
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
- Pathophysiology
- Primordial Drivers of Diabetes Heart Disease: Comprehensive Insights into Insulin Resistance
- Yajie Fan, Zhipeng Yan, Tingting Li, Aolin Li, Xinbiao Fan, Zhongwen Qi, Junping Zhang
- Diabetes Metab J. 2024;48(1):19-36. Published online January 3, 2024
- DOI: https://doi.org/10.4093/dmj.2023.0110
- 2,217 View
- 185 Download
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
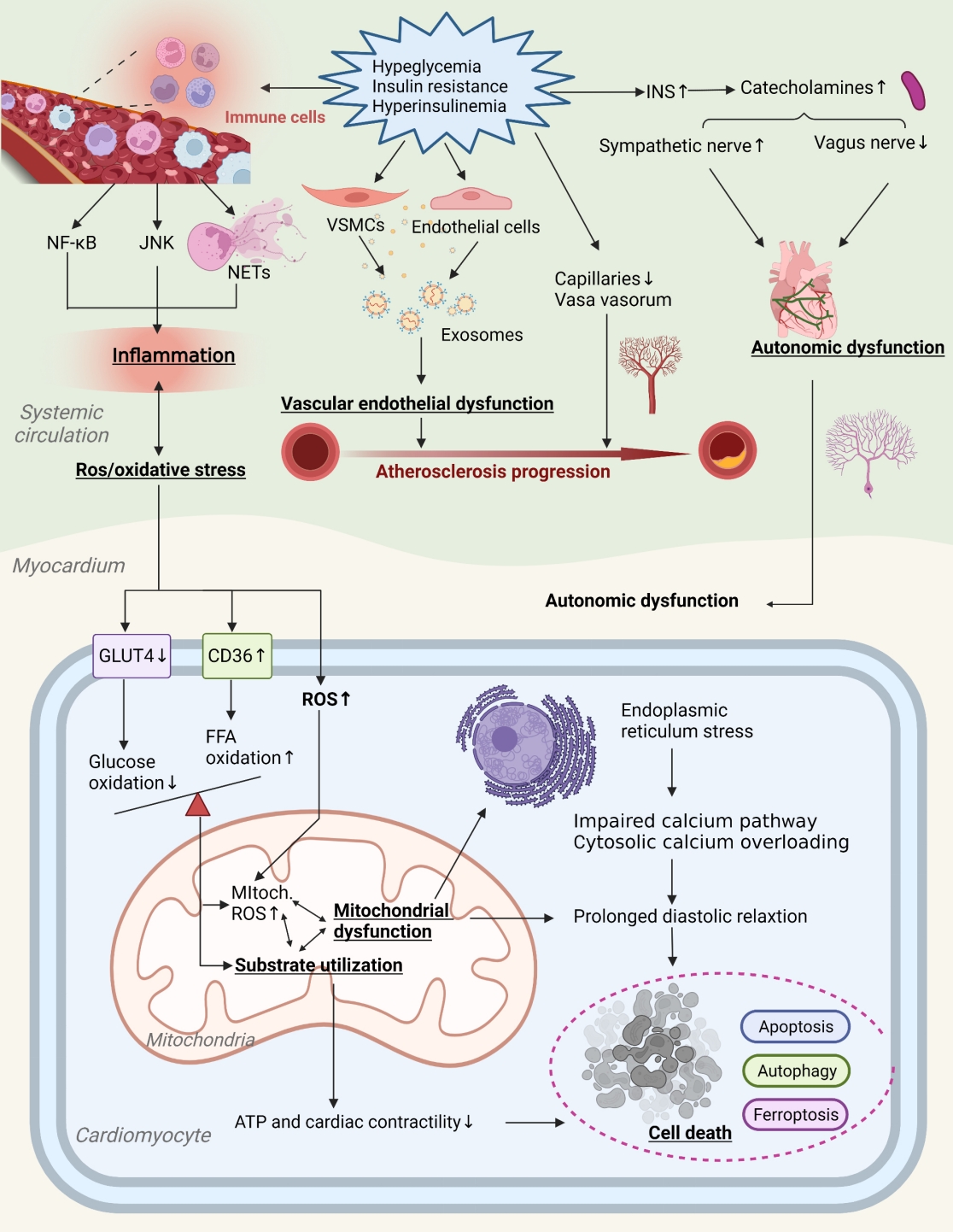
ePub - Insulin resistance has been regarded as a hallmark of diabetes heart disease (DHD). Numerous studies have shown that insulin resistance can affect blood circulation and myocardium, which indirectly cause cardiac hypertrophy and ventricular remodeling, participating in the pathogenesis of DHD. Meanwhile, hyperinsulinemia, hyperglycemia, and hyperlipidemia associated with insulin resistance can directly impair the metabolism and function of the heart. Targeting insulin resistance is a potential therapeutic strategy for the prevention of DHD. Currently, the role of insulin resistance in the pathogenic development of DHD is still under active research, as the pathological roles involved are complex and not yet fully understood, and the related therapeutic approaches are not well developed. In this review, we describe insulin resistance and add recent advances in the major pathological and physiological changes and underlying mechanisms by which insulin resistance leads to myocardial remodeling and dysfunction in the diabetic heart, including exosomal dysfunction, ferroptosis, and epigenetic factors. In addition, we discuss potential therapeutic approaches to improve insulin resistance and accelerate the development of cardiovascular protection drugs.
- Basic Research
- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition
- Ho Gyun Lee, Il Hyeon Jung, Byong Seo Park, Hye Rim Yang, Kwang Kon Kim, Thai Hien Tu, Jung-Yong Yeh, Sewon Lee, Sunggu Yang, Byung Ju Lee, Jae Geun Kim, Il Seong Nam-Goong
- Diabetes Metab J. 2023;47(6):784-795. Published online August 23, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0261
- 1,441 View
- 149 Download
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
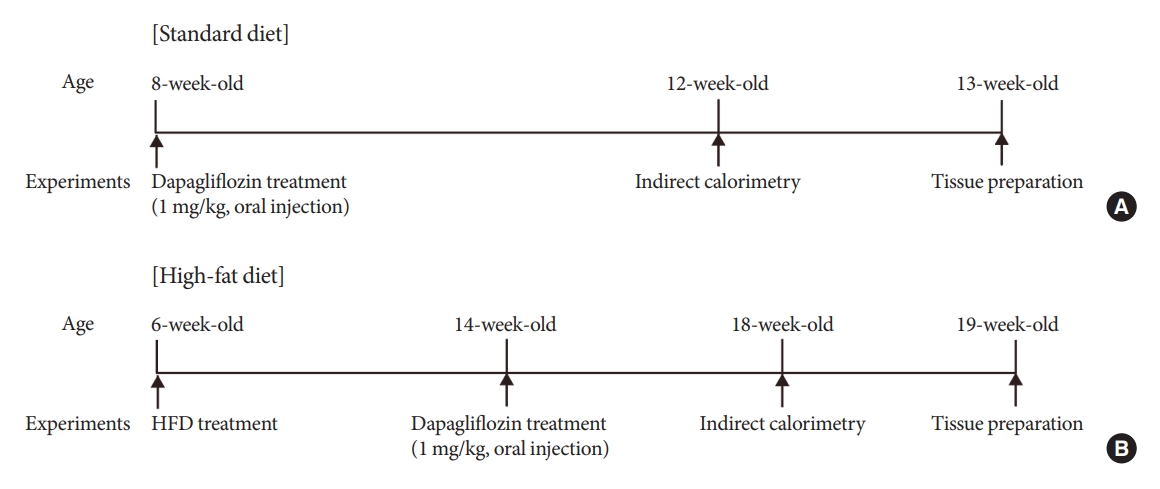
Sodium-glucose cotransporter 2 (SGLT-2) inhibitors are currently used to treat patients with diabetes. Previous studies have demonstrated that treatment with SGLT-2 inhibitors is accompanied by altered metabolic phenotypes. However, it has not been investigated whether the hypothalamic circuit participates in the development of the compensatory metabolic phenotypes triggered by the treatment with SGLT-2 inhibitors.
