- Skip Navigation
- Skip to contents

- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 41(6); 2017 > Article
-
Original ArticleObesity and Metabolic Syndrome Beneficial Effects of Aerobic Exercise Training Combined with Rosiglitazone on Glucose Metabolism in Otsuka Long Evans Tokushima Fatty Rats
-
Shan-Ji Piao1,2, So Hun Kim1, Young Ju Suh3, Seong-Bin Hong1, Seong Hee Ahn1, Da Hae Seo1, In-Sun Park4
 , Moonsuk Nam1
, Moonsuk Nam1
-
Diabetes & Metabolism Journal 2017;41(6):474-485.
DOI: https://doi.org/10.4093/dmj.2017.41.6.474
Published online: November 15, 2017
1Department of Internal Medicine, Inha University School of Medicine, Incheon, Korea.
2Qingdao Endocrine and Diabetes Hospital, Qingdao, China.
3Department of Biomedical Sciences, Inha University School of Medicine, Incheon, Korea.
4Department of Anatomy, Inha University School of Medicine, Incheon, Korea.
- Corresponding authors: Moonsuk Nam. Department of Internal Medicine, Inha University School of Medicine, 27 Inhang-ro, Jung-gu, Incheon 22332, Korea. namms@inha.ac.kr
- In-Sun Park. Department of Anatomy, Inha University School of Medicine, 100 Inha-ro, Nam-gu, Incheon 22212, Korea. sunpark@inha.ac.kr
- *Shan-Ji Piao and So Hun Kim contributed equally to this study as first authors.
Copyright © 2017 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Regular aerobic exercise is essential for the prevention and management of type 2 diabetes mellitus and may be particularly beneficial for those treated with thiazolidinediones, since it may prevent associated weight gain. This study aimed to evaluate the effect of combined exercise and rosiglitazone treatment on body composition and glucose metabolism in obese diabetes-prone animals.
-
Methods
- We analyzed metabolic parameters, body composition, and islet profiles in Otsuka Long Evans Tokushima Fatty rats after 28 weeks of aerobic exercise, rosiglitazone treatment, and combined exercise and rosiglitazone treatment.
-
Results
- Combined exercise with rosiglitazone showed significantly less increase in weight and epididymal fat compared to rosiglitazone treatment. Aerobic exercise alone and combined rosiglitazone and exercise treatment led to similar retention of lean body mass. All experimental groups showed a decrease in fasting glucose. However, the combined exercise and rosiglitazone therapy group showed prominent improvement in glucose tolerance compared to the other groups. Rescue of islet destruction was observed in all experimental groups, but was most prominent in the combined therapy group.
-
Conclusion
- Regular aerobic exercise combined with rosiglitazone treatment can compensate for the adverse effect of rosiglitazone treatment and has benefit for islet preservation.
- Keywords: Beta-cell; Body composition; Diabetes; Exercise; Rats, inbred OLETF; Rosiglitazone
- Obesity is known to be a major predisposing risk factor for type 2 diabetes mellitus (T2DM) [1]. Obesity may lead to insulin resistance and β-cell dysfunction [23]. Major strategies for T2DM therapy focuses on the alleviation of insulin resistance, improvement of β-cell function, and weight loss for obese patients [4]. Rosiglitazone, a thiazolidinedione (TZD) class antidiabetic agent, acts through activating the nuclear transcription factor peroxisome proliferator-activated receptor γ [567]. In animal models of insulin resistance, rosiglitazone has been reported to decrease plasma glucose and insulin levels, improve insulin action in skeletal muscles, and prevent β-cell atrophy [8]. It also has been reported that rosiglitazone promotes β-cell survival and preserves β-cell mass in human islet amyloid polypeptide transgenic mice [89]. However, rosiglitazone causes weight gain as an adverse effect, leading to overweight and obesity, which may be a major limitation of its use [10].
- Aerobic exercise has been traditionally prescribed for diabetes prevention and management. Aerobic exercise has beneficial effects on glucose metabolism, mainly through improvement of insulin sensitivity [11]. Aerobic exercise is an important component of lifestyle modification interventions for weight loss. Therefore, aerobic exercise that can lead to increased energy consumption may be beneficial in preventing the weight gain associated with the use of rosiglitazone [12]. Previous studies have shown the beneficial effects of combined exercise and rosiglitazone in improving cardiovascular risk factors in patients with T2DM [1314].
- Beneficial effects of aerobic exercise on β-cell preservation has been suggested in animal and human studies [15]. However, the effect of combined regular aerobic exercise and rosiglitazone treatment on the pancreas islet in T2DM has not been evaluated. We hypothesized that the combination of aerobic exercise with rosiglitazone treatment can enhance positive effects by compensating for the adverse effect of weight gain caused by TZDs, as well as by accelerating the improvement of insulin resistance and β-cell preservation. In the present study, we investigated changes in body composition, glucose metabolism, insulin resistance, and islet morphology in a T2DM animal model after administrating aerobic exercise and rosiglitazone, alone and concomitantly.
INTRODUCTION
- Animals
- The Otsuka Long Evans Tokushima Fatty (OLETF) rat is a useful animal model for T2DM with obesity [16]. At 5 to 6 weeks, the OLETF rat develops obesity, while diabetic symptoms appear at about 24 weeks [17]. Male OLETF rats and age-matched male Long-Evans Tokushima Otsuka (LETO) rats were supplied by Tokushima Research Institute, Otsuka Pharmaceuticals (Tokushima, Japan). The rats were individually housed in a standard animal facility with access to standard food and water ad libitum and maintained on a 12-hour light-dark cycle (lights on at 7:00 AM) in a temperature- (22° to 25℃) and humidity- (50% to 60%) controlled colony room. All procedures were in accordance with institutional guidelines for animal research at Inha University in Incheon, the Republic of Korea.
