Serotonin as a New Therapeutic Target for Diabetes Mellitus and Obesity
Article information
Abstract
Serotonin (5-hydroxytryptamine [5-HT]) is a monoamine that has various functions in both neuronal and non-neuronal systems. In the central nervous system, 5-HT regulates mood and feeding behaviors as a neurotransmitter. Thus, there have been many trials aimed at increasing the activity of 5-HT in the central nervous system, and some of the developed methods are already used in the clinical setting as anti-obesity drugs. Unfortunately, some drugs were withdrawn due to the development of unwanted peripheral side effects, such as valvular heart disease and pulmonary hypertension. Recent studies revealed that peripheral 5-HT plays an important role in metabolic regulation in peripheral tissues, where it suppresses adaptive thermogenesis in brown adipose tissue. Inhibition of 5-HT synthesis reduced the weight gain and improved the metabolic dysfunction in a diet-induced obesity mouse model. Genome-wide association studies also revealed genetic associations between the serotonergic system and obesity. Several genetic polymorphisms in tryptophan hydroxylase and 5-HT receptors were shown to have strong associations with obesity. These results support the clinical significance of the peripheral serotonergic system as a therapeutic target for obesity and diabetes.
INTRODUCTION
Serotonin (5-hydroxytryptamine [5-HT]) has diverse functions in neuronal and non-neuronal systems. It acts as a hormone and a mitogen, as well as a neurotransmitter. At the time of its discovery (1918), 5-HT was thought to be a vasoconstrictor stored in platelets, so it was named serotonin after the Latin word serum and the Greek word tonic [12]. In 1937, 5-HT was found in the enterochromaffin (EC) cells of the gastrointestinal tract and named enteramine because it was thought to cause smooth muscle contraction in the gut [3]. It was not until 1952 that scientists reported 5-HT's role as a neurotransmitter [4]. It has since been discovered that 5-HT is a neurotransmitter associated with mood, behavior, sleep cycles, and appetite [2].
The synthesis of 5-HT is initiated with the hydroxylation of tryptophan by tryptophan hydroxylase (TPH), which is the rate-limiting enzyme for 5-HT synthesis. Thus, 5-HT production is regulated by the activity of TPH and the availability of tryptophan. The biological actions of 5-HT are mediated through the membrane-bound 5-HT receptors and terminated by its uptake into cells through a 5-HT transporter (SERT, Slc6a4) [5]. There are two isoforms of TPH. TPH1 is predominantly expressed in the peripheral tissues including the pineal gland, whereas TPH2 is exclusively expressed in the brain and the enteric nervous system [67]. Because 5-HT cannot cross the blood-brain barrier, the peripheral 5-HT system is functionally separate from the central 5-HT system.
Within the central and enteric nervous systems, 5-HT is synthesized and stored in the presynaptic neurons. Most 5-HT in the periphery is synthesized in EC cells [8] and platelets are the major storage site for circulating 5-HT. Several studies recently reported that 5-HT can be synthesized in other peripheral tissues (e.g., the heart, pancreas, and adipose tissue) and plays a role in a cell-autonomous manner [91011].
In this review, we will discuss the recent findings on the roles of central/peripheral 5-HT in metabolism and its clinical implications for the treatment of obesity.
