The Phospholipid Linoleoylglycerophosphocholine as a Biomarker of Directly Measured Insulin Resistance
Article information
Abstract
Background
Plasma concentrations of some lysophospholipids correlate with metabolic alterations in humans, but their potential as biomarkers of insulin resistance (IR) is insufficiently known. We aimed to explore the association between plasma linoleoylglycerophosphocholine (LGPC) and objective measures of IR in adults with different metabolic profiles.
Methods
We studied 62 men and women, ages 30 to 69 years, (29% normal weight, 59% overweight, 12% obese). Participants underwent a 5-point oral glucose tolerance test (5p-OGTT) from which we calculated multiple indices of IR and insulin secretion. Fifteen participants additionally underwent a hyperinsulinemic-euglycemic clamp for estimation of insulin-stimulated glucose disposal. Plasma LGPC was determined using high performance liquid chromatography/time-of-flight mass spectrometry. Plasma LGPC was compared across quartiles defined by the IR indices.
Results
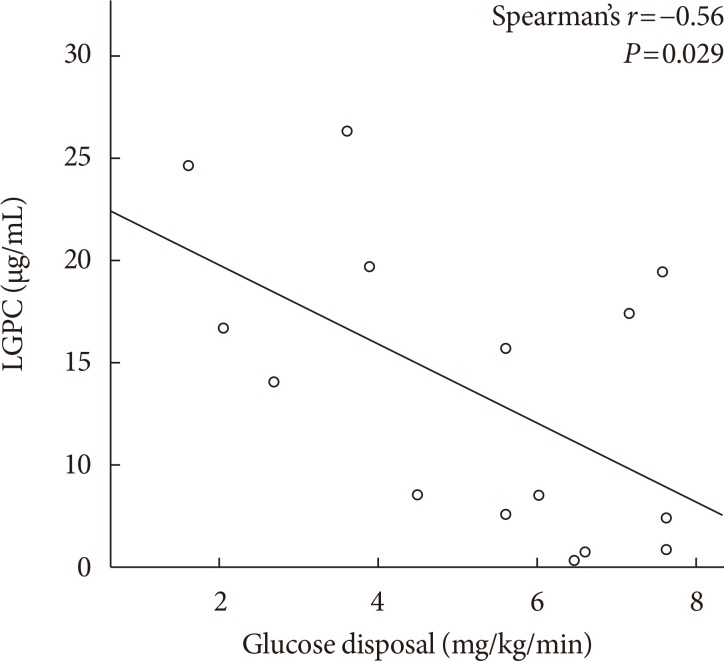
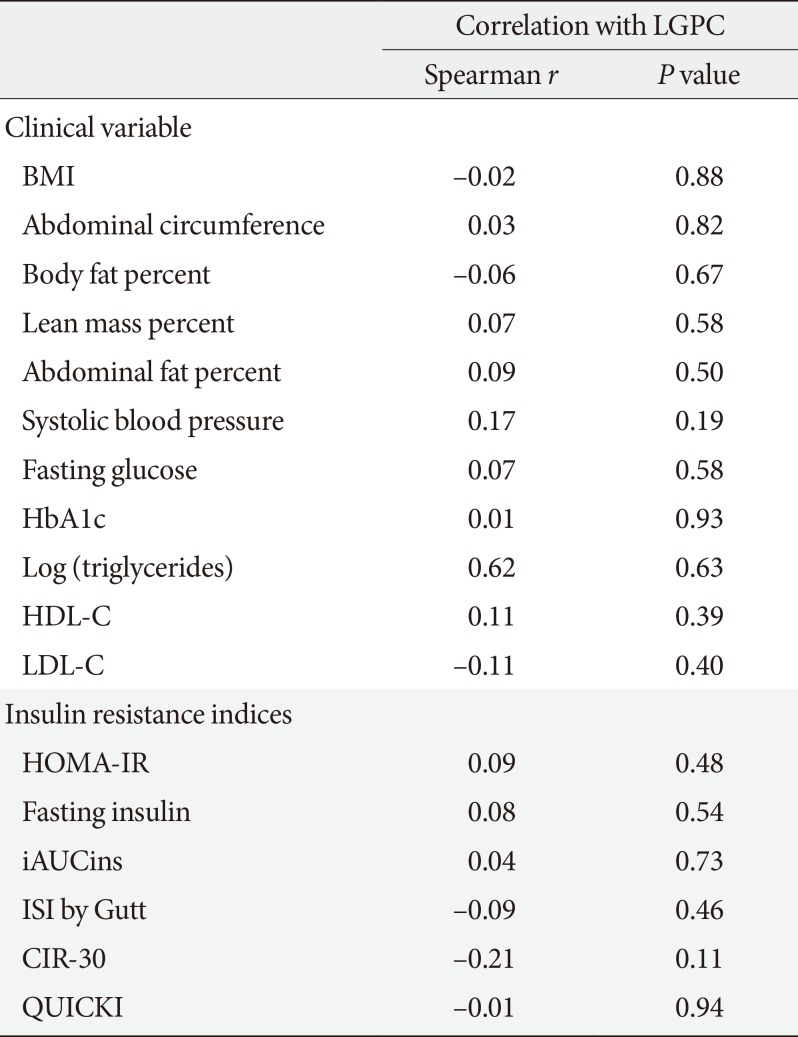
Mean LGPC was 15.4±7.6 ng/mL in women and 14.1±7.3 ng/mL in men. LGPC did not correlate with body mass in-dex, percent body fat, waist circumference, blood pressure, glycosylated hemoglobin, log-triglycerides, or high density lipoprotein cholesterol. Plasma LGPC concentrations was not systematically associated with any of the studied 5p-OGTT-derived IR indices. However, LGPC exhibited a significant negative correlation with glucose disposal in the clamp (Spearman r=−0.56, P=0.029). Despite not being diabetic, participants with higher plasma LGPC exhibited significantly higher post-challenge plasma glucose excursions in the 5p-OGTT (P trend=0.021 for the increase in glucose area under the curve across quartiles of plasma LGPC).
