
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
Review
- Pathophysiology
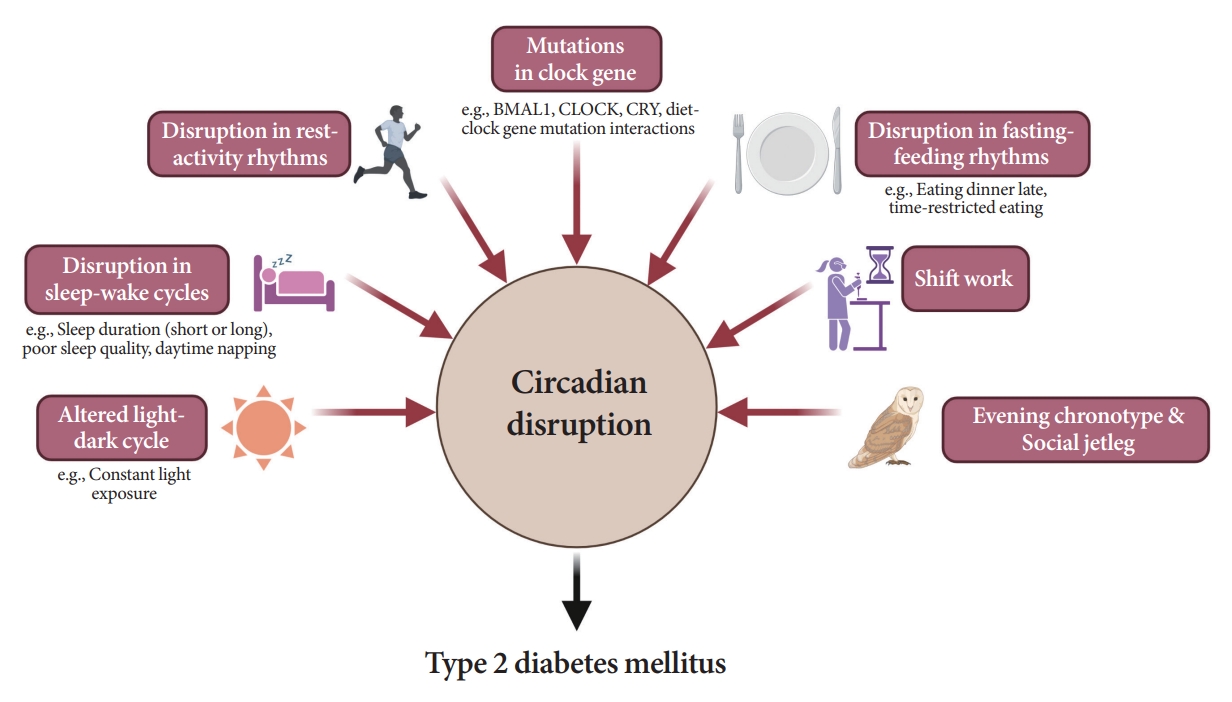
- Attention to Innate Circadian Rhythm and the Impact of Its Disruption on Diabetes
- Da Young Lee, Inha Jung, So Young Park, Ji Hee Yu, Ji A Seo, Kyeong Jin Kim, Nam Hoon Kim, Hye Jin Yoo, Sin Gon Kim, Kyung Mook Choi, Sei Hyun Baik, Nan Hee Kim
- Diabetes Metab J. 2024;48(1):37-52. Published online January 3, 2024
- DOI: https://doi.org/10.4093/dmj.2023.0193
- 2,489 View
- 230 Download
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Novel strategies are required to reduce the risk of developing diabetes and/or clinical outcomes and complications of diabetes. In this regard, the role of the circadian system may be a potential candidate for the prevention of diabetes. We reviewed evidence from animal, clinical, and epidemiological studies linking the circadian system to various aspects of the pathophysiology and clinical outcomes of diabetes. The circadian clock governs genetic, metabolic, hormonal, and behavioral signals in anticipation of cyclic 24-hour events through interactions between a “central clock” in the suprachiasmatic nucleus and “peripheral clocks” in the whole body. Currently, circadian rhythmicity in humans can be subjectively or objectively assessed by measuring melatonin and glucocorticoid levels, core body temperature, peripheral blood, oral mucosa, hair follicles, rest-activity cycles, sleep diaries, and circadian chronotypes. In this review, we summarized various circadian misalignments, such as altered light-dark, sleep-wake, rest-activity, fasting-feeding, shift work, evening chronotype, and social jetlag, as well as mutations in clock genes that could contribute to the development of diabetes and poor glycemic status in patients with diabetes. Targeting critical components of the circadian system could deliver potential candidates for the treatment and prevention of type 2 diabetes mellitus in the future.
Original Article
- Role of Glucocorticoid Receptor on Insulin Secretion and Synthesis in INS-1 Cells.
- Ju Yeon Yang, Myong Su Kang, Tak Ho Song, In Kook Jeong, Pyong Ju Seo, Hee Jin Kim
- Korean Diabetes J. 2006;30(6):428-434. Published online November 1, 2006
- DOI: https://doi.org/10.4093/jkda.2006.30.6.428
- 1,606 View
- 24 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Glucocorticoids play important roles in the regulation of glucose homeostasis. It is well known that glucocorticoids reduce hepatic and peripheral tissue sensitivity to insulin, but the roles of glucocorticoids on insulin secretion and synthesis in pancreatic beta cells are still unclear. We have investigated the direct effects of glucocorticoids on insulin secretion and synthesis in rat insulinoma (INS-1) cells. METHODS: Insulin content and 11.2 mM glucose-stimulated insulin secretion (GSIS) were measured in INS-1 cells after culture with or without 1 micrometer dexamethasone (DEX). Preproinsulin mRNA levels were analyzed by real-time RT-PCR and normalized to the internal control. Effect of RU486 on DEX-induced inhibition of GSIS and preproinsulin mRNA synthesis was evaluated. RESULTS: Insulin content of INS-1 cells cultured in RPMI containing 11.2 mM glucose in the presence of DEX was not different from that of control cells. After 1-h preincubation in 2.8 mM glucose, basal insulin secretion from cells treated with DEX did not differ from that of controls, but GSIS was significantly reduced in the cells treated with DEX in comparison to control cells. The expression of preproinsulin mRNA relative to beta-actin mRNA was also lower in the cells treated with DEX. Glucocorticoid receptor antagonist improved DEX-induced inhibition of GSIS and preproinsulin mRNA synthesis. CONCLUSION: DEX inhibited GSIS and preproinsulin mRNA synthesis in INS-1 cells. Glucocorticoid receptor antagonist ameliorated the reduced GSIS and preproinsulin mRNA synthesis induced by DEX.

 KDA
KDA First
First Prev
Prev





