
- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Search
- Page Path
- HOME > Search
Original Articles
- Metabolic Risk/Epidemiology
- Healthy Lifestyle and the Risk of Metabolic Dysfunction-Associated Fatty Liver Disease: A Large Prospective Cohort Study
- Qing Chang, Yixiao Zhang, Tingjing Zhang, Zuyun Liu, Limin Cao, Qing Zhang, Li Liu, Shaomei Sun, Xing Wang, Ming Zhou, Qiyu Jia, Kun Song, Yang Ding, Yuhong Zhao, Kaijun Niu, Yang Xia
- Received April 27, 2023 Accepted November 30, 2023 Published online March 19, 2024
- DOI: https://doi.org/10.4093/dmj.2023.0133 [Epub ahead of print]
- 730 View
- 46 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
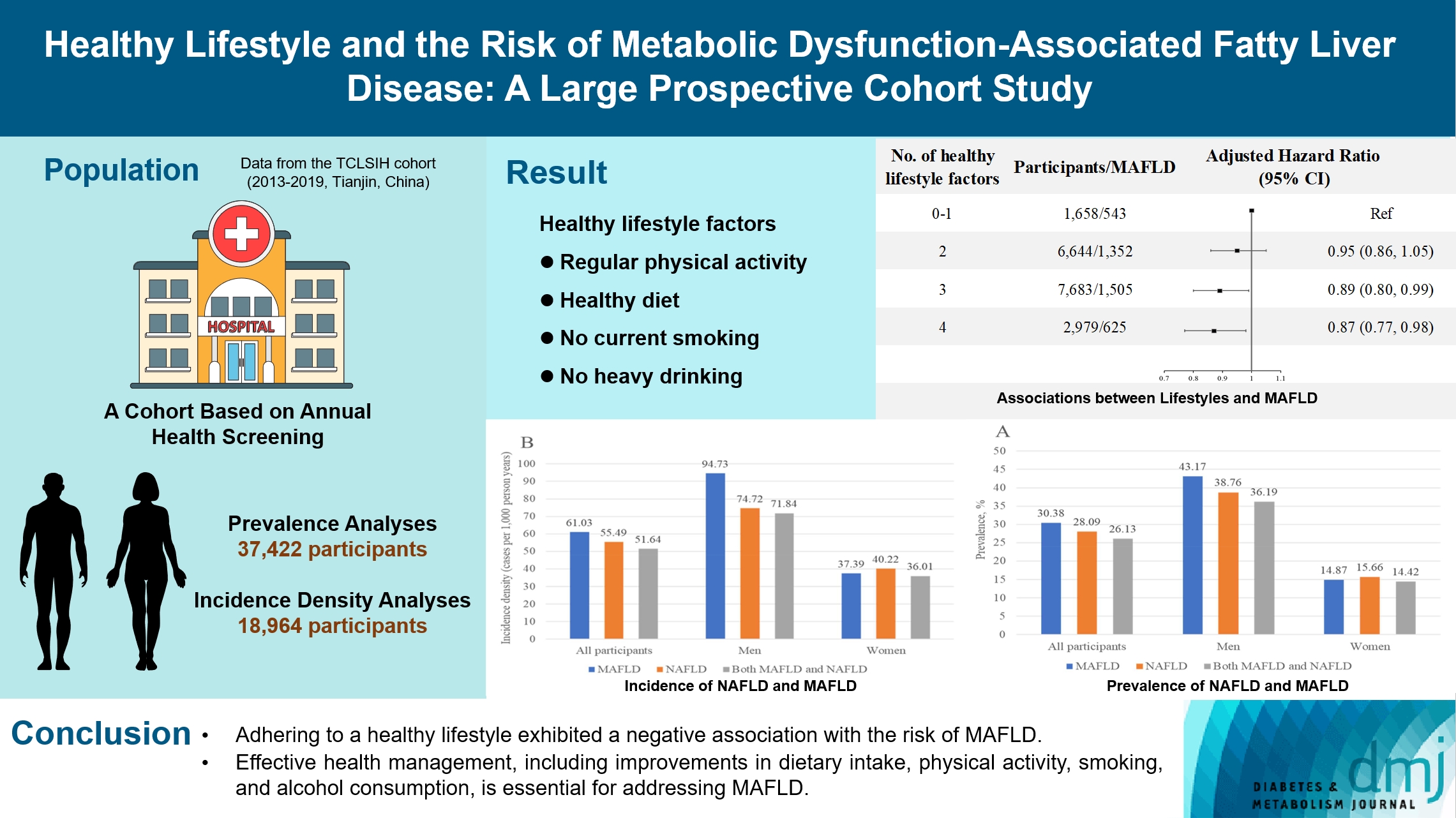
The incidence density of metabolic dysfunction-associated fatty liver disease (MAFLD) and the effect of a healthy lifestyle on the risk of MAFLD remain unknown. We evaluated the prevalence and incidence density of MAFLD and investigated the association between healthy lifestyle and the risk of MAFLD.
