
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 39(4); 2015 > Article
-
Original ArticleOthers Predictive Factors for Efficacy of Dipeptidyl Peptidase-4 Inhibitors in Patients with Type 2 Diabetes Mellitus
- Shusuke Yagi1, Ken-ichi Aihara2, Masashi Akaike3, Daiju Fukuda1, Hotimah Masdan Salim1, Masayoshi Ishida1, Tomomi Matsuura1, Takayuki Ise1, Koji Yamaguchi1, Takashi Iwase1, Hirotsugu Yamada1, Takeshi Soeki1, Tetsuzo Wakatsuki1, Michio Shimabukuro4, Toshio Matsumoto2, Masataka Sata1
-
Diabetes & Metabolism Journal 2015;39(4):342-347.
DOI: https://doi.org/10.4093/dmj.2015.39.4.342
Published online: July 21, 2015
1Department of Cardiovascular Medicine, Institute of Biomedical Sciences, Tokushima University Graduate School, Tokushima, Japan.
2Department of Hematology, Endocrinology and Metabolism, Institute of Biomedical Sciences, Tokushima University Graduate School, Tokushima, Japan.
3Department of Medical Education, Institute of Biomedical Sciences, Tokushima University Graduate School, Tokushima, Japan.
4Department of Cardio-Diabetes Medicine, Institute of Biomedical Sciences, Tokushima University Graduate School, Tokushima, Japan.
- Corresponding author: Shusuke Yagi. Department of Cardiovascular Medicine, Institute of Biomedical Sciences, Tokushima University Graduate School, 3-18-15 Kuramoto-cho, Tokushima 770-8503, Japan. syagi@tokushima-u.ac.jp
Copyright © 2015 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Predictive factors for the efficacy of dipeptidyl peptidase-4 (DPP-4) inhibitors for lowering glycosylated hemoglobin (HbA1c) remain unclear in patients with type 2 diabetes mellitus. The aim of this study is therefore to clarify predictive factors of the efficacy of DPP-4 inhibitors for lowering HbA1c after 12 months of treatment.
-
Methods
- A total of 191 consecutive type 2 diabetic patients (male sex 55%, mean age, 68.3±35.8 years), who had been treated with DPP-4 inhibitors for 12 months, were enrolled in this study and evaluated retrospectively.
-
Results
- After 12 months of DPP-4 inhibitor treatment, random blood glucose level, and HbA1c level, decreased from 167±63 to 151±49 mg/dL (P<0.01), and from 7.5%±1.3% to 6.9%±0.9% (P<0.01) respectively, without severe side effects. Multiple regression analysis showed that predictors of DPP-4 inhibitor treatment efficacy in lowering HbA1c level after 12 months were a decrease in HbA1c level after 3 months of treatment, a high baseline HbA1c level, a low baseline body mass index, and the absence of coronary artery disease.
-
Conclusion
- Most suitable candidates for treatment with DPP-4 inhibitors are diabetics who are not obese and do not have coronary artery disease. In addition, long-term efficacy of DPP-4 inhibitors can be predicted by decrement of HbA1c after 3 months of treatment.
- Dipeptidyl peptidase-4 (DPP-4) inhibitors improve glucose metabolism by inhibiting the breakdown of incretins (a group of gastrointestinal hormones), including glucagon-like peptide 1 and glucose-dependent insulinotropic polypeptide. Glucose in the small intestine stimulates incretin release, and then these secreted incretins stimulate insulin secretion from β-cells and suppress inappropriate glucagon secretion from α-cells. Thus, DPP-4 inhibitors have a glucose-dependent anti-hyperglycemic action with a low incidence of hypoglycemia. DPP-4 inhibitors are widely used for the treatment of type 2 diabetes [1]. Reaching a glycosylated hemoglobin (HbA1c) level <7.0% is recommended as a treatment target [1]; however, predictive factors for the efficacy of these inhibitors in lowering HbA1c levels have not yet been identified. The aim of this study is to clarify factors including age, gender, body mass index (BMI), estimated glomerular filtration rate (eGFR), presence of dyslipidemia, hypertension, and coronary artery disease (CAD), that are predictive of the efficacy of DPP-4 inhibitors in lowering HbA1c level after 12 months of treatment.