Conclusion
In our sample of Latino adults without known diabetes, LGPC showed potential as a biomarker of IR and impaired glucose metabolism.
INTRODUCTION
Insulin resistance (IR) is a physiological alteration involved in the pathogenesis of type 2 diabetes mellitus (T2DM), several forms of cancer and many other diseases [1]. Despite its importance, IR is often overlooked in the clinical setting because of the high difficulty and cost of hyperinsulinemic/euglycemic clamp, the gold standard method for measurement of insulin action [2]. Several phospholipids of the phosphatidylcholine (PC) family are associated with metabolic abnormalities and could serve as potential IR biomarkers [3]. However, the robustness of their association with IR and its diagnostic performance is insufficiently known, particularly in non-Caucasian populations.
The phospholipid composition of cell membranes largely determines their biochemical properties, including responsiveness to hormones [4]. In fact, the phospholipid composition of muscle cell membranes modulates insulin action, so that a reduction in membrane polyunsaturated fatty acids (PUFAs) correlates with reduced insulin sensitivity [5]. PCs are some of the most abundant constituents of cell membranes, and there are multiple different PC species depending on the fatty acids present in the sn-1 and sn-2 positions of their glyc-erol backbone. Linoleoylglycerophosphocholine (LGPC) is a lysophosphatidylcholine in which linoleic acid (18:2, n-6) occupies the sn-1 position, and there is no fatty acid in the sn-2 position. Plasma concentrations of LGPC have been associated with increased risk of impaired glucose tolerance or T2DM [678]. In two cohorts of Caucasian participants, LGPC was nega-tively associated with dysglycemia even after adjustment for major known T2DM risk factors [9].