Methods
Mice were fed a standard diet or high-fat diet and treated with dapagliflozin, an SGLT-2 inhibitor. Food intake and energy expenditure were observed using indirect calorimetry system. The activity of hypothalamic neurons in response to dapagliflozin treatment was evaluated by immunohistochemistry with c-Fos antibody. Quantitative real-time polymerase chain reaction was performed to determine gene expression patterns in the hypothalamus of dapagliflozin-treated mice.
Results
Dapagliflozin-treated mice displayed enhanced food intake and reduced energy expenditure. Altered neuronal activities were observed in multiple hypothalamic nuclei in association with appetite regulation. Additionally, we found elevated immunosignals of agouti-related peptide neurons in the paraventricular nucleus of the hypothalamus.
Conclusion
This study suggests the functional involvement of the hypothalamus in the development of the compensatory metabolic phenotypes induced by SGLT-2 inhibitor treatment. -
Citations
Citations to this article as recorded by- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
Jae Hyun Bae
Diabetes & Metabolism Journal.2024; 48(1): 157. CrossRef - Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
Ho Gyun Lee, Il Hyeon Jung, Byong Seo Park, Hye Rim Yang, Kwang Kon Kim, Thai Hien Tu, Jung-Yong Yeh, Sewon Lee, Sunggu Yang, Byung Ju Lee, Jae Geun Kim, Il Seong Nam-Goong
Diabetes & Metabolism Journal.2024; 48(1): 159. CrossRef
- Altered Metabolic Phenotypes and Hypothalamic Neuronal Activity Triggered by Sodium-Glucose Cotransporter 2 Inhibition (Diabetes Metab J 2023;47:784-95)
- Basic Research
- Brown Fat as a Regulator of Systemic Metabolism beyond Thermogenesis
- Okamatsu-Ogura Yuko, Masayuki Saito
- Diabetes Metab J. 2021;45(6):840-852. Published online June 25, 2021
- DOI: https://doi.org/10.4093/dmj.2020.0291

- 9,135 View
- 510 Download
- 18 Crossref
-
 Graphical Abstract
Graphical Abstract
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub 
- Brown adipose tissue (BAT) is a specialized tissue for nonshivering thermogenesis to dissipate energy as heat. Although BAT research has long been limited mostly in small rodents, the rediscovery of metabolically active BAT in adult humans has dramatically promoted the translational studies on BAT in health and diseases. Moreover, several remarkable advancements have been made in brown fat biology over the past decade: The molecular and functional analyses of inducible thermogenic adipocytes (socalled beige adipocytes) arising from a developmentally different lineage from classical brown adipocytes have been accelerated. In addition to a well-established thermogenic activity of uncoupling protein 1 (UCP1), several alternative thermogenic mechanisms have been discovered, particularly in beige adipocytes. It has become clear that BAT influences other peripheral tissues and controls their functions and systemic homeostasis of energy and metabolic substrates, suggesting BAT as a metabolic regulator, other than for thermogenesis. This notion is supported by discovering that various paracrine and endocrine factors are secreted from BAT. We review the current understanding of BAT pathophysiology, particularly focusing on its role as a metabolic regulator in small rodents and also in humans.
-
Citations
Citations to this article as recorded by- Brown adipose tissue evaluation using water and triglyceride as indices by diffuse reflectance spectroscopy
Tomomi Iida, Yukio Ueda, Hideo Tsukada, Dai Fukumoto, Takafumi Hamaoka
Journal of Biophotonics.2024;[Epub] CrossRef - White-brown adipose tissue interplay in polycystic ovary syndrome: Therapeutic avenues
Khadijeh Abbasi, Reza Zarezadeh, Amir Valizadeh, Amir Mehdizadeh, Hamed Hamishehkar, Mohammad Nouri, Masoud Darabi
Biochemical Pharmacology.2024; 220: 116012. CrossRef - Brown Adipose Tissue, Batokines, and Bioactive Compounds in Foods: An Update
Fabiane Ferreira Martins, Bruna Cadete Martins, Ananda Vitoria Silva Teixeira, Matheus Ajackson, Vanessa Souza‐Mello, Julio Beltrame Daleprane
Molecular Nutrition & Food Research.2024;[Epub] CrossRef - Plasticity of Adipose Tissues: Interconversion among White, Brown, and Beige Fat and Its Role in Energy Homeostasis
Yanqiu Peng, Lixia Zhao, Min Li, Yunfei Liu, Yuke Shi, Jian Zhang
Biomolecules.2024; 14(4): 483. CrossRef - Homotaurine exhibits contrasting effects of DRD1-mediated thermogenesis-related regulators in C2C12 myoblasts and 3T3−L1 white adipocytes
Kiros Haddish, Jong Won Yun
Biotechnology and Bioprocess Engineering.2024;[Epub] CrossRef - The role of brown adipose tissue in mediating healthful longevity
Jie Zhang, Berhanu Geresu Kibret, Dorothy E. Vatner, Stephen F. Vatner
The Journal of Cardiovascular Aging.2024;[Epub] CrossRef - Thermogenic Brown Fat in Humans: Implications in Energy Homeostasis, Obesity and Metabolic Disorders
Masayuki Saito, Yuko Okamatsu-Ogura
The World Journal of Men's Health.2023; 41(3): 489. CrossRef - Interplay of skeletal muscle and adipose tissue: sarcopenic obesity
Min Jeong Park, Kyung Mook Choi
Metabolism.2023; 144: 155577. CrossRef - White adipose tissue undergoes browning during preweaning period in association with microbiota formation in mice
Anju Tsukada, Yuko Okamatsu-Ogura, Emi Futagawa, Yuki Habu, Natsumi Takahashi, Mira Kato-Suzuki, Yuko Kato, Satoshi Ishizuka, Kei Sonoyama, Kazuhiro Kimura
iScience.2023; 26(7): 107239. CrossRef - In situ fluorescence-photoacoustic measurement of the changes of brown adipose tissue in mice under hindlimb unloading
Baojie Gong, Jianxin Tang, Xiaoxiao Jiang, Zhe Zhang, Shiying Li, Hongjun Jin, Liming Nie, Guojia Huang