- Experimental protocol
- At 12 weeks of age, a total of 56 OLETF rats were randomly allocated into four groups: (1) OLETF rats with no treatment (OC, n=14), (2) OLETF rats with rosiglitazone treatment only (OR, n=14), (3) OLETF rats with exercise only (OEx, n=14), and (4) OLETF rats with combined treatment of rosiglitazone and exercise (OREx, n=14). LETO rats were used as normal controls (LETO, n=14). Additional non-treated OLETF rats (n=7) and LETO rats (n=7) were sacrificed at baseline (12 weeks old) for pathologic examination. Rosiglitazone maleate (3 mg/kg/day, Avandia; GSK Inc., Philadelphia, PA, USA) was orally administered to OR and OREx once a day, 6 days/week from week 12 as previously described [18] for 12 or 28 weeks. The OEx and OREx groups were submitted to a 30 minutes exercise at 15 m/min speed (low-intensity) on a forced exercise wheel system (rat activity cage; JD-A-06; Jungdo B&P, Seoul, Korea) 5 days a week for 12 or 28 weeks. After 12 weeks (24 weeks old) and 28 weeks (40 weeks old) of treatment, intraperitoneal glucose tolerance test (IPGTT) was performed for the evaluation of glucose tolerance, and the animals were sacrificed for evaluation of the pancreatic islet cells and molecular analysis.
- Analysis of blood samples
- Blood samples were drawn after an overnight fast, and the serum samples were kept at −80℃ for subsequent assays. Insulin and leptin levels were determined with enzyme-linked immunosorbent assay (ELISA) kits (Linco Research Inc., St. Charles, MA, USA). Adiponectin levels were determined with an ELISA kit (ALPCO Diagnostics, Windham, NH, USA). Glucose, total cholesterol, high density lipoprotein cholesterol, low density lipoprotein cholesterol, triglycerides, and free fatty acids were assessed with an automated chemistry analyzer (ADVIA 1650; Bayer, Tokyo, Japan). A surrogate index of insulin sensitivity was calculated from fasting blood glucose and plasma insulin concentrations as follows: homeostasis model assessment of insulin resistance (HOMA-IR)=(G0×I0)/22.5, with glucose expressed as mmol/L and insulin expressed as µU/mL [19].
- Glucose tolerance test
- The IPGTT was performed as described previously [17]. Blood glucose level was analyzed using a glucometer (One Touch Instrument; Roche, Basel, Swiss). Area under the curve (AUC) for glucose was calculated according to the trapezoidal rule from the glucose measurements at baseline, 30, 60, 90, 120, and 180 minutes.
- Dual energy X-ray absorptiometry
- Body composition was measured at 30 weeks of age by dual-energy X-ray absorptiometry (DEXA). Before undergoing a whole-body scan, the rats were anaesthetized with Ketamine mixtures (Ketamine 35 mg/kg+Xylazine 3 mg/kg+Acetylpro-mazine 0.75 mg/kg), and then total body mass, bone mineral content, lean body mass, and fat mass were measured in vivo with a QDR 4500A DEXA using small animal analysis software (DEXA; Hologic Inc., Waltham, MA, USA).
- The β-cell and islet morphology
- Immunocytochemistry was performed as described previously [20]. For insulin staining, tissue sections were incubated with anti-insulin antibody (1:1,000; Biogenex, San Ramon, CA, USA) for 24 hours at 4℃. In situ hybridization for insulin was carried out on tissue sections. After treating with proteinase K, the sections were incubated with alkaline phosphatase-conjugated anti-digoxigenin antibody. Hybridization signals were revealed by nitroblue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate toluidinium. For the double immunostaining, tissue sections were treated with mouse anti-rat insulin antibody (1:1,000; Biogenex) and rabbit anti-rat glucose transporter 2 (GLUT-2) antibody (1:1,000; Alpha Diagnostic International, San Antonio, TX, USA). After applying the secondary antibody, fluorescein isothiocyanate (FITC) conjugated anti-mouse and rhodamine conjugated anti-rabbit antibody (Jackson Immuno-Research Lab. Inc., West Grove, PA, USA) were added. The tissue sections were observed using a Radiance 2100 confocal microscope (Bio-Rad and Nikon, Tokyo, Japan).
- For morphometric analysis, insulin-positive cells were counted in the entire area of the tissue sections, and β-cell number per unit area (1 µm2) was calculated as reported previously [21]. To assess islet preservation, the islet destruction ratio (%) was calculated as (the number of destructed islets)/(the total islet number evaluated)×100 (%). For each pancreas, two slides were reviewed at 10× magnification and 20 photos without overlap were randomly taken to count the number of islets. The corresponding islets were examined at 40× magnification to evaluate whether there were signs of destruction. Destructed islets were defined as having lymphocyte infiltration and a distorted outline disabling clear demarcation of the islet area. The mean of the islet destruction ratios of the two slides was calculated. Image files were analyzed using Quantity One (Bio-Rad, Hercules, CA, USA).
- Statistical analysis
- Nonparametric tests were performed to compare the metabolic parameters, body weight, and body composition of the LETO, OC, OR, OEx, and OREx groups at each age point, due to the relatively small sample size of each group. The Mann-Whitney U test was used to compare the glucose tolerance levels and AUCs between LETO and OC groups at the 12-week age point. We compared body weight, body composition, glucose metabolic parameters, and AUCs among LETO, OC, OR, OEx, and OREx groups at 24 and 40 weeks using the Kruskal-Wallis test and Tukey method, using ranks for multiple comparisons. Bonferroni adjustments for multiple tests were additionally carried out to compare body and epididymal fat weight variables. SPSS version 14.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analysis. A level of 5% was used for statistical significance.