INTRODUCTION
- We enrolled 191 consecutive type 2 diabetic patients who were recruited from Tokushima University Hospital during the period from April 2010 to January 2012, and who had been treated with DPP-4 inhibitors for at least 12 months with medical records. Anti-hyperglycemic efficacy of DPP-4 inhibitors was assessed by levels of HbA1c (National Glycohemoglobin Standardization Program) before and 12 months after treatment retrospectively.
- Subjects with the following diseases/conditions were excluded: active malignant diseases, connective tissue diseases treated by immunosuppressant and/or steroid therapy, renal failure (defined as serum creatinine >3.0 mg/dL), and liver dysfunction (defined as aspartate aminotransferase >100 IU/L, alanine aminotransferase >100 IU/L). Also excluded were patients receiving insulin or steroid therapy and patients whose treatment-e.g., with angiotensin-converting enzyme inhibitors/angiotensin II receptor blockers, statins, diuretics, or β-blockers-could affect glucose metabolism if that treatment was changed during the observational period. This study was carried out in conformance with the Declaration of Helsinki and the study protocol was approved by the Tokushima University Hospital Ethics Committee (No. 1760).
- Statistical analysis
- Continuous variables were averaged and each value expressed as the mean±standard deviation, or as a percentage for categorical parameters. HbA1c levels, random blood sugar levels, and parameters associated with dyslipidemia including low density lipoprotein cholesterol (LDL-C), triglyceride, high density lipoprotein cholesterol (HDL-C) before and 12 months after treatment with DPP-4 inhibitors were compared using the paired t-test. CAD was defined as angina pectoris, myocardial infarction, and silent myocardial ischemia with or without percutaneous coronary intervention or coronary artery bypass surgery. Gender and the presence of dyslipidemia, hypertension, and CAD were coded as dummy variables. The degrees of association among independent variables for HbA1c level after 12 months, including age, gender, BMI, eGFR, presence of dyslipidemia, hypertension, and CAD, were assessed by multiple regression analyses (stepwise regression model). All statistical analyses were performed using SPSS II version 11 software (SPSS Inc., Chicago, IL, USA). Statistical significance was defined as P<0.05.
METHODS
- Clinical characteristics of study subjects
- Characteristics of the patients enrolled in this study are shown in Table 1. Overall, these patients had moderate diabetes, with a mean HbA1c level of 7.5%±1.3%.
- Effects of DPP-4 inhibitors on blood glucose and HbA1c level
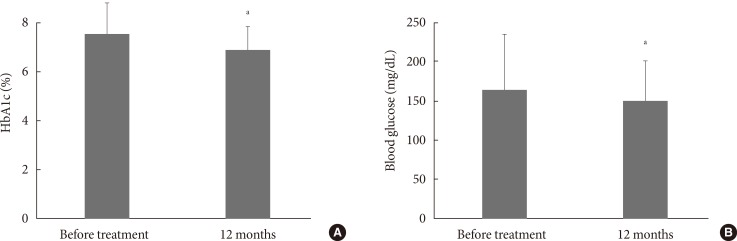
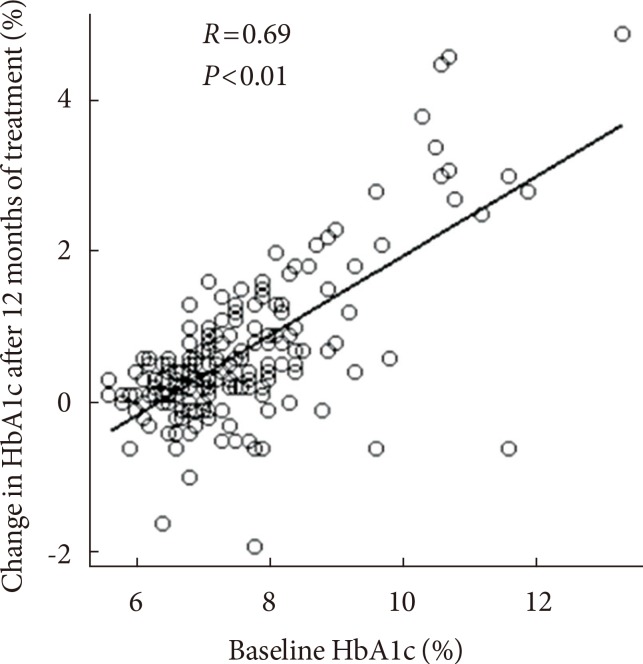
- Twelve months of treatment with DPP-4 inhibitors led to a significant decrease in random blood glucose level, from 167±63 to 151±49 mg/dL (P<0.01), and in HbA1c level, from 7.5%±1.3% to 6.9%±0.9% (P<0.01) (Fig. 1). Single regression analysis showed that the magnitude of decrease in HbA1c level after 12 months was positively associated with the baseline (the initial level at pre-treatment) HbA1c (P<0.01) (Fig. 2). In addition, the percentage of patients with a HbA1c level <7.0% after 12 months treatment increased from 36.6% to 60.2%.