Journal of Applied Physiology.2023; 135(2): 251. CrossRef - Age-Related Expression Dynamics of Uncoupling Protein 1 in Adipose Tissues of ICR Outbred Mice during Postnatal Ontogenesis
A. V. Yakunenkov, E. I. Elsukova, I. O. Natochy
Journal of Evolutionary Biochemistry and Physiology.2023; 59(4): 1020. CrossRef - UNCOUPLING PROTEIN UCP1 EXPRESSION DYNAMICS IN ADIPOSE TISSUES OF THE OUTBRED ICR MICE IN POSTNATAL ONTOGENESIS
A. V. Yakunenkov, E. I. Elsukova, I. O. Natochy
Журнал эволюционной биохимии и физиологии.2023; 59(4): 255. CrossRef - Antibodies Regulate Dual-Function Enzyme IYD to Induce Functional Synergy between Metabolism and Thermogenesis
Sunghyun Kang, Hwan-Woo Park, Kyung Ho Han
International Journal of Molecular Sciences.2022; 23(14): 7834. CrossRef - Machine learning-featured Secretogranin V is a circulating diagnostic biomarker for pancreatic adenocarcinomas associated with adipopenia
Yunju Jo, Min-Kyung Yeo, Tam Dao, Jeongho Kwon, Hyon‐Seung Yi, Dongryeol Ryu
Frontiers in Oncology.2022;[Epub] CrossRef - Possible roles of exercise and apelin against pregnancy complications
Hamed Alizadeh Pahlavani
Frontiers in Endocrinology.2022;[Epub] CrossRef - Relationships between the expression of adipose genes and profiles of hospitalized dogs
Yukina Sugiyama, Fumie Shimokawa, Kazutoshi Sugiyama, Takashi Kobayashi, Yusuke Yamashita, Kei Kazama, Ken Onda, Masayuki Funaba, Masaru Murakami
Veterinary Research Communications.2022; 46(4): 1239. CrossRef - Garlic (Allium sativum L.) in diabetes and its complications: Recent advances in mechanisms of action
Yayi Jiang, Rensong Yue, Guojie Liu, Jun Liu, Bo Peng, Maoyi Yang, Lianxue Zhao, Zihan Li
Critical Reviews in Food Science and Nutrition.2022; : 1. CrossRef - Fruit of Gardenia jasminoides Induces Mitochondrial Activation and Non-Shivering Thermogenesis through Regulation of PPARγ
Woo Yong Park, Gahee Song, Ja Yeon Park, Kwan-Il Kim, Kwang Seok Ahn, Hyun Jeong Kwak, Jungtae Leem, Jae-Young Um, Jinbong Park
Antioxidants.2021; 10(9): 1418. CrossRef
- Brown adipose tissue evaluation using water and triglyceride as indices by diffuse reflectance spectroscopy
- Basic Research
- Revisiting the Bacterial Phylum Composition in Metabolic Diseases Focused on Host Energy Metabolism
- Yeonmi Lee, Hui-Young Lee
- Diabetes Metab J. 2020;44(5):658-667. Published online July 9, 2020
- DOI: https://doi.org/10.4093/dmj.2019.0220
- 9,049 View
- 131 Download
- 19 Web of Science
- 19 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
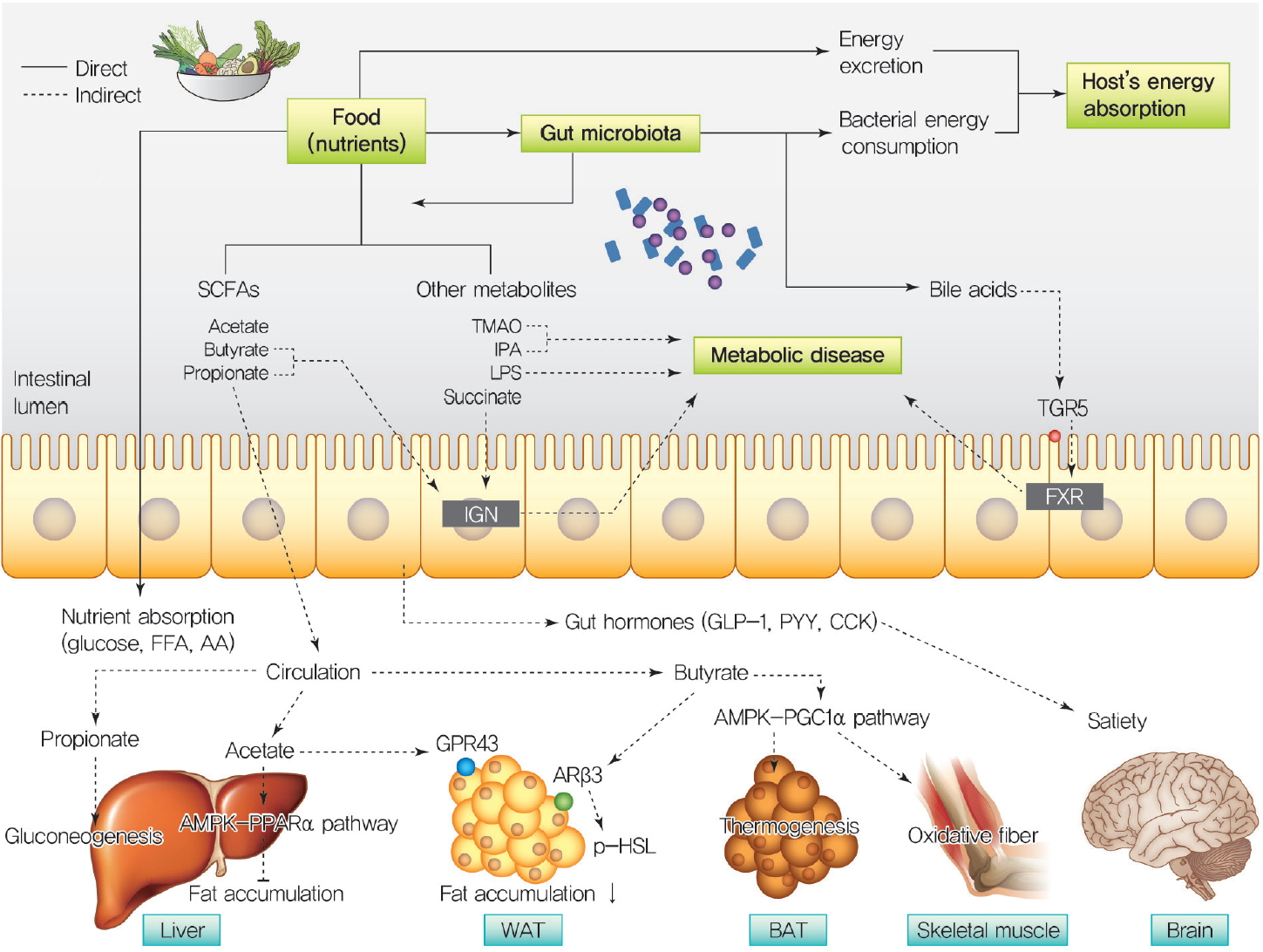
ePub Over a hundred billion bacteria are found in human intestines. This has emerged as an environmental factor in metabolic diseases, such as obesity and related diseases. The majority of these bacteria belong to two dominant phyla,
Bacteroidetes andFirmicutes . Since the ratio ofFirmicutes toBacteroidetes increases in people with obesity and in various animal models, it has been assumed that phylum composition causes the increase in occurrence of metabolic diseases over the past decade. However, this assumption has been challenged by recent studies that have found even an opposite association of phylum composition within metabolic diseases. Moreover, the gut microbiota affects host energy metabolism in various ways including production of metabolites and interaction with host intestinal cells to regulate signaling pathways that affect energy metabolism. However, the direct effect of gut bacteria on host energy intake, such as energy consumption by the bacteria itself and its effects on intestinal energy absorption, has been underestimated. This review aims to discuss whether increased ratio ofFirmicutes toBacteroidetes is associated with the development of metabolic diseases, and whether energy competition between the bacteria and host is a missing part of the mechanism linking gut microbiota to metabolic diseases.-
Citations
Citations to this article as recorded by- Behavior, intestinal health, and growth of small sea cucumbers Apostichopus japonicus in different color morphs
Peng Ding, Yushi Yu, Zihe Zhao, Xiang Li, Xiajing Wang, Huiyan Wang, Xiyuan Huang, Jun Ding, Chong Zhao
Marine Environmental Research.2024; 193: 106300. CrossRef - Traditional Chinese Medicine formula Dai-Zong-Fang alleviating hepatic steatosis in db/db mice via gut microbiota modulation
Li-Wei Zhang, Li-Li Zhu, Xiao-Yun Zhu, Shou-Qiang Fu, Xi-Ming Liu
Frontiers in Pharmacology.2024;[Epub] CrossRef - Repeated inoculation with rumen fluid accelerates the rumen bacterial transition with no benefit on production performance in postpartum Holstein dairy cows
Fanlin Kong, Feiran Wang, Yijia Zhang, Shuo Wang, Wei Wang, Shengli Li
Journal of Animal Science and Biotechnology.2024;[Epub] CrossRef - Identification of oncogenic signatures in the inflammatory colon of C57BL/6 mice fed a high-fat diet
Huawei Zeng, Bryan D. Safratowich, Wen-Hsing Cheng, Michael R. Bukowski
The Journal of Nutritional Biochemistry.2023; 111: 109188. CrossRef - Evaluation of the gut microbiome alterations in healthy rats after dietary exposure to different synthetic ZnO nanoparticles
Xinyi Zhu, Henghui Li, Liuzhu Zhou, Huijun Jiang, Minghui Ji, Jin Chen
Life Sciences.2023; 312: 121250. CrossRef - Microplastic-induced gut microbiota and serum metabolic disruption in Sprague-Dawley rats
Nan Zhao, Meirong Zhao, Hangbiao Jin
Environmental Pollution.2023; 320: 121071. CrossRef - Effects of neutral polysaccharide from Platycodon grandiflorum on high-fat diet-induced obesity via the regulation of gut microbiota and metabolites
Jing Song, Qin liu, Mengqi Hao, Xiaohu Zhai, Juan Chen
Frontiers in Endocrinology.2023;[Epub] CrossRef - Metabolite interactions between host and microbiota during health and disease: Which feeds the other?