METHODS
- Metabolic characteristics
- Table 1 reports the metabolic parameters of the LETO and OLETF rats of each group at 40 weeks (after the 28-week intervention). The plasma glucose and insulin levels were higher in the OC group, but reduced in the OR, OEx, and OREx groups. Serum adiponectin concentration was elevated in the OR (P<0.01) and OREx groups (P<0.01). The serum leptin concentration was decreased in the OEx group (P<0.01). Regarding plasma lipid profiles, significant decreases in serum total cholesterol (P<0.05 and P<0.01, respectively) and triglyceride (P=0.02 and P=0.03, respectively) levels were observed in both the OR and OREx groups. HOMA-IR, a surrogate marker of insulin resistance, was significantly increased in the OC group compared to the LETO group. The OR, OEx, and OREx groups all showed improved insulin sensitivity compared to the OC group to a similar extent (Table 1).
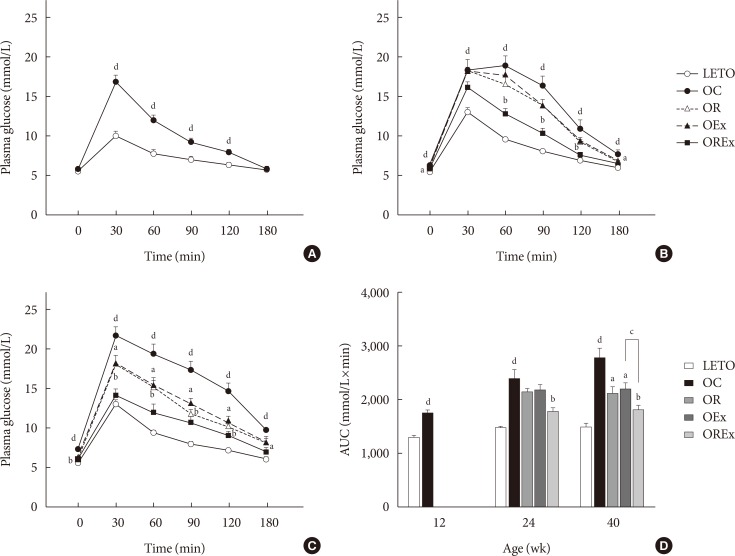
- Effects of rosiglitazone and exercise on glucose tolerance
- The IPGTT was performed at 12, 24, and 40 weeks of age to assess glucose tolerance (Fig. 1). A greater increase in blood glucose levels in response to the intraperitoneal glucose load was seen in the OC group compared to LETO (P<0.01) (Fig. 2A–C). At 24 weeks, the OC group showed significantly higher fasting glucose levels compared to the LETO group, and the OREx group showed lower fasting glucose levels compared to the OC group (Fig. 2B). At 40 weeks, fasting plasma glucose was significantly higher in the OC group compared to LETO, and the OR, OEx, OREx groups all showed a lower fasting plasma glucose level compared to the OC group (Fig. 2C). Glucose tolerance was further impaired with aging for the OC group, but significant improvement in glucose tolerance was seen in the OR, OEx, and OREx groups at 24 and 40 weeks. The OREx group showed a further improvement compared to the OR and OEx groups (Fig. 2B and C). At 24 weeks of age, the glucose AUCs of the OREx group were significantly lower than those of the OC group, while no significant changes were found in the OR and OEx groups. At 40 weeks, the OR, OEx, and OREx groups all showed lower glucose AUCs compared to the OC group, and the OREx group showed further improvement compared to the OR and OEx groups (Fig. 2D).
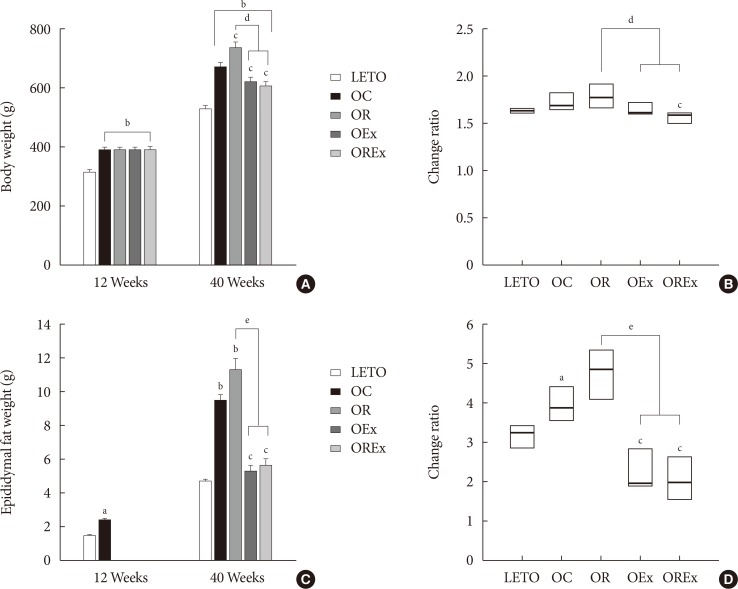
- Effects of rosiglitazone and exercise on body weight and composition
- Changes of body weight and visceral fat are presented in Fig. 3. At 12 weeks of age, OLETF rats demonstrated significantly higher body weight than the LETO controls (393.8 [377.5 to 408.0] vs. 320.0 [307.5 to 328.75], P<0.01) (Fig. 3A). At 40 weeks of age (after 28 weeks of treatment), the OR group demonstrated a significantly higher weight gain (1.78 [1.70 to 1.93] fold) compared to the OC group (1.69 [1.64 to 1.76] fold), whereas weight gains were considerably lower in the OEx (1.62 [1.52 to 1.70] fold) and OREx (1.57 [1.49 to 1.60] fold) groups (Fig. 3B). This shows that rosiglitazone induced undesirable weight gain, and that this weight gain can be rescued by regular aerobic exercise. Epididymal fat in the experimental animals paralleled changes in body weight (Fig. 3C and D), implying that weight control by aerobic exercise is accompanied by suppressed visceral fat increase. These findings were closely related to the body composition evaluated by DEXA at 30 weeks of age (Table 2), showing a significant improvement in composition of both lean body mass and fat body mass in the OEx and OREX groups compared to the OR group. There was no significant difference in food intake among the groups, although there was a trend of increased intake in the OC group compared to the LETO group.