THE 5-HT RECEPTOR FAMILIES
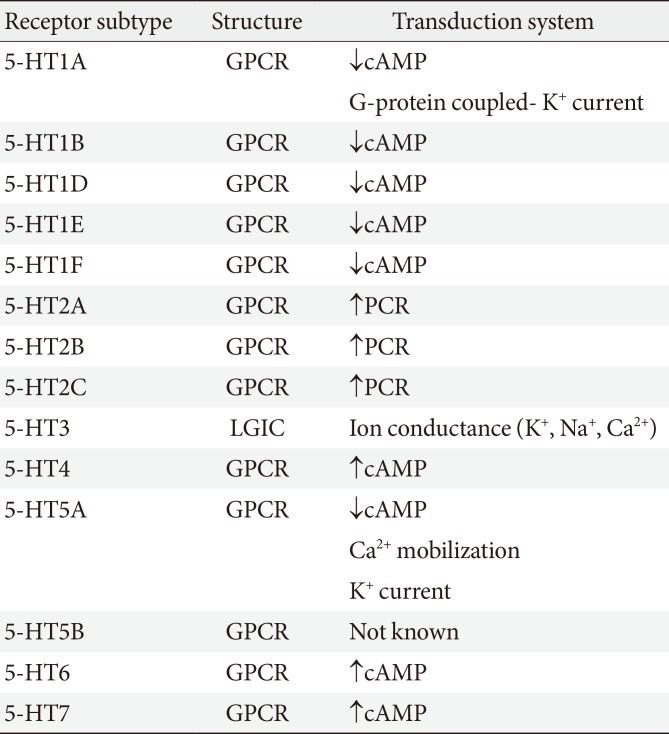
5-HT regulates various physiological and pathological processes, which are mediated through numerous 5-HT receptors. The 5-HT receptors are phylogenetically ancient and present in the lowest invertebrates as well as the higher mammals [12]. More than fourteen 5-HT receptors in seven families are known to mediate the diverse effects of 5-HT (Table 1) [1314]. All 5-HT receptors are G-protein-coupled receptors except for the 5-HT3 receptor, which is a ligand-gated cation channel [1215].
The 5-HT1 receptor family consists of five receptors: the 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1E, and 5-HT1F receptors. All these receptors are Gαi/o protein-coupled receptors that inhibit adenyl cyclase and reduce the cyclic adenosine monophosphate levels. The 5-HT2 receptor family consists of the 5-HT2A, 5-HT2B, and 5-HT2C receptors. These receptors are Gαq/11 protein-coupled receptors that mediate excitatory neurotransmission. The 5-HT2A and 5-HT2C receptors are mainly expressed in the central nervous system (CNS). The 5-HT2B receptor is expressed in peripheral tissues including the liver, kidney, heart, and stomach. It is associated with cardiac function, valvular heart disease, and heart morphogenesis. The 5-HT3 receptor is a member of the Cys-loop ligand-gated ion channel family. This channel is formed by a homopentamer of 5-HT3A subunits or heteropentamer of 5-HT3A subunits with 5-HT3B-E subunits. Opening of the channel in response to 5-HT activates an inward current and depolarizes the membrane.
REGULATION OF APPETITE BY CENTRAL 5-HT
Appetite is regulated by hypothalamic feeding circuits. Briefly, anorexigenic proopiomelanocortin (POMC) neurons release α-melanocyte-stimulating hormone, the endogenous ligand for the melanocortin 4 receptor (MC4R), to reduce appetite and food intake. Orexigenic neuropeptide Y/agouti-related peptide (NPY/AgRP) neurons increase appetite and food intake by releasing the endogenous MC4R antagonist, AgRP, and they suppress POMC neurons by releasing γ-aminobutyric acid (GABA).
It has long been known that 5-HT in the hypothalamus plays an important role in the control of appetite and energy balance. Central 5-HT has suppressive effects on food intake and body weight. Fenfluramine, which enhances 5-HT release, is a prototypical agent that leads to serotonergic suppression of feeding [16]. 5-HT reuptake inhibitors and monoamine oxidase inhibitors, which can increase the central serotonergic activity, also reduce food intake [1718]. In contrast, inhibition of central 5-HT synthesis by intraventricular injection of a TPH inhibitor induced hyperphagia and obesity in rats [19].
Studies using receptor-specific knock-out (KO) mice have provided more precise insight into the roles of 5-HT in appetite regulation. The involvement of central 5-HT1B and 5-HT2C receptors in the regulation of appetite was initially suggested by the anorectic effects of a central serotonergic agonist (m-chlorophenylpiperazine [mCPP]) [20]. A major contribution of 5-HT2C receptor to feeding control was confirmed by a study using Htr2c KO mice [21]. Htr2c KO mice are hyperphagic and obese, and the anorectic effects of mCPP do not occur in Htr2c KO mice [22]. Furthermore, studying the effects of 5-HT on the hypothalamic feeding circuit revealed that 5-HT activates POMC neurons through the 5-HT2C receptor and inhibits NPY/AgRP neurons through the 5-HT1B receptor [23]. Thus, central 5-HT inhibits food intake by modulating the hypothalamic feeding circuits. Many drugs targeting 5-HT2C receptor have been produced and some are currently being used in the clinical setting. However, others were discontinued because of severe complications such as pulmonary hypertension and valvular heart disease (Table 2) [2425]. Recently, a selective 5-HT2C receptor agonist (Belviq, lorcaserin hydrochloride; Arena, Zofingen, Switzerland) was officially approved for use in the treatment of obesity [26].