- There was a significant decrease in LDL-C level, from 108±33 to 104±30 mg/dL (P<0.05), in response to treatment with DPP-4 inhibitors; however, no effects were observed for triglyceride, or HDL-C levels. There were no significant changes in eGFR values, and no cases of severe liver dysfunction were reported post-treatment.
- Contributors to decreased HbA1c levels after treatment with DPP-4 inhibitors
- Stepwise multiple regression analysis was performed to elucidate independent determinants for decreased HbA1c level 12 months after the start of treatment (baseline HbA1c to HbA1c at 12 months). It was shown that the degree of change in HbA1c level 3 months after the start of treatment (baseline HbA1c to HbA1c at 3 months) and baseline HbA1c level were positive contributors to change in HbA1c at 12 months after the start of treatment, and that BMI, and a history of CAD were negative contributors to change. This implies that the decrease in HbA1c level is greater in patients with a high baseline HbA1c level, a low baseline BMI, and an absence of CAD. However, associations with gender, age, eGFR, and presence of hypertension or dyslipidemia, were statistically excluded (Table 2).
- Safety of DPP-4 inhibitor treatment
- Over the 12-month DPP-4 inhibitor treatment period, no patients experienced severe side effects, such as hypoglycemic episodes or pancreatitis, which resulted in a hospital visit or hospitalization.
RESULTS
- We have shown that DPP-4 inhibitors are effective in decreasing HbA1c level after 12 months of treatment with no severe side effects. We also show that predictors of the efficacy of treatment were a decrease in HbA1c level after 3 months of treatment, a higher baseline HbA1c level, a lower baseline BMI, and the absence of CAD which is a novel finding of the study.
- It has not yet been established which patients would benefit from the effects of DPP-4 inhibitor treatment. For example, Monami et al. [2] showed that DPP-4 inhibitors were more effective in older patients with a mild/moderate fasting hyperglycemia, while Kim et al. [3] showed they were more effective in Asians than in other ethnic groups, especially in a lower BMI group of <30 kg/m2. In terms of predicting efficacy, Nomiyama et al. [4] and Maeda et al. [5] showed that a higher baseline HbA1c level, lower BMI, and shorter period since the onset/diagnosis of diabetes were significantly correlated with a greater HbA1c reduction. Iwasaki et al. [6] also reported that increased serum levels of eicosapentaenoic acid and docosahexaenoic acid could predict DPP-4 inhibitor efficacy. Lim et al. [7] showed that low insulinogenic index, indicating β-cell dysfunction, was a predictor of combination therapy of sitagliptin and metformin. Consistent with previous studies, our results also show that a decrease in HbA1c level is dependent on baseline HbA1c. Secondary failure such as desensitization of insulin secretion with sulfonylurea has been known in some antidiabetics [8]; however, we have shown that the decrease in HbA1c level after 12 months of treatment is associated with the decrease seen after 3 months of treatment, suggesting that long-term effects of DPP-4 inhibitors on glucose metabolism can be predicted by the short-term effects of this treatment. Another predictor of DPP-4 inhibitor response is low BMI. Insufficiency in insulin secretion occurs predominantly in patients with low BMI, while insulin resistance occurs predominantly in patients with high BMI. Thus DPP-4 inhibitors, which improve insulin secretion as well as insulin resistance, are more effective in patients with low BMI. Increased DPP-4 activity in patients with high BMI may account for the lower efficacy of DPP-4 inhibitors in this group [9]. Furthermore, DPP-4 inhibitors were more effective in patients without CAD than in patients with CAD. The mechanisms of the results were unknown, further basic studies are therefore needed to clarify the issue. In addition, whether DPP-4 inhibitors decrease cardiovascular events or mortality is still debatable [10111213], further clinical studies are also needed.