- Effects of rosiglitazone and exercise on islet morphology
- Fig. 3A shows chronological changes in islets in the LETO and OLETF rats. All experimental rats retained normal islet features with round or oval shape, compact cellular mass, and a distinct demarcation from the exocrine tissue at 12 weeks of age. Development of atypical islets were detected at 24 weeks of age in OLETF rats. The islets were somewhat inflated and started to lose outer demarcation, particularly in the OC group, whereas the islets of the LETO control group demonstrated no alteration at all. Alterations of the islet were aggravated at 40 weeks in the OC group, which showed a looser islet constitution owing to increased fiber substance as well as lymphocyte infiltration. The β-cells in the islets display relatively weak immunoreactivity for insulin (Fig. 4A). However, improvement in islet preservation was obtained by rosiglitazone treatment as well as by regular aerobic exercises. The islets of the OR and OEx groups revealed relatively well-organized islet features retaining a compact β-cell mass with distinguishable contours. Moreover, preservation of the islet was prominent in the OLETF rats treated with combined rosiglitazone and aerobic exercise (OREx) which showed a compact islet mass with strong insulin immunoreactive cells (Fig. 4A).
- To assess islet preservation, we assessed the islet destruction ratio as the percentage of islets showing an apparent morphological alteration (Fig. 4B). The β-cells mass was estimated by counting the number of β-cells per unit area (µm2) of the same tissue sections used for the determination of altered islets (Fig. 4B). Alteration and destruction of the islet was found to be severe in the OC group, especially at the age of 40 weeks (>80%). However, the islets of the OR and OEx groups demonstrated a significant reduction of this islet alteration and destruction. Furthermore, we found a remarkable improvement of islet preservation in experimental rats treated with the combination of rosiglitazone and aerobic exercise (OREx) compared to the OR and OE groups. Only a small number of altered islets (<5%) was detected in the OREx group. We also observed a significant increase of β-cell numbers in the OR, OEx, and OREx groups at 40 weeks of age (Fig. 4C).
- Insulin expression in β-cells was confirmed at the insulin mRNA level by in situ hybridization, as an indication of insulin synthesis (Fig. 4D). Expression signal of insulin mRNA in the OC group was lower compared to the LETO controls, whereas the hybridization signals appeared to be improved in the OR, OEx and OREx groups.
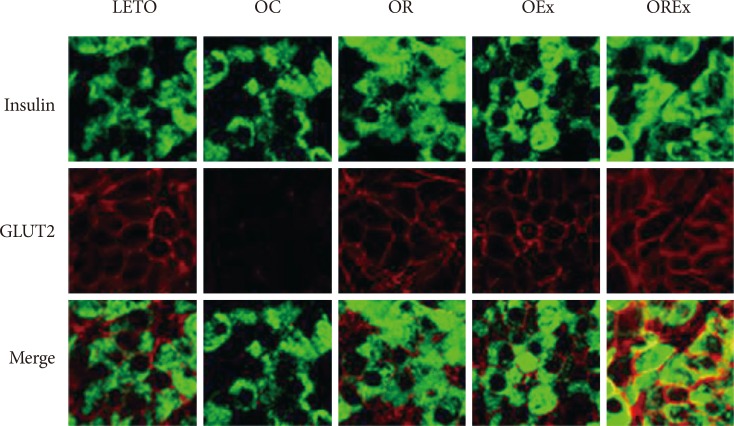
- Pancreatic GLUT-2 expression
- To estimate glucose sensitivity of the β-cells, we examined GLUT-2 immunoreactivity of the β-cell membrane (Fig. 5). We found substantial loss of GLUT-2 immunolabeling in the β-cells of the OC rats compared to the LETO controls, but intact expression of GLUT-2 in the β-cells of the OR, OEx, and OREx rats.
RESULTS
- Rosiglitazone is an antidiabetic agent used in patients with T2DM to improve insulin sensitivity and glucose metabolism [5622]. Rosiglitazone has superior durability when used for a long period of time compared to other antidiabetic agents, probably due to its beneficial effect on preserving β-cell function [23]. However, the adverse effect of body weight gain [10] may limit its clinical use. Therefore, an effort to limit unwanted weight gain will be necessary, and sufficient regular aerobic exercise may be beneficial in this clinical setting [24]. In this study, we evaluated the antidiabetic effects of aerobic exercise and its synergic advantages by combining it with TZD treatment. The combination of aerobic exercise and rosiglitazone treatment was able to further improve glucose tolerance, favorably affect body composition, and prevent destruction of pancreatic islets in OLETF rats as an additional benefit.