Yan Zhang, Rui Chen, DuoDuo Zhang, Shuang Qi, Yan Liu
Biomedicine & Pharmacotherapy.2023; 160: 114295. CrossRef - Connecting Gut Microbial Diversity with Plasma Metabolome and Fecal Bile Acid Changes Induced by the Antibiotics Tobramycin and Colistin Sulfate
Aishwarya Murali, Varun Giri, Franziska Maria Zickgraf, Philipp Ternes, Hunter James Cameron, Saskia Sperber, Volker Haake, Peter Driemert, Hennicke Kamp, Dorothee Funk-Weyer, Shana J. Sturla, Ivonne M.C.M. Rietjens, Bennard van Ravenzwaay
Chemical Research in Toxicology.2023; 36(4): 598. CrossRef - Short-Term Alternate Feeding between Terrestrially Sourced Oil- and Fish Oil-Based Diets Modulates the Intestinal Microecology of Juvenile Turbot
Xiuhua Ma, Yaoyao Kong, Houguo Xu, Qingzhu Bi, Mengqing Liang, Kangsen Mai, Yanjiao Zhang
Biology.2023; 12(5): 650. CrossRef - Effects and action mechanisms of lotus leaf (Nelumbo nucifera) ethanol extract on gut microbes and obesity in high-fat diet-fed rats
Zhang Yanan, Ma Lu, Zhang Lu, Huo Jinhai, Wang Weiming
Frontiers in Nutrition.2023;[Epub] CrossRef - Effects of coffee with different roasting degrees on obesity and related metabolic disorders
Claudia I. Gamboa-Gómez, Laura J. Barragán-Zúñiga, Fernando Guerrero-Romero, Gerardo Martínez-Aguilar, José Luis Gónzalez, Almendra A. Valenzuela-Ramírez, Juan A. Rojas-Contreras, Monica Anese, Maribel Cervantes Flores, Marilisa Alongi
Journal of Functional Foods.2023; 111: 105889. CrossRef - Gut Microbiota and Bacterial Translocation in the Pathogenesis of Liver Fibrosis
Roman Maslennikov, Elena Poluektova, Oxana Zolnikova, Alla Sedova, Anastasia Kurbatova, Yulia Shulpekova, Natyia Dzhakhaya, Svetlana Kardasheva, Maria Nadinskaia, Elena Bueverova, Vladimir Nechaev, Anna Karchevskaya, Vladimir Ivashkin
International Journal of Molecular Sciences.2023; 24(22): 16502. CrossRef - Eugenol, A Major Component of Clove Oil, Attenuates Adiposity, and Modulates Gut Microbiota in High‐Fat Diet‐Fed Mice
Mengjie Li, Yuhan Zhao, Yanan Wang, Ruixuan Geng, Jingjing Fang, Seong‐Gook Kang, Kunlun Huang, Tao Tong
Molecular Nutrition & Food Research.2022;[Epub] CrossRef - Heimao tea polysaccharides ameliorate obesity by enhancing gut microbiota-dependent adipocytes thermogenesis in mice fed with high fat diet
Yu Wang, Ting Li, Yueyue Liu, Chengcheng Yang, Lei Liu, Xiangnan Zhang, Xingbin Yang
Food & Function.2022; 13(24): 13014. CrossRef - The Interplay of Sex Steroids, the Immune Response, and the Intestinal Microbiota
Fernanda Pace, Paula I. Watnick
Trends in Microbiology.2021; 29(9): 849. CrossRef - Heat stress on microbiota composition, barrier integrity, and
nutrient transport in gut, production performance, and its amelioration in farm
animals
Amlan Kumar Patra, Indrajit Kar
Journal of Animal Science and Technology.2021; 63(2): 211. CrossRef - Mechanisms linking gut microbial metabolites to insulin resistance
Hye Rim Jang, Hui-Young Lee
World Journal of Diabetes.2021; 12(6): 730. CrossRef - The impact of gut microbiota metabolites on cellular bioenergetics and cardiometabolic health
Lenka Tomasova, Marian Grman, Karol Ondrias, Marcin Ufnal
Nutrition & Metabolism.2021;[Epub] CrossRef
- Behavior, intestinal health, and growth of small sea cucumbers Apostichopus japonicus in different color morphs
- Basic Research
- Effects of Microbiota on the Treatment of Obesity with the Natural Product Celastrol in Rats
- Weiyue Hu, Lingling Wang, Guizhen Du, Quanquan Guan, Tianyu Dong, Ling Song, Yankai Xia, Xinru Wang
- Diabetes Metab J. 2020;44(5):747-763. Published online May 11, 2020
- DOI: https://doi.org/10.4093/dmj.2019.0124
- 9,361 View
- 136 Download
- 16 Web of Science
- 17 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub Background Obesity has become one of the most serious issues threatening the health of humankind, and we conducted this study to examine whether and how celastrol protects against obesity.
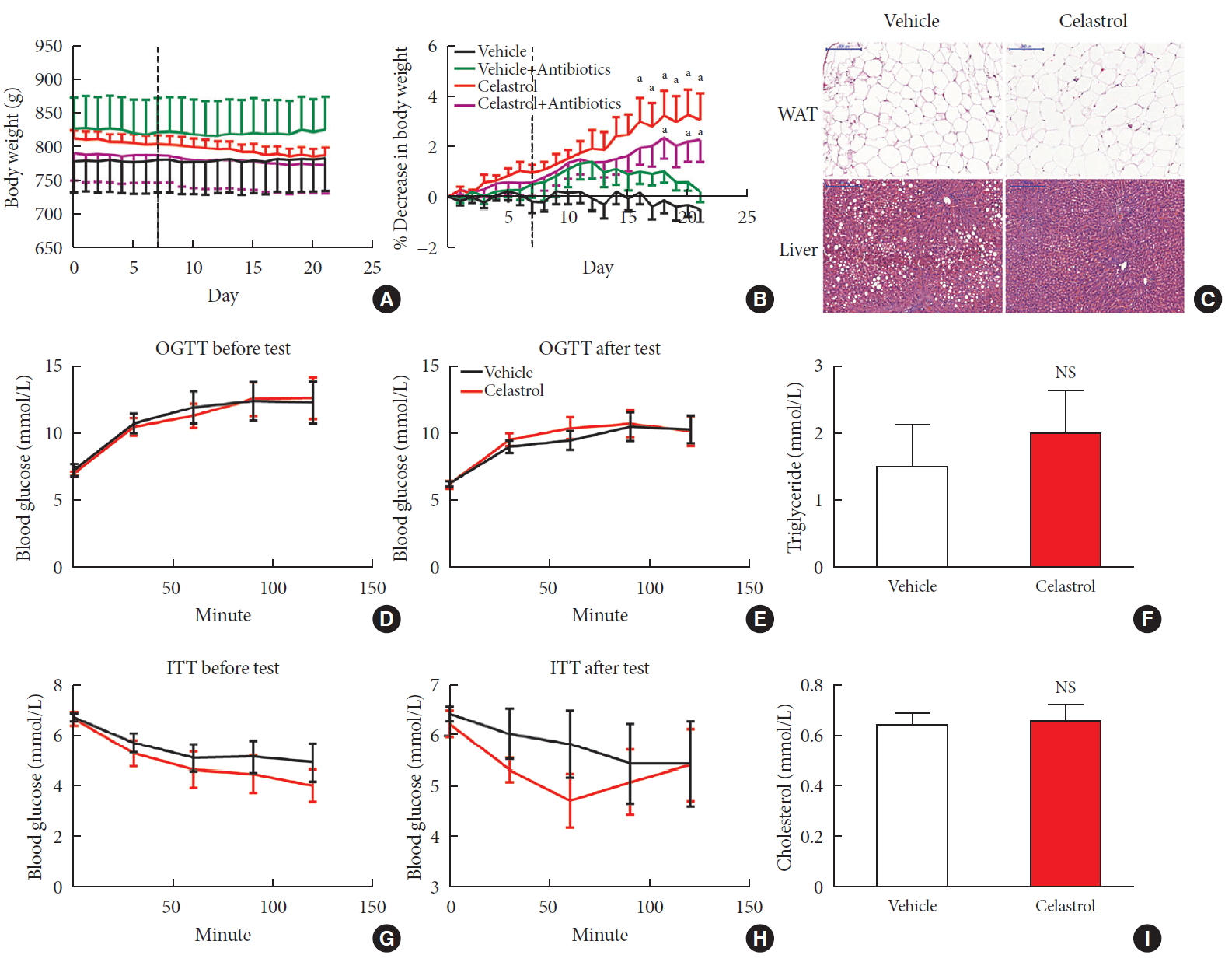
Methods We fed male Sprague-Dawley rats a high-fat diet and administered celastrol to obese rats for 3 weeks. By recording body weight (BW) and other measures, we identified the effective dose of celastrol for obesity treatment. Feces were collected to perform 16S rRNA sequencing, and hypothalami were extracted for transcriptome sequencing. We then treated leptin knockout rats with celastrol and explored the changes in energy metabolism. Male Institute of Cancer Research (ICR) mice were used to test the acute toxicity of celastrol.