Even though prior evidence supports an association between plasma LGPC and the incidence of clinical T2DM, the relationship between LGPC and objectively measured IR is not well known. In addition, virtually no study has explored the potential of LGPC as an IR biomarker in non-Caucasian populations. The development of new and practical IR biomarkers requires their evaluation in different populations and an exploration of their diagnostic potential in individuals with different clinical characteristics but without overt T2DM. Identification of novel IR biomarkers is a starting point for the development of diagnostic tests that identify subjects most likely to benefit from preventive interventions. Biomarkers may also reveal new pathogenic pathways involved in IR and T2DM.
With this background, we evaluated the association between fasting plasma levels of LGPC and objective indicators of IR derived from a 5-point oral glucose tolerance test (5p-OGTT) and a hyperinsulinemic-euglycemic clamp in adults with different metabolic risk profiles but without a prior diagnosis of T2DM.
METHODS
Study subjects
We included adults of both sexes, aged 30 to 69 years, with no prior diagnosis of diabetes mellitus. We intentionally included patients with a wide range of body mass index (BMI) in order to achieve considerable variation in insulin sensitivity in the study sample. Exclusion criteria were pregnancy, use of any anti-diabetic medication, endocrine diseases, and use of anticoagulants. We also excluded patients who were acutely ill, or with plasma C-reactive protein above 10 mg/L.
Clinical assessment, 5p-OGTT, and hyperinsulinemic clamp
We measured in all participants resting blood pressure (mean of two measurements), height, weight and abdominal circumference. Percent body fat, percent lean mass, and percent abdominal fat were determined with a tetrapolar biological impedance meter (BC545; TANITA, Tokyo, Japan). Fasting blood samples were obtained in potassium oxalate tubes for measurement of fasting glucose and in EDTA (ethylenediaminetetraacetic acid) tubes for all other determinations including LGPC. After prompt plasma separation, a protease inhibitor cocktail was added and total plasma was separated in aliquots and frozen at −80℃ until analyzed.
For the 5p-OGTT, patients arrived after a 8- to 12-hour fast and received a load of 75 g of glucose (20% w/v in water) to be consumed in less than 5 minutes. Blood samples for the measurement of plasma glucose and insulin were drawn at 0, 30, 60, 90, and 120 minutes post-glucose challenge. Patients could not smoke, ingest food or do significant physical activity during the 5p-OGTT. A subgroup of 15 participants selected entirely at random from the complete study sample additionally underwent a hyperinsulinemic-euglycemic clamp as described by DeFronzo et al. [10]. After an overnight fast, subjects were admitted to a clinical research center, where two intravenous catheters were placed in the antecubital area of both arms for simultaneous insulin and dextrose infusion. Another catheter was placed in the dorsal area of the hand ipsilateral to the dextrose infusion for blood sampling. The hand for capillary blood sampling and glucose measurement was placed inside a heated-hand box (The University of Vermont Medical, Burlington, VT, USA). This device keeps a constant temperature (56℃ to 58℃) in order to arterialize venous blood. A short-acting human insulin analog (Humalog; Lilly, Indianapolis, IN, USA) was infused at an initial priming rate of 100 mU/m2/min that was reduced to 90 mU/m2/min after 2 minutes and then in 20 mU/m2/min steps every 2 minutes until a dose of 40 mU/m2/min was reached. This rate was then kept constant for the next 110 minutes, the whole procedure lasted 120 minutes. A variable infusion of 20% glucose was started at the 4th minute and adjusted every 5 minutes in order to maintain the arterialized venous glucose concentration at 100 mg/dL (95 to 105 mg/dL). Plasma glucose was measured by the glucose oxidase method using Accu-Chek Performa glucose meters (Roche, Mannheim, Germany). The main result of the clamp was whole-body insulin-stimulated glucose disposal at steady state (M-value) (mg [glucose]/kg [body weight]/min).