Given that TPH2 is exclusively expressed in the brain, Tph2 KO mice were expected to be hyperphagic and obese. However, Tph2 KO mice do not show an obese phenotype although their central 5-HT level is decreased [2728]. In addition, Htr1b KO mice do not become obese although they are hyperphagic [29]. There have also been reports supporting that central 5-HT increases the sympathetic tone and activates brown adipose tissue (BAT) [3031]. Taken together with the known adverse effects of anti-obesity drugs with regard to their increasing the 5-HT activity in the brain, these controversial data indicate that increasing the 5-HT activity in the brain may not be a good strategy for anti-obesity treatment.
ROLES OF 5-HT IN PERIPHERAL TISSUES
Although TPH1 is widely expressed in the peripheral tissues, over 90% of the total body 5-HT is produced in the gut [32]. The majority of the peripheral 5-HT is stored in platelets and controls the hemodynamics upon the activation of platelets. 5-HT is also present in other peripheral tissues and acts autonomously on many cells in the cardiovascular, gastrointestinal, hematopoietic, and immune systems, as well as in the bone, liver, and placenta [33].
Gut
EC cells are dispersed throughout the intestinal mucosa and are the main source of 5-HT in the gut. The major targets of the gut-derived 5-HT (GDS) are the mucosal projections of primary afferent neurons. These include extrinsic nerves and afferent neurons. The extrinsic nerves transmit sensations of nausea and discomfort to the CNS. There are two types of intrinsic primary afferent neurons (IPANs). The submucosal IPAN regulate the peristaltic and secretory reflexes, whereas myenteric IPAN regulate giant migrating contractions. The 5-HT secreted from myenteric neurons mediates fast and slow excitatory neurotransmission and regulates gastrointestinal motility.
Many 5-HT receptor agonists/antagonists are already used to regulate gut motility in the clinical setting (Table 3). Pressure, acid, and/or cancer therapeutic agents are known to release 5-HT from EC cells, which provokes abdominal discomfort and nausea [3435]. 5-HT3 receptor antagonists, such as ondansetron (Zofran; GSK, London, UK), granisetron (Kytril; Roche Laboratories, Basel, Switzerland), or alosetron (Lotronex; GSK, London, UK), alleviate the 5-HT-induced discomfort that extrinsic sensory nerves convey to the CNS. Irritable bowel syndrome (IBS) is a disease linked to the serotonergic system. The persistent visceral hypersensitivity of the bowel in IBS patients can be blunted by the administration of a 5-HT4 receptor agonist (Tegaserod; Novartis, Basel, Switzerland) [36]. Thus, drugs that inhibit the 5-HT3 receptor or enhance the 5-HT4 receptor have been used to treat IBS.
Many studies suggest that 5-HT plays a role in the pathogenesis of gastrointestinal diseases by affecting the production of pro-inflammatory mediators and the immune system [37]. 5-HT receptors have been identified on various immune cells such as lymphocytes, monocytes, macrophages, and dendritic cells [3839]. The T lymphocyte-derived 5-HT may act as an autocrine factor and has demonstrated a fundamental role as an intrinsic cofactor in T-cell activation and function through the 5-HT7 receptor [40].
Liver
5-HT is known to regulate liver regeneration. Partial hepatectomy has been widely used as an experimental model to understand the role of 5-HT in liver regeneration. The number of platelets (filled with 5-HT) strongly correlates with the hepatocyte proliferative capacity [41]. In contrast, several studies using SERT-deficient rats reported that the regenerative process is not influenced when the platelet 5-HT level is reduced [42]. Thus, the mechanism underlying liver regeneration is more complex than has previously been thought, and more studies are needed to elucidate the main source of 5-HT to regulate liver regeneration. To directly examine the function of 5-HT in liver regeneration, partial hepatectomy was performed on Tph1 KO and wild type (WT) control mice. All markers of hepatocyte proliferation were reduced in Tph1 KO mice, suggesting that 5-HT is involved in the induction of hepatocyte proliferation after a major loss of hepatic tissue [43]. It has also been demonstrated that 5-HT can promote liver regeneration after liver injury through the 5-HT2 receptor or 5-HT7 receptor [4445].