Results We observed that celastrol reduced BW and promoted energy expenditure at a dose of 500 µg/kg BW but that food intake was not changed after administration. The diversity of the gut microbiota was improved, with an increased ratio of
Bacteroidetes toFirmicutes , and the gut microbiota played an important role in the anti-obesity effects of celastrol. Hypothalamic transcriptome analysis showed a significant enrichment of the leptin signaling pathway, and we found that celastrol significantly enhanced energy expenditure, which was mediated by the leptin signaling pathway. Acute lethal toxicity of celastrol was not observed at doses ranging from 0 to 62.5 mg/kg BW.Conclusion Our study revealed that celastrol decreased the BW of obese rats by enhancing energy expenditure but not by suppressing food intake and that this effect was mediated by the improvement of the gut microbiota and the activation of the hypothalamic leptin signaling pathway.
-
Citations
Citations to this article as recorded by- Natural compounds as obesity pharmacotherapies
Xin‐Yuan Zhao, Ji‐Qiu Wang, G. Gregory Neely, Yan‐Chuan Shi, Qiao‐Ping Wang
Phytotherapy Research.2024; 38(2): 797. CrossRef - Celastrol functions as an emerging manager of lipid metabolism: Mechanism and therapeutic potential
Jia Gu, Ya-Ning Shi, Neng Zhu, Hong-Fang Li, Chan-Juan Zhang, Li Qin
Biomedicine & Pharmacotherapy.2023; 164: 114981. CrossRef - Tripterygium hypoglaucum extract ameliorates adjuvant-induced arthritis in mice through the gut microbiota
Jianghui HU, Jimin NI, Junping ZHENG, Yanlei GUO, Yong YANG, Cheng YE, Xiongjie SUN, Hui XIA, Yanju LIU, Hongtao LIU
Chinese Journal of Natural Medicines.2023; 21(10): 730. CrossRef - Health improvements of type 2 diabetic patients through diet and diet plus fecal microbiota transplantation
Lili Su, Zhifan Hong, Tong Zhou, Yuanyuan Jian, Mei Xu, Xuanping Zhang, Xiaoyan Zhu, Jiayin Wang
Scientific Reports.2022;[Epub] CrossRef - Tripterygium hypoglaucum (Levl.) Hutch: A systematic review of its traditional uses, botany, phytochemistry, pharmacology, pharmacokinetics and toxicology
Jiangping Wei, Liyun Chen, Sijia Gao, Jirui Wang, Yunhong Wang, Zhiwei Zhang, Yuyu Zhang, Xiaomei Zhang, Yong Yang, Dajian Yang
Pharmacological Research - Modern Chinese Medicine.2022; 3: 100094. CrossRef - Celastrol: An Update on Its Hepatoprotective Properties and the Linked Molecular Mechanisms
Mengzhen Li, Faren Xie, Lu Wang, Guoxue Zhu, Lian-Wen Qi, Shujun Jiang
Frontiers in Pharmacology.2022;[Epub] CrossRef - Celastrol inhibits the proliferation and migration of MCF-7 cells through the leptin-triggered PI3K/AKT pathway
Pingping Chen, Bin Wang, Meng Li, Chunxue Cui, Fei Liu, Yonggang Gao
Computational and Structural Biotechnology Journal.2022; 20: 3173. CrossRef - Investigating Celastrol’s Anti-DCM Targets and Mechanisms via Network Pharmacology and Experimental Validation
Rui Xi, Yongxin Wan, Lihong Yang, Jingying Zhang, Liu Yang, Shuai Yang, Rui Chai, Fengchen Mu, Qiting Sun, Rui Yan, Zhifang Wu, Sijin Li, Zhijun Liao
BioMed Research International.2022; 2022: 1. CrossRef - Celastrol inhibits TXNIP expression to protect pancreatic β cells in diabetic mice
Si-wei Wang, Tian Lan, Fang Zheng, Hui Huang, Hang-fei Chen, Qi Wu, Feng Zhang
Phytomedicine.2022; 104: 154316. CrossRef - Celastrol: A Promising Agent Fighting against Cardiovascular Diseases
Zhexi Li, Jingyi Zhang, Xulei Duan, Guoan Zhao, Min Zhang
Antioxidants.2022; 11(8): 1597. CrossRef - Celastrol: A lead compound that inhibits SARS‐CoV‐2 replication, the activity of viral and human cysteine proteases, and virus‐induced IL‐6 secretion
Carlos A. Fuzo, Ronaldo B. Martins, Thais F. C. Fraga‐Silva, Martin K. Amstalden, Thais Canassa De Leo, Juliano P. Souza, Thais M. Lima, Lucia H. Faccioli, Débora Noma Okamoto, Maria Aparecida Juliano, Suzelei C. França, Luiz Juliano, Vania L. D. Bonato,
Drug Development Research.2022; 83(7): 1623. CrossRef - In vitro activity of celastrol in combination with thymol against carbapenem-resistant Klebsiella pneumoniae isolates
Mahmoud Saad Abdel-Halim, Momen Askoura, Basem Mansour, Galal Yahya, Amira M. El-Ganiny
The Journal of Antibiotics.2022; 75(12): 679. CrossRef - Celastrol alleviates metabolic disturbance in high‐fat diet‐induced obese mice through increasing energy expenditure by ameliorating metabolic inflammation
Xueping Yang, Fan Wu, Lingli Li, Ernest C. Lynch, Linglin Xie, Yan Zhao, Ke Fang, Jingbin Li, Jinlong Luo, Lijun Xu, Xin Zou, Fuer Lu, Guang Chen
Phytotherapy Research.2021; 35(1): 297. CrossRef - Celastrol in metabolic diseases: Progress and application prospects
Shaohua Xu, Yaqian Feng, Weishen He, Wen Xu, Wei Xu, Hongjun Yang, Xianyu Li
Pharmacological Research.2021; 167: 105572. CrossRef - The Anti-Obesity Effect of Traditional Chinese Medicine on Lipid Metabolism
Qijing Fan, Furong Xu, Bin Liang, Xiaoju Zou
Frontiers in Pharmacology.2021;[Epub] CrossRef - Serum Metabolome Mediates the Antiobesity Effect of Celastrol-Induced Gut Microbial Alterations
Shaohua Xu, Liwei Lyu, Huaichang Zhu, Xiaoqiang Huang, Wei Xu, Wen Xu, Yaqian Feng, Yong Fan
Journal of Proteome Research.2021; 20(10): 4840. CrossRef - Interrelated Mechanism by Which the Methide Quinone Celastrol, Obtained from the Roots of Tripterygium wilfordii, Inhibits Main Protease 3CLpro of COVID-19 and Acts as Superoxide Radical Scavenger
Francesco Caruso, Manrose Singh, Stuart Belli, Molly Berinato, Miriam Rossi
International Journal of Molecular Sciences.2020; 21(23): 9266. CrossRef
- Natural compounds as obesity pharmacotherapies
- Basic Research
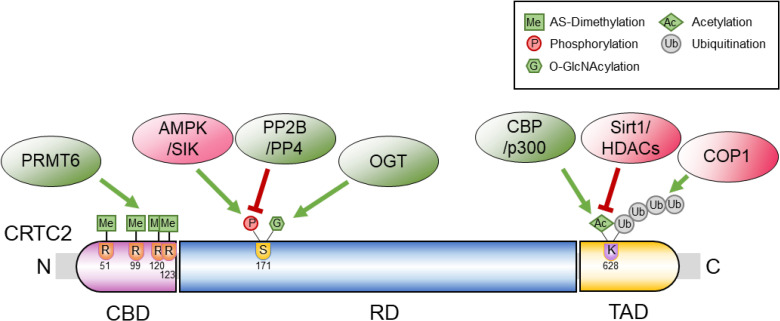
- Role of CRTC2 in Metabolic Homeostasis: Key Regulator of Whole-Body Energy Metabolism?