Biochemical measurements
Fasting plasma glucose, plasma lipids, and creatinine were measured with enzymatic-colorimetric assays (Biosystems, Barcelona, Spain). Glycosylated hemoglobin (HbA1c) was determined using a National Glycohemoglobin Standardization Program-certified boronate affinity technique (NycoCard Reader II; Alere Technologies, Oslo, Norway). Plasma LGPC was measured using high performance liquid chromatography-time of flight mass spectrometry (HPLC-MS-QToF). Fifty microliters of each fasting plasma sample were subjected to protein precipitation by mixing with 250 µL of methanol. After centrifugation, the clear supernatant was separated and an aliquot of it injected onto a HPLC-MS-QToF system (Agilent Technologies 1260 Infinity HPLC/Agilent 6520 MS system; Agilent Technologies, Santa Clara, CA, USA). LGPC was eluted with a 0.01% formic acid in water/acetonitrile-water-ammonium formate (700:300:2.7) gradient on a Thermo BioBasic SCX (Waltham, MA, USA) column (50×2.1 mm, 5 µm) at a mobile phase flow rate of 0.5 mL/min during 2.5 minutes plus column equilibration, at 40℃. Ionization was achieved in positive ESI mode. We monitored a single ion with m/z value=520.34. LGPC standards for calibration curves were custom-made by Ambinter (Orleans, France).
Statistical analyses
Using plasma glucose and insulin values from the 5p-OGTT, we calculated IR indices based on fasting values: homeostasis model assessment of insulin resistance (HOMA-IR; higher values indicate more IR) [11], quantitative insulin sensitivity check index (QUICKI; higher values indicate less IR) [12]; IR indices based on post-load insulin and glucose values: incremental area under the insulin curve (iAUCin; higher values numbers indicate more IR) [13], insulin sensitivity index by Gutt (ISI-Gutt; higher values numbers indicate less IR) [14]; and insulin secretion: corrected insulin response at 30 minutes (CIR-30; higher values indicate a sharper first peak of insulin secretion) [15].
Plasma triglycerides had a very skewed distribution and hence were log-transformed before analyses. Given the small sample size and non-normal nature of most variables under study, comparisons of numerical variables between groups were made with the non-parametric Mann-Whitney U test. Comparisons of categorical variables between groups were made with exact chi-square tests. Participants were placed in quartiles of LGPC and the mean area under the glucose curve in the 5p-OGTT (AUCglu) was calculated for each quartile. Reported P values for trend correspond to those associated with the coefficient of the slope in a linear regression in which mean LGPC values in each of the LGPC quartiles were the independent variables, and mean values of AUCglu in each quartile were the dependent variable. The linear correlations between IR indices and LGPC, and between clinical variables and LGPC were assessed using Spearman correlation coefficients. For exploratory receiver operating characteristic (ROC) analyses, IR was defined as belonging to the highest quartile of iAUCins, or as belonging to the lowest quartile of glucose disposal. All statistical tests were two-tailed at a significance level of 0.05. Analyses were performed with SPSS version 23.0 (IBM Co., Armonk, NY, USA).
Ethical considerations
The study was approved by the Institutional Review Board of Universidad de los Andes according to minute 307 of 2014. The study complied with scientific, technical, and administrative norms for health research as mandated by resolution 008430–1993 of the Colombian Ministry of Health and with the principles stated by the Declaration of Helsinki. All study subjects underwent a thorough informed consent procedure and provided written informed consent.
RESULTS
Sixty-two patients were enrolled, 44% men and 56% women. Mean age was 50.7±10.2, BMI 26.7 kg/m2 (range, 16.0 to 39.9 kg/m2), abdominal circumference was 96.4±12.4 cm in men and 82.9±11.8 cm in women. HbA1c was 5.64%±1.05% in men and 5.45%±1.02% in women. Mean blood pressure, total cholesterol, low density lipoprotein cholesterol, creatinine, and estimated glomerular filtration rate were within the normal range. Mean high density lipoprotein cholesterol (HDL-C) was normal in women at 48.8±12.3 mg/dL and borderline low in men at 39.0±10.5 mg/dL. There was no significant difference in plasma LGPC levels by sex (P=0.56). Characteristics of study participants are summarized in Table 1.