Crane et al. [46] reported a role of peripheral 5-HT in the liver after exposure to a high fat diet (HFD). When fed a HFD, WT mice become obese and develop fatty liver disease. In contrast, Tph1 KO mice do not develop a fatty liver after exposure to a HFD and have reduced fat accumulation in the liver. 5-HT increases hepatic glucose uptake and fat content in liver [4748]. The duodenal 5-HT content increases in ob/ob mice and treatment with a 5-HT3 receptor antagonist caused the reduction of the elevated 5-HT levels and an increase in SERT in the duodenum. In these mice, the 5-HT3 receptor antagonist also reduced the fat content, inflammation, and necrosis of the liver [48].
With regard to insulin resistance, Sumara et al. [8] reported that inhibition of GDS improves the glucose tolerance in mice fed a HFD. In hepatocytes, GDS signaling through the 5-HT2B receptor promotes gluconeogenesis. In addition, GDS prevents glucose uptake into hepatocytes in a glucose transporter 2-dependent manner. Therefore, gut-specific Tph1 KO mice and liver-specific Htr2b KO mice showed improved glucose tolerance compared to WT mice [8].
Pancreatic β-cells and the impact of 5-HT on diabetes mellitus
It has long been known that 5-HT is present in the same vesicle with insulin in pancreatic β-cell after preloading of 5-hydroxytryptophan, a precursor of 5-HT, and 5-HT has been associated with the regulation of the blood glucose concentration [4950]. Recently, 5-HT has been identified as a downstream molecule of placental lactogen that mediates the adaptation of β-cells to pregnancy [10]. In response to the lactogens produced during pregnancy, the expression of Tph1 rises sharply in pancreatic β-cells, resulting in massive production of 5-HT in β-cells [10]. Islet 5-HT acts in an autocrine/paracrine manner through the 5-HT3 receptor to increase the glucose responsiveness of β-cells and thereby increases the overall islet glucose-stimulated insulin secretion (GSIS) [5152]. In addition, the expression of the 5-HT2B receptor increases during mid-gestation to stimulate β-cell proliferation and increase the β-cell mass, whereas 5-HT1D receptor expression increases at the end of gestation to reduce the β-cell mass [10].
5-HT also regulates insulin secretion in a diet-induced insulin-resistant state. The β-cell-specific Tph1 KO mice and Htr3a KO mice developed glucose intolerance compared to WT mice after exposure to an HFD. The GSIS was impaired in islets isolated from HFD-fed Htr3a KO and β-cell-specific Tph1 KO mice, and 5-HT treatment improved the insulin secretion from the islets of β-cell-specific Tph1 KO mice, but not from the Htr3a KO islets [51]. In addition, 5-HT production has been observed in the embryonic pancreas. In particular, TPH1 expression is upregulated in newborn β-cells during the perinatal period while β-cells are actively proliferating [53]. Given that β-cells produce 5-HT under two physiological conditions of β-cell proliferation, the perinatal period and pregnancy, 5-HT is thought to be an important regulator of β-cell proliferation as well as insulin secretion [10515254].