- Hye-Sook Han, Yongmin Kwon, Seung-Hoi Koo
- Diabetes Metab J. 2020;44(4):498-508. Published online March 5, 2020
- DOI: https://doi.org/10.4093/dmj.2019.0200
- 6,977 View
- 163 Download
- 14 Web of Science
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub Cyclic adenosine monophosphate (cAMP) signaling is critical for regulating metabolic homeostasis in mammals. In particular, transcriptional regulation by cAMP response element-binding protein (CREB) and its coactivator, CREB-regulated transcription coactivator (CRTC), is essential for controlling the expression of critical enzymes in the metabolic process, leading to more chronic changes in metabolic flux. Among the CRTC isoforms, CRTC2 is predominantly expressed in peripheral tissues and has been shown to be associated with various metabolic pathways in tissue-specific manners. While initial reports showed the physiological role of CRTC2 in regulating gluconeogenesis in the liver, recent studies have further delineated the role of this transcriptional coactivator in the regulation of glucose and lipid metabolism in various tissues, including the liver, pancreatic islets, endocrine tissues of the small intestines, and adipose tissues. In this review, we discuss recent studies that have utilized knockout mouse models to delineate the role of CRTC2 in the regulation of metabolic homeostasis.
-
Citations
Citations to this article as recorded by- Integration of genomic and transcriptomic data of inbred mouse models for polygenic obesity and leanness revealed “obese” and “lean” candidate alleles in polyadenylation signals
Martin Šimon, Špela Mikec, Nicholas M. Morton, Santosh S. Atanur, Simon Horvat, Tanja Kunej
Gene Reports.2024; 35: 101903. CrossRef - Mylabris phalerata induces the apoptosis and cell cycle delay in HCC, and potentiates the effect of sorafenib based on the molecular and network pharmacology approach
Young Woo Kim, Seon Been Bak, Su Youn Baek, Il Kon Kim, Won-Yung Lee, Un-Jung Yun, Kwang-Il Park
Molecular & Cellular Toxicology.2023; 19(4): 731. CrossRef - Emerging Role of SMILE in Liver Metabolism
Nanthini Sadasivam, Kamalakannan Radhakrishnan, Hueng-Sik Choi, Don-Kyu Kim
International Journal of Molecular Sciences.2023; 24(3): 2907. CrossRef - PIMT regulates hepatic gluconeogenesis in mice
Bandish Kapadia, Soma Behera, Sireesh T. Kumar, Tapan Shah, Rebecca Kristina Edwin, Phanithi Prakash Babu, Partha Chakrabarti, Kishore V.L. Parsa, Parimal Misra
iScience.2023; 26(3): 106120. CrossRef - Biological functions of CRTC2 and its role in metabolism-related diseases
Hong-Yu Zheng, Yan-Xia Wang, Kun Zhou, Hai-Lin Xie, Zhong Ren, Hui-Ting Liu, Yang-Shao Ou, Zhi-Xiang Zhou, Zhi-Sheng Jiang
Journal of Cell Communication and Signaling.2023; 17(3): 495. CrossRef - An insulin-regulated arrestin domain protein controls hepatic glucagon action
Sezin Dagdeviren, Megan F. Hoang, Mohsen Sarikhani, Vanessa Meier, Jake C. Benoit, Marinna C. Okawa, Veronika Y. Melnik, Elisabeth M. Ricci-Blair, Natalie Foot, Randall H. Friedline, Xiaodi Hu, Lauren A. Tauer, Arvind Srinivasan, Maxim B. Prigozhin, Sudha
Journal of Biological Chemistry.2023; 299(8): 105045. CrossRef - The Pleiotropic Face of CREB Family Transcription Factors
Md. Arifur Rahman Chowdhury, Jungeun An, Sangyun Jeong
Molecules and Cells.2023; 46(7): 399. CrossRef - It is a branched road to adipose tissue aging
N. Touitou, B. Lerrer, H. Y. Cohen
Nature Aging.2023; 3(8): 911. CrossRef - Impaired BCAA catabolism in adipose tissues promotes age-associated metabolic derangement
Hye-Sook Han, Eunyong Ahn, Eun Seo Park, Tom Huh, Seri Choi, Yongmin Kwon, Byeong Hun Choi, Jueun Lee, Yoon Ha Choi, Yujin L. Jeong, Gwang Bin Lee, Minji Kim, Je Kyung Seong, Hyun Mu Shin, Hang-Rae Kim, Myeong Hee Moon, Jong Kyoung Kim, Geum-Sook Hwang, S
Nature Aging.2023; 3(8): 982. CrossRef - Exploring the diagnostic value, prognostic value, and biological functions of NPC gene family members in hepatocellular carcinoma based on a multi-omics analysis
Keheng Chen, Xin Zhang, Huixin Peng, Fengdie Huang, Guangyu Sun, Qijiang Xu, Lusheng Liao, Zhiyong Xing, Yanping Zhong, Zhichao Fang, Meihua Liao, Shihua Luo, Wencheng Chen, Mingyou Dong
Functional & Integrative Genomics.2023;[Epub] CrossRef - MicroRNA regulation of AMPK in nonalcoholic fatty liver disease
Hao Sun, Jongsook Kim Kemper
Experimental & Molecular Medicine.2023; 55(9): 1974. CrossRef - Serine active site containing protein 1 depletion alters lipid metabolism and protects against high fat diet-induced obesity in mice
Miaomiao Du, Xueyun Li, Fangyi Xiao, Yinxu Fu, Yu Shi, Sihan Guo, Lifang Chen, Lu Shen, Lan Wang, Huang Cheng, Hao Li, Anran Xie, Yaping Zhou, Kaiqiang Yang, Hezhi Fang, Jianxin Lyu, Qiongya Zhao
Metabolism.2022; 134: 155244. CrossRef - cAMP Signaling in Cancer: A PKA-CREB and EPAC-Centric Approach
Muhammad Bilal Ahmed, Abdullah A. A. Alghamdi, Salman Ul Islam, Joon-Seok Lee, Young-Sup Lee
Cells.2022; 11(13): 2020. CrossRef - Hepatic Sam68 Regulates Systemic Glucose Homeostasis and Insulin Sensitivity
Aijun Qiao, Wenxia Ma, Ying Jiang, Chaoshan Han, Baolong Yan, Junlan Zhou, Gangjian Qin
International Journal of Molecular Sciences.2022; 23(19): 11469. CrossRef - The Role of Small Heterodimer Partner-Interacting Leucine Zipper
(SMILE) as a Transcriptional Corepressor in Hepatic Glucose and Lipid
Metabolism
Woo-Ram Park, Byungyoon Choi, Nanthini Sadasivam, Don-Kyu Kim
Trends in Agriculture & Life Sciences.2022; 60: 7. CrossRef - AMPK Localization: A Key to Differential Energy Regulation
Qonita Afinanisa, Min Kyung Cho, Hyun-A Seong
International Journal of Molecular Sciences.2021; 22(20): 10921. CrossRef
- Integration of genomic and transcriptomic data of inbred mouse models for polygenic obesity and leanness revealed “obese” and “lean” candidate alleles in polyadenylation signals
- Obesity and Metabolic Syndrome
- Two Faces of White Adipose Tissue with Heterogeneous Adipogenic Progenitors
- Injae Hwang, Jae Bum Kim
- Diabetes Metab J. 2019;43(6):752-762. Published online December 26, 2019
- DOI: https://doi.org/10.4093/dmj.2019.0174
- 8,943 View
- 181 Download
- 36 Web of Science
- 38 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Chronic energy surplus increases body fat, leading to obesity. Since obesity is closely associated with most metabolic complications, pathophysiological roles of adipose tissue in obesity have been intensively studied. White adipose tissue is largely divided into subcutaneous adipose tissue (SAT) and visceral adipose tissue (VAT). These two white adipose tissues are similar in their appearance and lipid storage functions. Nonetheless, emerging evidence has suggested that SAT and VAT have different characteristics and functional roles in metabolic regulation. It is likely that there are intrinsic differences between VAT and SAT. In diet-induced obese animal models, it has been reported that adipogenic progenitors in VAT rapidly proliferate and differentiate into adipocytes. In obesity, VAT exhibits elevated inflammatory responses, which are less prevalent in SAT. On the other hand, SAT has metabolically beneficial effects. In this review, we introduce recent studies that focus on cellular and molecular components modulating adipogenesis and immune responses in SAT and VAT. Given that these two fat depots show different functions and characteristics depending on the nutritional status, it is feasible to postulate that SAT and VAT have different developmental origins with distinct adipogenic progenitors, which would be a key determining factor for the response and accommodation to metabolic input for energy homeostasis.