- Several studies have demonstrated that sufficient amount of exercise can reduce body weight or prevent regain in both humans and rats [11222425]. We found that regular aerobic exercise effectively mitigates spontaneous obesity as well as rosiglitazone-induced weight gain in OLETF rats. Exercise is also of great benefit since it can reduce abdominal obesity, visceral fat, and cardiovascular risk factors, and increase both skeletal muscle mass and cardiorespiratory fitness, even without significant weight loss [26]. Indeed, aerobic exercise resulted in successful attenuation of total fat mass and visceral fat mass increases caused by rosiglitazone suggesting that the major obstacle of TZDs therapy can be eliminated by physical exercise.
- Exercise and rosiglitazone as a combination treatment was shown to have complementary and additional beneficial effects on glycemic indexes, insulin sensitivity, and cardiovascular risk factors compared to either exercise or rosiglitazone by itself in previous studies [131427282930]. In patients with T2DM, simultaneous treatment with rosiglitazone and exercise counteracted rosiglitazone induced weight gain, extended improvements of insulin sensitivity, glycemic control and fitness beyond those expected by their complementary actions in patients with T2DM after 8 months [13]. In obese Zucker fatty rats, exercise and troglitazone alone each had a beneficial effect on insulin sensitivity, while the combination of both treatments completely normalized insulin sensitivity to the level of lean control rats. Exercise and troglitazone displayed different mechanisms, and therefore the combined effect was able to have additional benefits [28]. The present study shows that at 40 weeks (after 28 weeks of treatment) groups treated with exercise alone or rosiglitazone alone showed improved glucose tolerance, and the combined rosiglitazone and exercise treatment group showed more prominent improvement in glucose tolerance, in accordance with previous studies. Adiponectin was significantly increased in the OR and OREx groups, while there was no significant change in the OEx group. This finding is consistent with previous studies in patients with T2DM [13]. The increase in plasma adiponectin level by rosiglitazone treatment, which is not seen in the exercise-alone group, would be an additional benefit of combining rosiglitazone to exercise, since adiponectin is known to have beneficial effects such as insulin sensitizing, anti-inflammatory, and anti-atherogenic properties [31]. Although we have not measured other cytokines, it is possible that other cytokines may have also played a role. In patients with T2DM, exercise, rosiglitazone, and combined treatment all suppressed interleukin 6 (IL-6), resistin, and IL-18, and increased IL-10 levels, while both combined treatment and rosiglitazone treatment alone decreased tumor necrosis factor α levels significantly [1314].
- In addition to the known beneficial effects of the combined treatment of aerobic exercise and rosiglitazone, the current study shows the benefit of preservation of pancreas islet morphology in a T2DM rat model. This is the first study to evaluate pancreas morphology after concurrent treatment with exercise and a TZD. Since direct evaluation of the pancreas after treatment is not feasible in human studies, this animal data should be informative in providing knowledge on the effects of combined treatment in the pancreas. Diabetes is closely associated with defects of islet β-cells, and β-cell dysfunction is an early event leading to the development of T2DM [32]. In obese humans, there is an increase in β-cell mass compared to non-obese humans. Obese patients with impaired fasting glucose and T2DM had a significantly decreased β-cell mass compared to non-diabetic people [33]. This coincides with significant alterations of pancreatic islets as shown in non-treated diabetic OLETF rats. Both rosiglitazone [83234] and exercise [153536] have been reported to have beneficial effects on pancreatic islet preservation in animals. The present study further shows that aerobic exercise protects islet destruction particularly when combined with rosiglitazone treatment. Hyperglycemia, hyperlipidemia, and inflammation are thought to contribute to β-cell dysfunction [32]. Rosiglitazone has been shown to improve indices of β-cell function when assessed by the HOMA in patients with T2DM. Exercise also has been shown to improve β-cell function measured by the disposition index [1537]. The benefit of both exercise and rosiglitazone on the pancreas may be indirect, mainly by reducing the secretory demand, but there is evidence supporting direct beneficial effects on the pancreas, especially for rosiglitazone [3839].
- In summary, the present study demonstrates that combined rosiglitazone with regular aerobic exercise can prominently improve glucose metabolism, and this is associated with protection from islet destruction in a T2DM prone-animal model. Combined regular aerobic exercise with rosiglitazone was also able to attenuate the increase in body weight and fat mass induced by rosiglitazone treatment. It is noticeable that aerobic exercise and rosiglitazone can complement each other and lead to additional benefits when combined, and can lead to better sustained glycemic control in accordance with its additional benefit on islet preservation. Therefore, participating in regular aerobic exercise should be strongly encouraged to patients with T2DM in conjunction with TZD prescription for better therapeutic effects, and to prevent the side effects of TZDs.