Adipose tissues
The biological functions of peripheral 5-HT in the regulation of energy homeostasis have been extensively studied using chemical agonists and antagonists since the 1960s. By taking advantage of mouse genetic studies, it has become possible to better understand the precise roles of 5-HT in energy metabolism. SERT KO mice exhibit an obese phenotype although they are expected to be slim due to the anorexigenic effects of central 5-HT [55]. Body weight is reduced in Tph1 and Tph2 double KO mice, as well as Tph1 KO mice [2728]. However, the body weights of gut-specific Tph1 KO mice are comparable to those of WT control mice [8]. These data suggest that 5-HT, other than GDS, may play a role in regulating systemic energy homeostasis. Recently, two independent studies have highlighted the role of adipocyte-derived 5-HT in energy storage in the white adipose tissue (WAT) and energy expenditure in the BAT [1146]. In a diet-induced mouse model of obesity, the Tph1 expression and tissue 5-HT concentrations were elevated in adipose tissues [1146]. Tph1 KO mice were protected from obesity and the related metabolic dysfunctions [46]. Tph1 KO mice gained significantly less weight and had lower adiposity when fed an HFD. The glycemic control is also improved in Tph1 KO mice, although the glucose uptake was similar in the muscle, liver and heart, indicating that the BAT make a major contribution to the increase in the basal metabolic rate [46]. Indeed, energy expenditure was enhanced in Tph1 KO mice when they were fed an HFD. The BAT activity increased in a Ucp1-dependent manner in Tph1 KO mice. The obesogenic actions of peripheral 5-HT were also confirmed using a peripheral TPH inhibitor (LP-533401) [1146]. Furthermore, a cell autonomous effect of 5-HT in adipose tissue has been shown in adipocyte-specific Tph1 KO mice [11]. The adipocyte-specific Tph1 KO induced Ucp1 and Dio2 expression in the BAT and subcutaneous WAT [11].
Regarding brown fat thermogenesis, the 5-HT3 receptor plays a major role in diet-induced thermogenesis [11]. Diet-induced thermogenesis was robustly increased in the BAT of Htr3a KO mice fed a HFD [11]. In addition to the role of 5-HT in the BAT, in vitro experiments using 3T3-L1 adipocytes provided a hint about the role of 5-HT in adipogenesis [1156]. It was known that 5-HT can increase the lipid accumulation in human and mouse fat cells [57]. It was also known that 5-HT2A receptor expression is increased in the hypertrophied 3T3L1 adipocytes and WAT of db/db mice, and the activation of the 5-HT2A receptor reduces adiponectin expression in hypertrophied 3T3L1 adipocytes [58]. Treatment with a 5-HT2A receptor antagonist increased the lipid accumulation and circulating adiponectin levels [1159]. An antagonist for the 5-HT2A receptor inhibited adipogenesis [56]. Thus, 5-HT increases energy storage in the WAT through 5-HT2A receptor and inhibits energy expenditure in the BAT through 5-HT3 receptor. Taken together, these recent findings suggest that 5-HT negatively regulates the sensitivity of BAT to β-adrenergic stimulation.
GENETICS OF 5-HT AND OBESITY
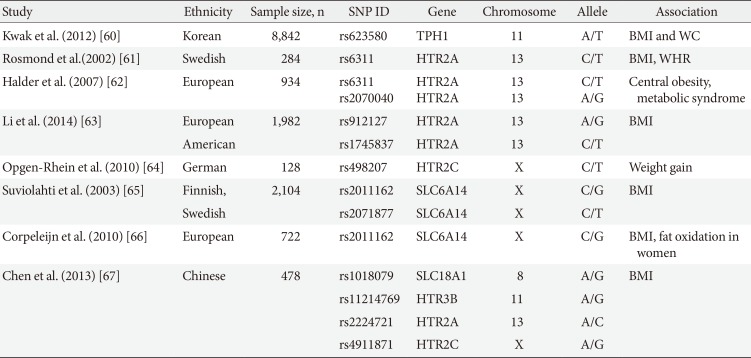
To understand the genetic basis of obesity, many genome-wide association studies (GWAS) have been conducted. Several GWAS revealed genetic associations between the serotonergic system and obesity. The first association between TPH1 and obesity was identified through a GWAS in Koreans [60]. A genetic variant of TPH1 (rs623580) was associated with the body mass index in a GWAS comprising 8,842 subjects (P=0.0047), and single nucleotide polymorphisms (SNPs) of TPH1 were significantly associated with weight gain during pregnancy in women with gestational diabetes mellitus. Several GWAS revealed associations between SNPs of the 5-HT receptors and obesity (Table 4) [6061626364656667]. SNPs of HTR2A had strong associations with obesity in both European and Asian ethnic groups [61626367]. SNPs in the promoter (rs6311, -1438 [G/A]) and intron (rs2070040, 2416 [C/T]) were associated with metabolic syndrome in a study of 934 European subjects. Central obesity (a high waist circumference) showed a significant association with the -1438 GG genotype (odds ratio [OR], 1.57; P=0.003), and the -1438 GG and 2416 TT genotypes were associated with an increased risk of metabolic syndrome (OR, 1.71; P=0.016) [62]. In 478 healthy Chinese subjects, 12 SNPs in seven genes (MAOB, SLC18A1, HTR2A, HTR2C, HTR3B, HTR4, and HTR7) were significantly associated with the body mass index [67]. These genetic studies indicate that the identification of serotonergic system defects can be used for risk prediction and to develop individualized treatments for obesity and metabolic syndrome.