-
Citations
Citations to this article as recorded by- Lipodystrophy as a target to delay premature aging
Daniela G. Costa, Marisa Ferreira-Marques, Cláudia Cavadas
Trends in Endocrinology & Metabolism.2024; 35(2): 97. CrossRef - Subcutaneous fat predicts bone metastasis in breast cancer: A novel multimodality-based deep learning model
Shidi Miao, Haobo Jia, Wenjuan Huang, Ke Cheng, Wenjin Zhou, Ruitao Wang
Cancer Biomarkers.2024; 39(3): 171. CrossRef - Association between abdominal adiposity and clinical outcomes in patients with acute ischemic stroke
Kayo Wakisaka, Ryu Matsuo, Fumi Irie, Yoshinobu Wakisaka, Tetsuro Ago, Masahiro Kamouchi, Takanari Kitazono, Masaki Mogi
PLOS ONE.2024; 19(1): e0296833. CrossRef - NOTCH1 as a Negative Regulator of Avian Adipocyte Differentiation: Implications for Fat Deposition
Zheng Wang, Yue Su, Mingyu Zhao, Zhenhua Ma, Jianhui Li, Zhuocheng Hou, Huifeng Li
Animals.2024; 14(4): 585. CrossRef - Green tea beneficial effects involve changes in the profile of immune cells in the adipose tissue of obese mice
Kaue Tognolli, Victoria Silva, Celso Pereira Batista Sousa-Filho, Claudia Andrea Lima Cardoso, Renata Gorjão, Rosemari Otton
European Journal of Nutrition.2023; 62(1): 321. CrossRef - Mechanic Insight into the Distinct and Common Roles of Ovariectomy Versus Adrenalectomy on Adipose Tissue Remodeling in Female Mice
Weihao Chen, Fengyan Meng, Xianyin Zeng, Xiaohan Cao, Guixian Bu, Xiaogang Du, Guozhi Yu, Fanli Kong, Yunkun Li, Tian Gan, Xingfa Han
International Journal of Molecular Sciences.2023; 24(3): 2308. CrossRef - High-fat diet consumption by male rat offspring of obese mothers exacerbates adipose tissue hypertrophy and metabolic alterations in adult life
Guadalupe L. Rodríguez-González, Sergio De Los Santos, Dayana Méndez-Sánchez, Luis A. Reyes-Castro, Carlos A. Ibáñez, Patricia Canto, Elena Zambrano
British Journal of Nutrition.2023; 130(5): 783. CrossRef - Obesity and the risk of cardiometabolic diseases
Pedro L. Valenzuela, Pedro Carrera-Bastos, Adrián Castillo-García, Daniel E. Lieberman, Alejandro Santos-Lozano, Alejandro Lucia
Nature Reviews Cardiology.2023; 20(7): 475. CrossRef - Abdominal fat and muscle distributions in different stages of colorectal cancer
Jun Han, Xinyang Liu, Min Tang, Fan Yang, Zuoyou Ding, Guohao Wu
BMC Cancer.2023;[Epub] CrossRef - Expression Analysis of hsa-miR-181a-5p, hsa-miR-143-3p, hsa-miR-132-3p and hsa-miR-23a-3p as Biomarkers in Colorectal Cancer—Relationship to the Body Mass Index
Sofía Elena Tesolato, Daniel González-Gamo, Ana Barabash, Paula Claver, Sofía Cristina de la Serna, Inmaculada Domínguez-Serrano, Jana Dziakova, Carmen de Juan, Antonio José Torres, Pilar Iniesta
Cancers.2023; 15(13): 3324. CrossRef - Lower subcutaneous fat index predicts bone metastasis in breast cancer
Wen Wang, Wen-Juan Huang, Ping-Ping Liu, Shuang Fu, Meng-Lin Zhang, Xin Zhang, Rui-Tao Wang, Yuan-Xi Huang
Cancer Biomarkers.2023; 38(1): 121. CrossRef - RabGAP AS160/TBC1D4 deficiency increases long-chain fatty acid transport but has little additional effect on obesity and metabolic syndrome in ADMSCs-derived adipocytes of morbidly obese women
Agnieszka Mikłosz, Bartłomiej Łukaszuk, Elżbieta Supruniuk, Kamil Grubczak, Magdalena Kusaczuk, Adrian Chabowski
Frontiers in Molecular Biosciences.2023;[Epub] CrossRef - White adipose tissue: Distribution, molecular insights of impaired expandability, and its implication in fatty liver disease
Griselda Rabadán-Chávez, Rocío I. Díaz de la Garza, Daniel A. Jacobo-Velázquez
Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease.2023; 1869(8): 166853. CrossRef - Histological pattern and gene expression profiling of thyroid tissue in subjects with obesity
A. Basolo, A. M. Poma, R. Giannini, G. Ceccarini, C. Pelosini, P. Fierabracci, M. U. Castany, S. Bechi Genzano, C. E. Ambrosini, G. Materazzi, L. Chiovato, F. Basolo, F. Santini, L. Torregrossa
Journal of Endocrinological Investigation.2022; 45(2): 413. CrossRef - Predictors of non-alcoholic fatty liver disease in children
Menglong Li, Wen Shu, Jiawulan Zunong, Nubiya Amaerjiang, Huidi Xiao, Dan Li, Sten H. Vermund, Yifei Hu
Pediatric Research.2022; 92(1): 322. CrossRef - Distinct properties of adipose stem cell subpopulations determine fat depot-specific characteristics
Hahn Nahmgoong, Yong Geun Jeon, Eun Seo Park, Yoon Ha Choi, Sang Mun Han, Jeu Park, Yul Ji, Jee Hyung Sohn, Ji Seul Han, Ye Young Kim, Injae Hwang, Yun Kyung Lee, Jin Young Huh, Sung Sik Choe, Tae Jung Oh, Sung Hee Choi, Jong Kyoung Kim, Jae Bum Kim
Cell Metabolism.2022; 34(3): 458. CrossRef - WT1 in Adipose Tissue: From Development to Adult Physiology
Karin M. Kirschner, Holger Scholz
Frontiers in Cell and Developmental Biology.2022;[Epub] CrossRef - Shall We Begin the Voyage of Adipose Tissue Exploration?