DISCUSSION
-
Acknowledgements
- This study was supported by a grant of the Korea Healthcare technology R&D Project Ministry of Health and Welfare, Republic of Korea (HI14C1062). Shan-Ji Piao received a grant supported by the Brain Korea 21 Project, Republic of Korea. This work was supported by the KOSEF (2006-05402, R01-2006-000-11386-0, and 2009-0091914) and NRF (2017R1D1A1B03034581), Republic of Korea.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Kriketos AD, Carey DG, Jenkins AB, Chisholm DJ, Furler SM, Campbell LV. Central fat predicts deterioration of insulin secretion index and fasting glycaemia: 6-year follow-up of subjects at varying risk of type 2 diabetes mellitus. Diabet Med 2003;20:294-300. ArticlePubMed
- 2. Kahn SE. Clinical review 135: the importance of beta-cell failure in the development and progression of type 2 diabetes. J Clin Endocrinol Metab 2001;86:4047-4058. PubMed
- 3. Guldstrand M, Ahren B, Adamson U. Improved beta-cell function after standardized weight reduction in severely obese subjects. Am J Physiol Endocrinol Metab 2003;284:E557-E565. PubMed
- 4. Inzucchi SE, Bergenstal RM, Buse JB, Diamant M, Ferrannini E, Nauck M, Peters AL, Tsapas A, Wender R, Matthews DR. Management of hyperglycaemia in type 2 diabetes: a patient-centered approach. Position statement of the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia 2012;55:1577-1596. ArticlePubMedPDF
- 5. Ferre P. The biology of peroxisome proliferator-activated receptors: relationship with lipid metabolism and insulin sensitivity. Diabetes 2004;53(Suppl 1):S43-S50. PubMed
- 6. Dana SL, Hoener PA, Bilakovics JM, Crombie DL, Ogilvie KM, Kauffman RF, Mukherjee R, Paterniti JR Jr. Peroxisome proliferator-activated receptor subtype-specific regulation of hepatic and peripheral gene expression in the Zucker diabetic fatty rat. Metabolism 2001;50:963-971. ArticlePubMed
- 7. Etgen GJ, Oldham BA, Johnson WT, Broderick CL, Montrose CR, Brozinick JT, Misener EA, Bean JS, Bensch WR, Brooks DA, Shuker AJ, Rito CJ, McCarthy JR, Ardecky RJ, Tyhonas JS, Dana SL, Bilakovics JM, Paterniti JR Jr, Ogilvie KM, Liu S, Kauffman RF. A tailored therapy for the metabolic syndrome: the dual peroxisome proliferator-activated receptor-alpha/gamma agonist LY465608 ameliorates insulin resistance and diabetic hyperglycemia while improving cardiovascular risk factors in preclinical models. Diabetes 2002;51:1083-1087. PubMed
- 8. Koh EH, Kim MS, Park JY, Kim HS, Youn JY, Park HS, Youn JH, Lee KU. Peroxisome proliferator-activated receptor (PPAR)-alpha activation prevents diabetes in OLETF rats: comparison with PPAR-gamma activation. Diabetes 2003;52:2331-2337. PubMed
- 9. Hull RL, Shen ZP, Watts MR, Kodama K, Carr DB, Utzschneider KM, Zraika S, Wang F, Kahn SE. Long-term treatment with rosiglitazone and metformin reduces the extent of, but does not prevent, islet amyloid deposition in mice expressing the gene for human islet amyloid polypeptide. Diabetes 2005;54:2235-2244. ArticlePubMedPDF
- 10. Umrani DN, Banday AA, Hussain T, Lokhandwala MF. Rosiglitazone treatment restores renal dopamine receptor function in obese Zucker rats. Hypertension 2002;40:880-885. ArticlePubMed
- 11. Colberg SR, Sigal RJ, Fernhall B, Regensteiner JG, Blissmer BJ, Rubin RR, Chasan-Taber L, Albright AL, Braun B. American College of Sports Medicine. American Diabetes Association. Exercise and type 2 diabetes: the American College of Sports Medicine and the American Diabetes Association. Joint position statement. Diabetes Care 2010;33:e147-e167. PubMedPMC
- 12. American Diabetes Association. Standards of medical care in diabetes: 2015. Summary of revisions. Diabetes Care 2015;38(Suppl):S4.ArticlePMCPDF
- 13. Kadoglou NP, Iliadis F, Liapis CD, Perrea D, Angelopoulou N, Alevizos M. Beneficial effects of combined treatment with rosiglitazone and exercise on cardiovascular risk factors in patients with type 2 diabetes. Diabetes Care 2007;30:2242-2244. ArticlePubMedPDF
- 14. Kadoglou NP, Iliadis F, Sailer N, Athanasiadou Z, Vitta I, Kapelouzou A, Karayannacos PE, Liapis CD, Alevizos M, Angelopoulou N, Vrabas IS. Exercise training ameliorates the effects of rosiglitazone on traditional and novel cardiovascular risk factors in patients with type 2 diabetes mellitus. Metabolism 2010;59:599-607. ArticlePubMed
- 15. Narendran P, Solomon TP, Kennedy A, Chimen M, Andrews RC. The time has come to test the beta cell preserving effects of exercise in patients with new onset type 1 diabetes. Diabetologia 2015;58:10-18. ArticlePubMedPDF
- 16. Okauchi N, Mizuno A, Zhu M, Ishida K, Sano T, Noma Y, Shima K. Effects of obesity and inheritance on the development of non-insulin-dependent diabetes mellitus in Otsuka-Long-Evans-Tokushima fatty rats. Diabetes Res Clin Pract 1995;29:1-10. ArticlePubMed
- 17. Vanheest JL, Rodgers CD. Effects of exercise in diabetic rats before and during gestation on maternal and neonatal outcomes. Am J Physiol 1997;273(4 Pt 1):E727-E733. ArticlePubMed
- 18. Johns DG, Ao Z, Eybye M, Olzinski A, Costell M, Gruver S, Smith SA, Douglas SA, Macphee CH. Rosiglitazone protects against ischemia/reperfusion-induced leukocyte adhesion in the zucker diabetic fatty rat. J Pharmacol Exp Ther 2005;315:1020-1027. ArticlePubMed