- HbA1c <7% is known as a treatment target with respect to diabetic complications from the guidelines [114]. In this study, 60.2% of patients with moderate diabetes reached this level, indicating that DPP-4 inhibitors are effective in lowering glucose.
- Lipid profile is an important determinant of cardiovascular risk in type 2 diabetic patients, and LDL-C is the most important risk factor. DPP-4 inhibitors have been reported to reduce LDL-C in addition to total cholesterol; however, results between studies are not consistent [151617]. The precise mechanism by which LDL-C is reduced by DPP-4 inhibitors has not been determined; although, DPP-4 inhibitors might reduce the expression of hepatic genes involved in cholesterol synthesis [18] or reduce intestinal secretion of cholesterol and apolipoprotein B-48 [19].
- The present study had several limitations. Only patients who had been treated with DPP-4 inhibitors for at least 12 months at our hospital were included, and thus, the study contains a patient selection bias. In addition, patients receiving insulin therapy were excluded, because co-administration of DPP-4 inhibitors and insulin was not permitted in Japan at the time of this study. This was a retrospective study with a small sample size; thus, we could not compare the effects of each DPP-4 inhibitor as the characteristics of patients varied between groups. We also had a comparatively short observation period, and thus could not analyze the long-term effects of DPP-4 inhibitor treatment. Randomized, or large, clinical cohort studies, with a longer observation period are needed to clarify these issues.
- In conclusion, suitable candidates for treatment with DPP-4 inhibitors are diabetic patients without obesity or CAD. In addition, the long-term efficacy of DPP-4 inhibitors can be predicted by a decrease in HbA1c level after 3 months of treatment.
DISCUSSION
-
Acknowledgements
- We thank the staff of the Hospital Information Center at Tokushima University Hospital for extracting patient data from medical records. This work was supported in part by JSPS KAKENHI (Grant Number 25461131, 25860605, and 25293184).
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: D.F. has received research funding from Takeda Pharmaceutical Company Ltd. and Ono Pharmaceutical Company Ltd. T.M. has received research funding from MSD Pharmaceuticals Private Ltd. and Takeda Pharmaceutical Company Ltd., and received lecture fees from Ono Pharmaceutical Company Ltd. and Novartis Pharmaceuticals Corporation. M.Sa. has received research funding from Takeda, Tanabe-Mitsubishi, Astellas, Daiichi-Sankyo, MSD, Byer Healthcare, and Ono, and lecture fees from Takeda, Boehringer Ingelheim, Byer Healthcare, Mochida, Astellas, Tanabe-Mitsubishi, Novartis, AstraZeneca, MSD, and Shionogi. The Department of Cardio-Diabetes Medicine, Tokushima University Graduate School, is supported in part by unrestricted research grants from Boehringer Ingelheim, Tanabe-Mitsubishi, Kowa, and Actelion. The others declare no conflict of interest.
NOTES
- 1. American Diabetes Association. Standards of medical care in diabetes: 2013. Diabetes Care 2013;36(Suppl 1):S11-S66. ArticlePubMedPDF
- 2. Monami M, Cremasco F, Lamanna C, Marchionni N, Mannucci E. Predictors of response to dipeptidyl peptidase-4 inhibitors: evidence from randomized clinical trials. Diabetes Metab Res Rev 2011;27:362-372. ArticlePubMed
- 3. Kim YG, Hahn S, Oh TJ, Kwak SH, Park KS, Cho YM. Differences in the glucose-lowering efficacy of dipeptidyl peptidase-4 inhibitors between Asians and non-Asians: a systematic review and meta-analysis. Diabetologia 2013;56:696-708. ArticlePubMedPDF