Yong Geun Jeon
Molecules and Cells.2022; 45(6): 362. CrossRef - Mammalian adipogenesis regulator (Areg) cells use retinoic acid signalling to be non‐ and anti‐adipogenic in age‐dependent manner
Magda Zachara, Pernille Y Rainer, Horia Hashimi, Julie M Russeil, Daniel Alpern, Radiana Ferrero, Maria Litovchenko, Bart Deplancke
The EMBO Journal.2022;[Epub] CrossRef - The fates of different types of adipose tissue after transplantation in mice
Shenglu Jiang, Jiayan Lin, Qian Zhang, Yunjun Liao, Feng Lu, Junrong Cai
The FASEB Journal.2022;[Epub] CrossRef - Analysis of different adipose depot gene expression in cachectic patients with gastric cancer
Jun Han, Zuoyou Ding, Qiulin Zhuang, Lei Shen, Fan Yang, Szechun Sah, Guohao Wu
Nutrition & Metabolism.2022;[Epub] CrossRef - Adipose Stromal/Stem Cell-Derived Extracellular Vesicles: Potential Next-Generation Anti-Obesity Agents
Mariachiara Zuccarini, Patricia Giuliani, Valentina Di Liberto, Monica Frinchi, Francesco Caciagli, Vanni Caruso, Renata Ciccarelli, Giuseppa Mudò, Patrizia Di Iorio
International Journal of Molecular Sciences.2022; 23(3): 1543. CrossRef - Is There a Link between Obesity Indices and Skin Autofluorescence? A Response from the ILERVAS Project
Enric Sánchez, Marta Sánchez, Carolina López-Cano, Marcelino Bermúdez-López, José Manuel Valdivielso, Cristina Farràs-Sallés, Reinald Pamplona, Gerard Torres, Dídac Mauricio, Eva Castro, Elvira Fernández, Albert Lecube
Nutrients.2022; 15(1): 203. CrossRef - Insights behind the Relationship between Colorectal Cancer and Obesity: Is Visceral Adipose Tissue the Missing Link?
Alice Chaplin, Ramon Maria Rodriguez, Juan José Segura-Sampedro, Aina Ochogavía-Seguí, Dora Romaguera, Gwendolyn Barceló-Coblijn
International Journal of Molecular Sciences.2022; 23(21): 13128. CrossRef - Potential effects of nutrition-based weight loss therapies in reversing obesity-related breast cancer epigenetic marks
Paula M. Lorenzo, Ana B. Crujeiras
Food & Function.2021; 12(4): 1402. CrossRef - Metabolomic Profiles in Adipocytes Differentiated from Adipose-Derived Stem Cells Following Exercise Training or High-Fat Diet
Seita Osawa, Hisashi Kato, Yuki Maeda, Hisashi Takakura, Junetsu Ogasawara, Tetsuya Izawa
International Journal of Molecular Sciences.2021; 22(2): 966. CrossRef - Adipocytes Are the Control Tower That Manages Adipose Tissue Immunity by Regulating Lipid Metabolism
Jeu Park, Jee Hyung Sohn, Sang Mun Han, Yoon Jeong Park, Jin Young Huh, Sung Sik Choe, Jae Bum Kim
Frontiers in Immunology.2021;[Epub] CrossRef - Ceramides and Sphingosino-1-Phosphate in Obesity
Ilona Juchnicka, Mariusz Kuźmicki, Jacek Szamatowicz
Frontiers in Endocrinology.2021;[Epub] CrossRef - Contribution of Adipose Tissue to the Chronic Immune Activation and Inflammation Associated With HIV Infection and Its Treatment
Christine Bourgeois, Jennifer Gorwood, Anaelle Olivo, Laura Le Pelletier, Jacqueline Capeau, Olivier Lambotte, Véronique Béréziat, Claire Lagathu
Frontiers in Immunology.2021;[Epub] CrossRef - Subcutaneous, but not visceral, adipose tissue as a marker for prognosis in gastric cancer patients with cachexia
Jun Han, Min Tang, Chaocheng Lu, Lei Shen, Jiaqi She, Guohao Wu
Clinical Nutrition.2021; 40(9): 5156. CrossRef - miR-410-3P inhibits adipocyte differentiation by targeting IRS-1 in cancer-associated cachexia patients
Diya Sun, Zuoyou Ding, Lei Shen, Fan Yang, Jun Han, Guohao Wu
Lipids in Health and Disease.2021;[Epub] CrossRef - Multipotent Stromal Cells from Subcutaneous Adipose Tissue of Normal Weight and Obese Subjects: Modulation of Their Adipogenic Differentiation by Adenosine A1 Receptor Ligands
Mariachiara Zuccarini, Catia Lambertucci, Marzia Carluccio, Patricia Giuliani, Maurizio Ronci, Andrea Spinaci, Rosaria Volpini, Renata Ciccarelli, Patrizia Di Iorio
Cells.2021; 10(12): 3560. CrossRef - Obesity: The Crossroads of Opinion, Knowledge, and Opportunity
L. A. Ruyatkina, D. S. Ruyatkin
Meditsinskiy sovet = Medical Council.2020; (7): 108. CrossRef - Sex Differences in Long-term Metabolic Effects of Maternal Resveratrol Intake in Adult Rat Offspring
Purificación Ros, Francisca Díaz, Alejandra Freire-Regatillo, Pilar Argente-Arizón, Vicente Barrios, Jesús Argente, Julie A Chowen
Endocrinology.2020;[Epub] CrossRef - Adipose stem cells in obesity: challenges and opportunities
Sunhye Shin, Asma S. El-Sabbagh, Brandon E. Lukas, Skylar J. Tanneberger, Yuwei Jiang
Bioscience Reports.2020;[Epub] CrossRef - Clinical and pathogenetic rationale for the prevention and treatment of obesity
O.M. Korzh
Shidnoevropejskij zurnal vnutrisnoi ta simejnoi medicini.2020; 2020(2): 146. CrossRef - OBESITY: CLINICAL AND PATHOGENETIC JUSTIFICATION OF PREVENTION AND TREATMENT
O. M. Korzh
International Medical Journal.2020; (2): 5. CrossRef - The Effect and Mechanism of Subcutaneous and Visceral Adipose Tissue Loss on Gastric Cancer Patients With Cachexia
Jun Han, Min Tang, Guyue Zhang, Chaocheng Lu, Jiaqi She, Guohao Wu
SSRN Electronic Journal .2020;[Epub] CrossRef
- Lipodystrophy as a target to delay premature aging
- Mitochondrial Dysfunction in Diabetic Cardiomyopathy.
- Ji Hyun Ahn, Jae Taek Kim
- Korean Diabetes J. 2008;32(6):467-473. Published online December 1, 2008
- DOI: https://doi.org/10.4093/kdj.2008.32.6.467
- 2,111 View
- 30 Download
-
 Abstract
Abstract
 PDF
PDF - Metabolic syndrome and diabetes are associated with increased risk of cardiac dysfunction independently of underlying coronary artery disease. The underlying pathogenesis is partially understood but accumulating evidence suggests that alterations of cardiac energy metabolism might contribute to the development of contractile dysfunction. Recent findings suggest that myocardial mitochondrial dysfunction may play an important role in the pathogenesis of cardiac contractile dysfunction in type 2 diabetes. This review is focused on evaluating mechanisms for the mitochondrial abnormalities that may be involved in the development and progression of cardiac dysfunction in diabetes.

 KDA
KDA
 First
First Prev
Prev