- 19. Muniyappa R, Chen H, Muzumdar RH, Einstein FH, Yan X, Yue LQ, Barzilai N, Quon MJ. Comparison between surrogate indexes of insulin sensitivity/resistance and hyperinsulinemic euglycemic clamp estimates in rats. Am J Physiol Endocrinol Metab 2009;297:E1023-E1029. ArticlePubMedPMC
- 20. Kim SY, Lee SH, Kim BM, Kim EH, Min BH, Bendayan M, Park IS. Activation of nestin-positive duct stem (NPDS) cells in pancreas upon neogenic motivation and possible cytodifferentiation into insulin-secreting cells from NPDS cells. Dev Dyn 2004;230:1-11. ArticlePubMed
- 21. Lee SH, Han YM, Min BH, Park IS. Cytoprotective effects of polyenoylphosphatidylcholine (PPC) on beta-cells during diabetic induction by streptozotocin. J Histochem Cytochem 2003;51:1005-1015. ArticlePubMedPDF
- 22. Saris WH, Blair SN, van Baak MA, Eaton SB, Davies PS, Di Pietro L, Fogelholm M, Rissanen A, Schoeller D, Swinburn B, Tremblay A, Westerterp KR, Wyatt H. How much physical activity is enough to prevent unhealthy weight gain? Outcome of the IASO 1st Stock Conference and consensus statement. Obes Rev 2003;4:101-114. ArticlePubMed
- 23. Kahn SE, Haffner SM, Heise MA, Herman WH, Holman RR, Jones NP, Kravitz BG, Lachin JM, O’Neill MC, Zinman B, Viberti G. ADOPT Study Group. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N Engl J Med 2006;355:2427-2443. ArticlePubMed
- 24. Minami A, Ishimura N, Harada N, Sakamoto S, Niwa Y, Nakaya Y. Exercise training improves acetylcholine-induced endothelium-dependent hyperpolarization in type 2 diabetic rats, Otsuka Long-Evans Tokushima fatty rats. Atherosclerosis 2002;162:85-92. ArticlePubMed
- 25. Schoeller DA, Shay K, Kushner RF. How much physical activity is needed to minimize weight gain in previously obese women? Am J Clin Nutr 1997;66:551-556. ArticlePubMed
- 26. Ross R, Bradshaw AJ. The future of obesity reduction: beyond weight loss. Nat Rev Endocrinol 2009;5:319-325. ArticlePubMedPDF
- 27. Hallsten K, Virtanen KA, Lonnqvist F, Sipila H, Oksanen A, Viljanen T, Ronnemaa T, Viikari J, Knuuti J, Nuutila P. Rosiglitazone but not metformin enhances insulin- and exercise-stimulated skeletal muscle glucose uptake in patients with newly diagnosed type 2 diabetes. Diabetes 2002;51:3479-3485. ArticlePubMedPDF
- 28. Hevener AL, Reichart D, Olefsky J. Exercise and thiazolidinedione therapy normalize insulin action in the obese Zucker fatty rat. Diabetes 2000;49:2154-2159. ArticlePubMedPDF
- 29. Yaspelkis BB 3rd, Lessard SJ, Reeder DW, Limon JJ, Saito M, Rivas DA, Kvasha I, Hawley JA. Exercise reverses high-fat diet-induced impairments on compartmentalization and activation of components of the insulin-signaling cascade in skeletal muscle. Am J Physiol Endocrinol Metab 2007;293:E941-E949. ArticlePubMed
- 30. Lessard SJ, Rivas DA, Chen ZP, Bonen A, Febbraio MA, Reeder DW, Kemp BE, Yaspelkis BB 3rd, Hawley JA. Tissue-specific effects of rosiglitazone and exercise in the treatment of lipid-induced insulin resistance. Diabetes 2007;56:1856-1864. ArticlePubMedPDF
- 31. Simpson KA, Singh MA. Effects of exercise on adiponectin: a systematic review. Obesity (Silver Spring) 2008;16:241-256. ArticlePubMedPDF
- 32. Leiter LA. Beta-cell preservation: a potential role for thiazolidinediones to improve clinical care in type 2 diabetes. Diabet Med 2005;22:963-972. ArticlePubMed
- 33. Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC. Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes. Diabetes 2003;52:102-110. PubMed
- 34. Finegood DT, McArthur MD, Kojwang D, Thomas MJ, Topp BG, Leonard T, Buckingham RE. Beta-cell mass dynamics in Zucker diabetic fatty rats. Rosiglitazone prevents the rise in net cell death. Diabetes 2001;50:1021-1029. PubMed
- 35. Calegari VC, Abrantes JL, Silveira LR, Paula FM, Costa JM Jr, Rafacho A, Velloso LA, Carneiro EM, Bosqueiro JR, Boschero AC, Zoppi CC. Endurance training stimulates growth and survival pathways and the redox balance in rat pancreatic islets. J Appl Physiol (1985) 2012;112:711-718. ArticlePubMed
- 36. Shima K, Shi K, Mizuno A, Sano T, Ishida K, Noma Y. Exercise training has a long-lasting effect on prevention of non-insulin-dependent diabetes mellitus in Otsuka-Long-Evans-Tokushima Fatty rats. Metabolism 1996;45:475-480. ArticlePubMed
- 37. Slentz CA, Tanner CJ, Bateman LA, Durheim MT, Huffman KM, Houmard JA, Kraus WE. Effects of exercise training intensity on pancreatic beta-cell function. Diabetes Care 2009;32:1807-1811. PubMedPMC
- 38. Gupta D, Peshavaria M, Monga N, Jetton TL, Leahy JL. Physiologic and pharmacologic modulation of glucose-dependent insulinotropic polypeptide (GIP) receptor expression in beta-cells by peroxisome proliferator-activated receptor (PPAR)-gamma signaling: possible mechanism for the GIP resistance in type 2 diabetes. Diabetes 2010;59:1445-1450. PubMedPMC
- 39. Meidute Abaraviciene S, Lundquist I, Galvanovskis J, Flodgren E, Olde B, Salehi A. Palmitate-induced beta-cell dysfunction is associated with excessive NO production and is reversed by thiazolidinedione-mediated inhibition of GPR40 transduction mechanisms. PLoS One 2008;3:e2182ArticlePubMedPMC
REFERENCES
Experimental design. Flow chart of the experimental design is shown. LETO, Long-Evans Tokushima Otsuka; OLETF, Otsuka Long Evans Tokushima Fatty.