Association between LGPC and clinical and biochemical variables
LGPC did not show a significant linear correlation with BMI, abdominal circumference, fat body percent, abdominal fat percent, blood pressure, HbA1c, fasting glucose, triglycerides, or HDL-C (Table 2). However, participants with higher plasma LGPC were characterized by higher glycemic excursions during the 5p-OGTT. There was a monotonic increase in the AUCglu from LGPC quartile 1 (AUCglu=237.7 mg/dL*hr) to LGPC quartile 4 (AUCglu=294.3 mg/dL*hr) (P trend=0.021) (Fig. 1).

Correlation between plasma LGPC and insulin resistance indices, and between plasma LGPC and insulin resistance-associated clinical and biochemical variables
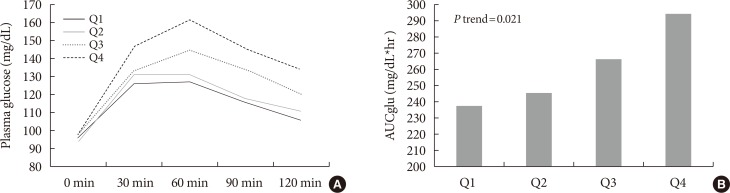
(A) Glycemic levels across the 5-point oral glucose tolerance test (5p-OGTT) by quartiles of plasma linoleoylglycerophosphocholine (LGPC). (B) Area under the glucose curve (AUCglu) in the 5p-OGTT across quartiles of plasma LGPC.
We performed a stepwise multivariable linear regression analysis that included AUCglu as the outcome variable, and the main determinants of IR (age, sex, and BMI) plus LGPC as predictors. In this mutually-adjusted model, only BMI and plasma LGPC were significant predictors of AUCglu (P=0.018 for LGPC, P<0.001 for BMI), while sex (P=0.76), and age (P=0.25) were not.
Association between LGPC and indirect IR indices
Plasma LGPC concentrations did not correlate with any of the 5p-OGTT-derived IR indices. We only found a non-significant trend towards a negative correlation with CIR-30 (r=−0.21, P=0.11) (Table 2).
Hyperinsulinemic-euglycemic clamp and ROC curve analysis
Mean whole-body insulin-stimulated glucose disposal at steady state was 5.2±1.93 mg/kg/min. Clinical and demographic characteristics of the group of patients who underwent the glucose clamp were on average not different from those of the complete study sample (Table 3). Glucose disposal was negatively correlated with body fat percent (r=−0.52, P=0.021), and positively with the ISI (r=0.52, P=0.023). Men had a non-significantly higher glucose disposal than women (5.53 mg/kg/min vs. 4.94 mg/kg/min, P=0.43). Plasma LGPC levels showed a substantial negative correlation with glucose disposal (r=−0.56, P=0.029) (Fig. 2). In an exploratory ROC curve analysis, a plasma LGPC cutoff of 8.6 µg/mL would have 100% sensitivity and 67% specificity for the detection of IR according to the glucose disposal definition (C-statistic, 0.85).

Demographic, clinical, and biochemical characteristics of participants, according to their participation in the hyperinsulinemic euglycemic clamp
DISCUSSION
Despite its high prevalence and great relevance, IR remains underdiagnosed and undertreated due the difficulty and expense of its determination. In our study we measured fasting concentrations of LGPC, a phospholipid with potential as an IR biomarker, in individuals without obvious IR (i.e., no diagnosis of diabetes, polycystic ovary syndrome or other endocrine disease), but with different degrees of body adiposity.