CONCLUSIONS
The human body has two separate serotonergic systems: the central 5-HT and peripheral 5-HT systems (Fig. 1). With regard to the central 5-HT system, increasing the 5-HT signaling has been utilized therapeutically to reduce body weight by reducing appetite. In 2010, the U.S. Food and Drug Administration approved a new selective 5-HT2C receptor agonist (Belviq, lorcaserin hydrochloride) for use in the treatment of obesity [26]. This drug was also approved by the Ministry of Food and Drug Substances in Korea. In peripheral tissues, suppressing 5-HT signaling might represent a new target for anti-obesity treatment by increasing energy expenditure and improving insulin resistance [1146]. Systemic TPH1 inhibitors and a peripheral TPH1 inhibitor (LP-533401) have already been patented for treating diabetes and obesity [6869]. However, there have not yet been any clinical trials using these drugs to treat obesity. Recent GWAS results support the clinical importance of serotonergic regulation in human metabolism, but most of these studies have small sample sizes and have not been validated. Therefore, to enhance our understanding of the role of 5-HT in metabolism and to provide effective therapeutic applications of 5-HT, more clinical studies are needed on drugs that can regulate the serotonergic system, and genetic studies are needed to investigate the associations between 5-HT and metabolic diseases.
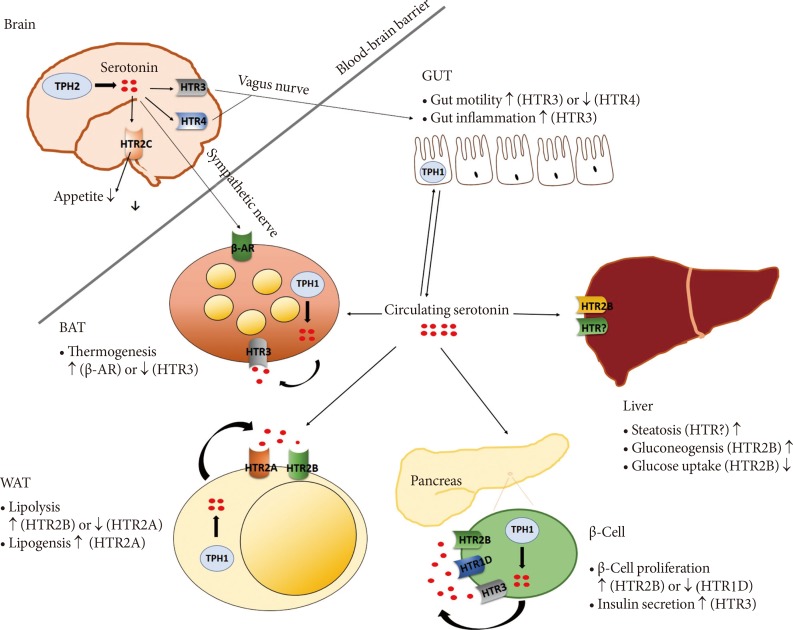
Roles of 5-hydroxytryptamine (5-HT) on metabolic organs. Central 5-HT regulates appetite and controls metabolic organs through sympathetic/parasympathetic nervous system. Peripheral 5-HT regulates glucose metabolism, insulin resistance and energy expenditure in an autocrine/paracrine or endocrine manner. TPH, tryptophan hydroxylase; HTR, 5-HT receptor; β-AR β-adrenergic receptor; BAT, brown adipose tissue; WAT, white adipose tissue.
ACKNOWLEDGMENTS
This work was supported by a grant from the National Research Foundation (NRF) funded by the Ministry of Science, ICT & Future Planning (NRF-2014M3A9D8034464, NRF-2014M3A9D5A0173546 to H.K.).
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.