Methods
A cross-sectional analysis was conducted on 37,422 participants to explore the prevalence of MAFLD. A cohort analysis of 18,964 individuals was conducted to identify the incidence of MAFLD, as well as the association between healthy lifestyle and MAFLD. Cox proportional hazards regression was used to calculate the hazard ratio (HR) and 95% confidence interval (CI) with adjustments for confounding factors.
Results
The prevalence of MAFLD, non-alcoholic fatty liver disease, and their comorbidities were 30.38%, 28.09%, and 26.13%, respectively. After approximately 70 thousand person-years of follow-up, the incidence densities of the three conditions were 61.03, 55.49, and 51.64 per 1,000 person-years, respectively. Adherence to an overall healthy lifestyle was associated with a 19% decreased risk of MAFLD (HR, 0.81; 95% CI, 0.72 to 0.92), and the effects were modified by baseline age, sex, and body mass index (BMI). Subgroup analyses revealed that younger participants, men, and those with a lower BMI experienced more significant beneficial effects from healthy lifestyle.
Conclusion
Our results highlight the beneficial effect of adherence to a healthy lifestyle on the prevention of MAFLD. Health management for improving dietary intake, physical activity, and smoking and drinking habits are critical to improving MAFLD.
- Technology/Device
- Glycemia according to the Use of Continuous Glucose Monitoring among Adults with Type 1 Diabetes Mellitus in Korea: A Real-World Study
- You-Bin Lee, Minjee Kim, Jae Hyeon Kim
- Diabetes Metab J. 2023;47(3):405-414. Published online March 6, 2023
- DOI: https://doi.org/10.4093/dmj.2022.0032

- 3,417 View
- 123 Download
- 3 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
We explored the association between continuous glucose monitoring (CGM) use and glycemia among adults with type 1 diabetes mellitus (T1DM) and determined the status of CGM metrics among adults with T1DM using CGM in the real-world.
Methods
For this propensity-matched cross-sectional study, individuals with T1DM who visited the outpatient clinic of the Endocrinology Department of Samsung Medical Center between March 2018 and February 2020 were screened. Among them, 111 CGM users (for ≥9 months) were matched based on propensity score considering age, sex, and diabetes duration in a 1:2 ratio with 203 CGM never-users. The association between CGM use and glycemic measures was explored. In a subpopulation of CGM users who had been using official applications (not “do-it-yourself” software) such that Ambulatory Glucose Profile data for ≥1 month were available (n=87), standardized CGM metrics were summarized.
Results
Linear regression analyses identified CGM use as a determining factor for log-transformed glycosylated hemoglobin. The fully-adjusted odds ratio (OR) and 95% confidence interval (CI) for uncontrolled glycosylated hemoglobin (>8%) were 0.365 (95% CI, 0.190 to 0.703) in CGM users compared to never-users. The fully-adjusted OR for controlled glycosylated hemoglobin (<7%) was 1.861 (95% CI, 1.119 to 3.096) in CGM users compared to never-users. Among individuals who had been using official applications for CGM, time in range (TIR) values within recent 30- and 90-day periods were 62.45%±16.63% and 63.08%±15.32%, respectively.
Conclusion
CGM use was associated with glycemic control status among Korean adults with T1DM in the real-world, although CGM metrics including TIR might require further improvement among CGM users. -
Citations
Citations to this article as recorded by- Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis
Ji Yoon Kim, Sang-Man Jin, Sarah B. Andrade, Boyang Chen, Jae Hyeon Kim
Diabetes Technology & Therapeutics.2024;[Epub] CrossRef - Accuracy and Safety of the 15-Day CareSens Air Continuous Glucose Monitoring System
Kyung-Soo Kim, Seung-Hwan Lee, Won Sang Yoo, Cheol-Young Park
Diabetes Technology & Therapeutics.2024; 26(4): 222. CrossRef - Navigating the Seas of Glycemic Control: The Role of Continuous Glucose Monitoring in Type 1 Diabetes Mellitus
Jun Sung Moon
Diabetes & Metabolism Journal.2023; 47(3): 345. CrossRef - Smart Insulin Pen: Managing Insulin Therapy for People with Diabetes in the Digital Era
Jee Hee Yoo, Jae Hyeon Kim
The Journal of Korean Diabetes.2023; 24(4): 190. CrossRef
- Real-World Continuous Glucose Monitoring Data from a Population with Type 1 Diabetes in South Korea: Nationwide Single-System Analysis

 KDA
KDA
 First
First Prev
Prev