Effects of rosiglitazone and exercise treatments on glucose tolerance. Time course of blood glucose during the intraperitoneal glucose tolerance test is reported at (A) 12 weeks, (B) 24 weeks, and (C) 40 weeks of age. (D) The area under the curve (AUC) for glucose at 40 weeks were calculated. Values are presented as mean±standard error (n=7). LETO, Long-Evans Tokushima Otsuka; OC, Otsuka Long Evans Tokushima Fatty (OLETF) rats with no treatment; OR, OLETF rats with rosiglitazone treatment only; OEx, OLETF rats with exercise only; OREx, OLETF rats with combined treatment of rosiglitazone and exercise. aP<0.05 vs. OC, bP<0.01 vs. OC, cP<0.05, dP<0.01 vs. LETO.
Effects of rosiglitazone and exercise on body weight and epididymal fat weight. (A) Body weight and (C) epididymal fat weight at 12 and 40 weeks are shown. Values are presented as mean±standard error (n=7). The change ratio of (B) body weight and (D) epididymal fat weight from 12 to 40 weeks were calculated. Each box (n=7) are presented as median (interquartile range). LETO, Long-Evans Tokushima Otsuka; OC, Otsuka Long Evans Tokushima Fatty (OLETF) rats with no treatment; OR, OLETF rats with rosiglitazone treatment only; OEx, OLETF rats with exercise only; OREx, OLETF rats with combined treatment of rosiglitazone and exercise. aP<0.05 vs. LETO, bP<0.01 vs. LETO, cP<0.01 vs. OC, dP<0.01, eP<0.001.
Profiles of the pancreatic islets in rosiglitazone, exercise, and combined treatment. (A) Chronological changes of pancreatic islets in LETO rats and OLETF rats with and without anti-diabetic treatment are presented with insulin immunostaining at 12, 24, and 40 weeks. (B) Changes in islet alterations by percentage at 40 weeks. The number of the altered islets was counted in the pancreatic tissues and the ratio was calculated. Each box (n=7×2 sections) are presented as median (interquartile range). (C) β-Cell number of the total area of the pancreas was calculated at 40 weeks. The pancreatic islet area was measured using Quantity One (Bio-Rad). (D) In situ hybridization: insulin mRNA in LETO, OC, OR, OEx, and OREx groups at 40 weeks. LETO, Long-Evans Tokushima Otsuka; OC, Otsuka Long Evans Tokushima Fatty (OLETF) rats with no treatment; OR, OLETF rats with rosiglitazone treatment only; OEx, OLETF rats with exercise only; OREx, OLETF rats with combined treatment of rosiglitazone and exercise. aP<0.001 vs. LETO, bP<0.01 vs. OC, cP<0.001 vs. OC, dP<0.01.

Glucose transporter 2 (GLUT-2) expression in pancreatic islets with rosiglitazone, exercise, and combined treatment at 40 weeks. The pancreatic tissues were immunolabeled for insulin (green fluorescence in upper panel) and GLUT-2 (red fluorescence in middle panel). The immunofluorescent signals were merged and are shown in the lower panel. LETO, Long-Evans Tokushima Otsuka; OC, Otsuka Long Evans Tokushima Fatty (OLETF) rats with no treatment; OR, OLETF rats with rosiglitazone treatment only; OEx, OLETF rats with exercise only; OREx, OLETF rats with combined treatment of rosiglitazone and exercise.
The characteristics of the experimental groups after 28 weeks of treatment (at 40 weeks of age) with rosiglitazone, exercise, and combined rosiglitazone and exercise
Values are presented as median (interquartile range).
LETO, Long-Evans Tokushima Otsuka; OC, Otsuka Long Evans Tokushima Fatty (OLETF) rats with no treatment; OR, OLETF rats with rosiglitazone treatment only; OEx, OLETF rats with exercise only; OREx, OLETF rats with combined treatment of rosiglitazone and exercise; HDL-C, high density lipoprotein cholesterol; HOMA-IR, homeostasis model assessment of insulin resistance.
aP<0.05 vs. LETO, bP<0.05 vs. OC, cP<0.05 vs. OR.
Effects of rosiglitazone and exercise on body composition at 30 weeks of age
Values are presented as median (interquartile range).
LETO, Long-Evans Tokushima Otsuka; OC, Otsuka Long Evans Tokushima Fatty (OLETF) rats with no treatment; OR, OLETF rats with rosiglitazone treatment only; OEx, OLETF rats with exercise only; OREx, OLETF rats with combined treatment of rosiglitazone and exercise.
aP<0.05 vs. LETO, bP<0.05 vs. OR, cP<0.05 vs. OC.
Figure & Data
References
Citations

- Impacts of an Exercise Intervention on the Health of Pancreatic Beta-Cells: A Review
Shuang Zhang, Yaru Wei, Chunxiao Wang
International Journal of Environmental Research and Public Health.2022; 19(12): 7229. CrossRef - Molecular mechanisms by which aerobic exercise induces insulin sensitivity
Habib Yaribeygi, Stephen L. Atkin, Luis E. Simental‐Mendía, Amirhossein Sahebkar
Journal of Cellular Physiology.2019; 234(8): 12385. CrossRef

 KDA
KDA PubReader
PubReader Cite
Cite