- 4. Nomiyama T, Akehi Y, Takenoshita H, Nagaishi R, Terawaki Y, Nagasako H, Kudo T, Kodera T, Kobayashi K, Urata H, Yanase T. CHAT. Contributing factors related to efficacy of the dipeptidyl peptidase-4 inhibitor sitagliptin in Japanese patients with type 2 diabetes. Diabetes Res Clin Pract 2012;95:e27-e28. ArticlePubMed
- 5. Maeda H, Kubota A, Tanaka Y, Terauchi Y, Matsuba I. ASSET-K Study group. The safety, efficacy and predictors for HbA1c reduction of sitagliptin in the treatment of Japanese type 2 diabetes. Diabetes Res Clin Pract 2012;95:e20-e22. ArticlePubMed
- 6. Iwasaki M, Hoshian F, Tsuji T, Hirose N, Matsumoto T, Kitatani N, Sugawara K, Usui R, Kuwata H, Sugizaki K, Kitamoto Y, Fujiwara S, Watanabe K, Hyo T, Kurose T, Seino Y, Yabe D. Predicting efficacy of dipeptidyl peptidase-4 inhibitors in patients with type 2 diabetes: association of glycated hemoglobin reduction with serum eicosapentaenoic acid and docosahexaenoic acid levels. J Diabetes Investig 2012;3:464-467.ArticlePubMedPMC
- 7. Lim S, An JH, Shin H, Khang AR, Lee Y, Ahn HY, Yoon JW, Kang SM, Choi SH, Cho YM, Park KS, Jang HC. Factors predicting therapeutic efficacy of combination treatment with sitagliptin and metformin in type 2 diabetic patients: the COSMETIC study. Clin Endocrinol (Oxf) 2012;77:215-223. ArticlePubMed
- 8. Satoh J, Takahashi K, Takizawa Y, Ishihara H, Hirai M, Katagiri H, Hinokio Y, Suzuki S, Tsuji I, Oka Y. Secondary sulfonylurea failure: comparison of period until insulin treatment between diabetic patients treated with gliclazide and glibenclamide. Diabetes Res Clin Pract 2005;70:291-297. ArticlePubMed
- 9. Kirino Y, Sei M, Kawazoe K, Minakuchi K, Sato Y. Plasma dipeptidyl peptidase 4 activity correlates with body mass index and the plasma adiponectin concentration in healthy young people. Endocr J 2012;59:949-953. ArticlePubMed
- 10. Scirica BM, Bhatt DL, Braunwald E, Steg PG, Davidson J, Hirshberg B, Ohman P, Frederich R, Wiviott SD, Hoffman EB, Cavender MA, Udell JA, Desai NR, Mosenzon O, McGuire DK, Ray KK, Leiter LA, Raz I. SAVOR-TIMI 53 Steering Committee and Investigators. Saxagliptin and cardiovascular outcomes in patients with type 2 diabetes mellitus. N Engl J Med 2013;369:1317-1326. ArticlePubMed
- 11. White WB, Cannon CP, Heller SR, Nissen SE, Bergenstal RM, Bakris GL, Perez AT, Fleck PR, Mehta CR, Kupfer S, Wilson C, Cushman WC, Zannad F. EXAMINE Investigators. Alogliptin after acute coronary syndrome in patients with type 2 diabetes. N Engl J Med 2013;369:1327-1335. ArticlePubMed
- 12. Monami M, Ahren B, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and cardiovascular risk: a meta-analysis of randomized clinical trials. Diabetes Obes Metab 2013;15:112-120. ArticlePubMed
- 13. Patil HR, Al Badarin FJ, Al Shami HA, Bhatti SK, Lavie CJ, Bell DS, O'Keefe JH. Meta-analysis of effect of dipeptidyl peptidase-4 inhibitors on cardiovascular risk in type 2 diabetes mellitus. Am J Cardiol 2012;110:826-833. ArticlePubMed
- 14. Teramoto T, Sasaki J, Ishibashi S, Birou S, Daida H, Dohi S, Egusa G, Hiro T, Hirobe K, Iida M, Kihara S, Kinoshita M, Maruyama C, Ohta T, Okamura T, Yamashita S, Yokode M, Yokote K. Diabetes mellitus. Executive summary of the Japan Atherosclerosis Society (JAS) guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan: 2012 version. J Atheroscler Thromb 2014;21:93-98. PubMed