Our results showed that LGPC had a strong negative correlation with glucose disposal in the clamp, despite not being associated with the more traditional surrogates of IR. Furthermore, in our sample of non-diabetic individuals, those with higher plasma LGPC had systematically higher glycemic excursions during the 5p-OGTT. These two findings suggest that LGPC has promise as a biomarker of IR and early deterioration of glucose metabolism in situations in which it would not be suspected clinically. Our sample size was insufficient for a thorough diagnostic evaluation of LGPC, but an exploratory ROC curve analysis in which belonging to the lowest quartile of glucose disposal was the outcome, a plasma LGPC cutoff of 8.6 µg/mL showed very high sensitivity and acceptable specificity. A major issue influencing the viability of new biomarkers is the accuracy of the measurement methodology. For our HPLC-MS-based measurement of LGPC, we obtained deviations between 5% to 10% when running prepared samples of known concentration, similar to what has been reported in prior studies [16].
Even though multiple other studies have examined a potential role for other complex phospholipids like lipopolysaccharides on the pathogenesis of IR through proinflammatory pathways [1718], studies on the relationship of plasma LGPC with direct measures of IR are very scarce. A lipidomic study in healthy young women found a negative association of LGPC with waist circumference after adjusting for the most relevant confounders, but no association between LGPC and HOMA-IR, a measure of IR in the fasting state [19]. Another study explored levels of total plasma lysophosphatidylcholines in patients with obesity and/or diabetes, both of which exhibited significantly decreased levels compared with lean subjects [16]. In an observational subanalysis of a clinical trial of omega-3 PUFAs, obese patients showed lower plasma LGPC than normal weight participants both at baseline and after omega-3 PUFAs supplementation [20]. This evidence supports the role of LGPC as a correlate of obesity and metabolic disease, but not necessarily as a true IR biomarker.
In order to explore possible biological mechanisms linking LGPC and IR, it is important to consider certain aspects of lysophospholipid metabolism. Plasma lysophosphadytilcholines may derive from PC by action of the enzyme lecithin cholesterol acyltransferase (LCAT). LCAT that travels with HDL in the blood removes fatty acids from the sn-1 or sn-2 positions of membrane-derived PC [1921]. Subjects with obesity and IR have higher circulating levels of LCAT [22]. This could in turn translate into more production and higher plasma concentrations of LGPC. Another explanation is that IR may impact the relative balance of PC and phosphatydilethanolamines (PE). About 70% of PC is synthesized via the cytidine 5′ diphosphate-choline pathway, which adds phosphocholine to the diacylglycerol scaffold, while 30% of PC is supplied from PE by the triple methylation of the choline head group, catalyzed by phosphatidylethanolamine N-methyltransferase (PEMT) [323]. Studies of human omental and subcutaneous adipose depots have found that obese, insulin resistant women have a lower adipose tissue expression of the PEMT gene and higher circulating PE levels [24]. Also, patients with non-alcoholic fatty liver disease have a reduced tissue PC/PE ratio [25]. Nonetheless, the exact biological mechanisms linking IR to changes in the production or degradation of LGCP remain to be fully elucidated.
The main limitations of our study are its cross-sectional nature and relatively small sample size, which limit the generalizability of our findings. Nonetheless, these observations show promise for LGPC as an IR biomarker, and will need to be confirmed in larger, prospective studies in which IR is also measured directly in study participants.
In conclusion, we found that plasma LGPC was not associated with clinical surrogates of IR or with IR indices derived from a 5p-OGTT. However, plasma LGPC had a negative association with insulin sensitivity as reflected by whole-body glucose disposal, and participants with higher fasting LGPC had significantly higher glucose excursions after ingestion of a glucose load. These findings suggest that LGPC has promise as an IR biomarker, especially in people in whom it would not be suspected otherwise. However, these findings will need to be expanded in larger, prospective studies.
ACKNOWLEDGMENTS
Funding for this project was provided by Departamento Administrativo de Ciencia, Tecnología e Innovación, COLCIENCIAS, Republic of Colombia. We want to thank our participants for their willingness and patience. We also thank Departamento Administrativo de Ciencia, Tecnología e Innovación, COLCIENCIAS, Republic of Colombia for its sponsorship.
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.