- 15. Sakamoto Y, Oyama J, Ikeda H, Kuroki S, Gondo S, Iwamoto T, Uchida Y, Kodama K, Hiwatashi A, Shimomura M, Taguchi I, Inoue T, Node K. S-DOG investigators. Effects of sitagliptin beyond glycemic control: focus on quality of life. Cardiovasc Diabetol 2013;12:35ArticlePubMedPMCPDF
- 16. Ayaori M, Iwakami N, Uto-Kondo H, Sato H, Sasaki M, Komatsu T, Iizuka M, Takiguchi S, Yakushiji E, Nakaya K, Yogo M, Ogura M, Takase B, Murakami T, Ikewaki K. Dipeptidyl peptidase-4 inhibitors attenuate endothelial function as evaluated by flow-mediated vasodilatation in type 2 diabetic patients. J Am Heart Assoc 2013;2:e003277ArticlePubMedPMC
- 17. Monami M, Lamanna C, Desideri CM, Mannucci E. DPP-4 inhibitors and lipids: systematic review and meta-analysis. Adv Ther 2012;29:14-25. ArticlePubMedPDF
- 18. Flock G, Baggio LL, Longuet C, Drucker DJ. Incretin receptors for glucagon-like peptide 1 and glucose-dependent insulinotropic polypeptide are essential for the sustained metabolic actions of vildagliptin in mice. Diabetes 2007;56:3006-3013. ArticlePubMedPDF
- 19. Hsieh J, Longuet C, Baker CL, Qin B, Federico LM, Drucker DJ, Adeli K. The glucagon-like peptide 1 receptor is essential for postprandial lipoprotein synthesis and secretion in hamsters and mice. Diabetologia 2010;53:552-561. ArticlePubMedPDF
REFERENCES
Effects of dipeptidyl peptidase-4 inhibitor treatment on (A) glycosylated hemoglobin (HbA1c) and (B) blood glucose levels. Values from before the onset of treatment and after 12 months of treatment are compared. aP<0.01.
Association between baseline glycosylated hemoglobin (HbA1c) level and changes in HbA1c after 12 months of treatment.
Clinical characteristics of study subjects
Values are presented as number (%) or mean±standard deviation.
HbA1c, glycosylated hemoglobin; LDL-C, low density lipoprotein cholesterol; HDL-C, high density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; DPP-4, dipeptidyl peptidase-4.
aNational Glycohemoglobin Standardization Program reference value.
Multiple regression analysis for determinants of degree of decrease in HbA1c level after 12 months of treatment: ΔHbA1c (baseline to 12 months)
Figure & Data
References
Citations

- Endogenous GLP-1 levels play an important role in determining the efficacy of DPP-IV Inhibitors in both prediabetes and type 2 diabetes
Shiau Chin Chong, Norlela Sukor, Sarah Anne Robert, Kim Fong Ng, Nor Azmi Kamaruddin
Frontiers in Endocrinology.2022;[Epub] CrossRef - Dipeptidyl peptidase‐4 inhibitor, anagliptin, alters hepatic insulin clearance in relation to the glycemic status in Japanese individuals with type 2 diabetes
Takahiro Abe, Yasuhiro Matsubayashi, Sayaka Muragishi, Akihiro Yoshida, Hideki Suganami, Kenichi Furusawa, Kazuya Fujihara, Shiro Tanaka, Kohei Kaku, Hirohito Sone
Journal of Diabetes Investigation.2021; 12(10): 1805. CrossRef - Effects of omarigliptin on glucose variability and oxidative stress in type 2 diabetes patients: A prospective study
Makoto Ohara, Hiroe Nagaike, Tomoki Fujikawa, Yo Kohata, Maiho Ogawa, Takemasa Omachi, Risa Sasajima, Hirotoshi Chiba, Toshimasa Ara, Ayuka Sugawara, Munenori Hiromura, Michishige Terasaki, Yusaku Mori, Tomoyasu Fukui, Tsutomu Hirano, Hiroki Yokoyama, Sho
Diabetes Research and Clinical Practice.2021; 179: 108999. CrossRef - Factors associated with the glucose‐lowering efficacy of sitagliptin in Japanese patients with type 2 diabetes mellitus: Pooled analysis of Japanese clinical trials
Naoko Tajima, Jun‐ichi Eiki, Taro Okamoto, Kotoba Okuyama, Masaru Kawashima, Samuel S Engel
Journal of Diabetes Investigation.2020; 11(3): 640. CrossRef - Efficacy and safety of evogliptin versus sitagliptin as add on to metformin alone in a combined russian-korean population. Evo-combi trial
Alina Y. Babenko, Anna A. Mosikian, Igor E. Makarenko, Victoriya V. Leusheva, Evgeny V. Shlyakhto
Diabetes mellitus.2018; 21(4): 241. CrossRef - Predictors for the Treatment Effect of Sodium Glucose Co-transporter 2 Inhibitors in Patients with Type 2 Diabetes Mellitus
Shusuke Yagi, Ken-ichi Aihara, Takeshi Kondo, Kiyoe Kurahashi, Sumiko Yoshida, Itsuro Endo, Daiju Fukuda, Yutaka Nakaya, Kin-ichiro Suwaki, Takashi Takeji, Toshihiro Wada, Hotimah Masdan Salim, Saori Hama, Tomomi Matsuura, Takayuki Ise, Kenya Kusunose, Ko
Advances in Therapy.2018; 35(1): 124. CrossRef - Ethnic Differences in Efficacy and Safety of Alogliptin: A Systematic Review and Meta-Analysis
Yuli Cai, Tianshu Zeng, Zhongyuan Wen, Lulu Chen
Diabetes Therapy.2018; 9(1): 177. CrossRef - Effectiveness prediction of Evogliptin treatment in type 2 diabetes mellitus in russian-korean population
Anna A. Mosikian, Alina Y. Babenko, Yulia A. Sevastyanova, Roman V. Drai, Evgenij V. Shlyakhto
Diabetes mellitus.2018; 21(5): 333. CrossRef - Efficacy and Safety of Linagliptin in 2681 Asian Patients Stratified by Age, Obesity, and Renal Function: A Pooled Analysis of Randomized Clinical Trials
Guang Ning, Tushar Bandgar, Uwe Hehnke, Jisoo Lee, Juliana C. N. Chan
Advances in Therapy.2017; 34(9): 2150. CrossRef - Potential regulatory mechanisms of lncRNA in diabetes and its complications
Shui-Dong Feng, Ji-Hua Yang, Chao Hua Yao, Si-Si Yang, Ze-Mei Zhu, Di Wu, Hong-Yan Ling, Liang Zhang
Biochemistry and Cell Biology.2017; 95(3): 361. CrossRef - Baseline Body Mass Index and the Efficacy of Hypoglycemic Treatment in Type 2 Diabetes: A Meta-Analysis
Xiaoling Cai, Wenjia Yang, Xueying Gao, Lingli Zhou, Xueyao Han, Linong Ji, Francesco Giorgino
PLOS ONE.2016; 11(12): e0166625. CrossRef - Efficacy of hypoglycemic treatment in type 2 diabetes stratified by age or diagnosed age: a meta-analysis
Xiaoling Cai, Wenjia Yang, Yifei Chen, Xueying Gao, Lingli Zhou, Simin Zhang, Xueyao Han, Linong Ji
Expert Opinion on Pharmacotherapy.2016; 17(12): 1591. CrossRef - Pharmacokinetic, pharmacodynamic and clinical evaluation of saxagliptin in type 2 diabetes
Rose Anderson, Jennifer Hayes, Jeffrey W. Stephens
Expert Opinion on Drug Metabolism & Toxicology.2016; 12(4): 467. CrossRef - Letter: Predictive Factors for Efficacy of Dipeptidyl Peptidase-4 Inhibitors in Patients with Type 2 Diabetes Mellitus (Diabetes Metab J2015;39:342-7)
Ye An Kim
Diabetes & Metabolism Journal.2015; 39(5): 444. CrossRef - Response: Predictive Factors for Efficacy of Dipeptidyl Peptidase-4 Inhibitors in Patients with Type 2 Diabetes Mellitus (Diabetes Metab J2015;39:342-7)
Shusuke Yagi, Ken-ichi Aihara, Masataka Sata
Diabetes & Metabolism Journal.2015; 39(5): 446. CrossRef
- Figure
- Related articles
-
- Risk Prediction and Management of Chronic Kidney Disease in People Living with Type 2 Diabetes Mellitus
- Comparative Efficacy of Rosuvastatin Monotherapy and Rosuvastatin/Ezetimibe Combination Therapy on Insulin Sensitivity and Vascular Inflammatory Response in Patients with Type 2 Diabetes Mellitus
- Clinical and Lifestyle Determinants of Continuous Glucose Monitoring Metrics in Insulin-Treated Patients with Type 2 Diabetes Mellitus
- The Beneficial Effect of Glycemic Control against Adverse Outcomes in Patients with Type 2 Diabetes Mellitus and Chronic Kidney Disease
- Non-Alcoholic Fatty Liver Disease with Sarcopenia and Carotid Plaque Progression Risk in Patients with Type 2 Diabetes Mellitus

 KDA
KDA PubReader
PubReader Cite
Cite





