Prevalence of Type 2 Diabetes Mellitus among Korean Children, Adolescents, and Adults Younger than 30 Years: Changes from 2002 to 2016
Article information
Abstract
Background
Despite the importance of and social concern regarding prevention of diabetes at younger ages, limited data are available. This study sought to analyze changes in the prevalence of type 2 diabetes mellitus (T2DM) in Koreans younger than 30 years according to sex, age, and level of income.
Methods
The dataset analyzed in this study was derived from health insurance claims recorded in the National Health Insurance Service (NHIS) database. Participants’ level of income was categorized as low (quintile 1, <20% of insurance premium) or others (quintile 2–5).
Results
In males and females, the prevalence of T2DM per 10,000 people steadily increased from 2.57 in 2002 to 11.41 in 2016, and from 1.96 in 2002 to 8.63 in 2016. The prevalence of T2DM in girls was higher in the age group of 5 to 14 years. Even though the prevalence was higher among those older than 20 years, the increase had started earlier, in the early 2000s, in younger age group. Adolescents aged 10 to 19 years in low-income families showed a remarkable increase in prevalence of T2DM, especially in boys.
Conclusion
The prevalence of T2DM in young Koreans increased more than 4.4-fold from 2002 to 2016, and the increase started in the early 2000s in younger age groups and in low-income families. This is the first study to examine the trend in prevalence of T2DM in children, adolescents, and young adults in Korea. Future studies and collaborations with social support systems to prevent T2DM at an early age group should be performed.
INTRODUCTION
Type 2 diabetes mellitus (T2DM) in young patients is on the rise given the increasing burden of obesity worldwide. In the United States, T2DM has been described as a new epidemic affecting the pediatric population [1]. The prevalence of T2DM in Korean adults aged ≥30 years increased 0.2% to 0.5% annually [2]. Similar to older-onset T2DM, the major predisposing risk factors for T2DM in children are obesity, family history, and a sedentary lifestyle [3]. Earlier onset of T2DM is associated with long-term disease exposure and an increased risk for chronic complications. Younger age at T2DM onset leads to more rapid deterioration of β-cell function [4] and was linked to more severe disease and an accelerated development of complications [5], as recently exemplified by the finding of an increased risk for cardiovascular mortality in this population [6,7]. A diagnosis of T2DM made in adolescence is predicted to shorten an individual’s life expectancy by 15 years relative to that of peers without T2DM [8]. Furthermore, T2DM has a disrupting effect on young people during their most productive and active years [9]. As such, in addition to its health impacts, the economic and social impacts of early-onset T2DM are concerning.
Diabetes affects 422 million people worldwide, and the global prevalence has been growing, with a disproportionate rise in numbers of younger patients [3]. Despite the importance of and social concern concerning the prevention of diabetes at younger ages and there were data of the prevalence of T2DM in Korean adults [2], limited data and reports exist in Koreans younger than 30 years. This study therefore sought to analyze changes in the prevalence of T2DM in Koreans younger than 30 years according to sex, age, and level of income based on health insurance premiums using national data.
METHODS
Data source
A compulsory National Health Insurance Service (NHIS) program was launched by the Korean government in February 1999. As of 2020, the NHIS program covers the entire population of South Korea. The dataset analyzed in this study was derived from health insurance claims recorded in the NHIS claims database, from which the NHIS provided data after deidentification. The claims case data contained patients’ age, sex, and level of income. The study protocol was reviewed and approved by the Konkuk University Medical Center Institutional Review Board (approval no. KUH1090064) and permission for the use of NHIS data was granted by the NHIS for the research (NHIS-2019-1-069). The need for written informed consent from the participants was waived due to the retrospective nature of this study.
Study population
Using NHIS claims data gathered from January 1, 2002 to December 31, 2016, we extracted information on patients (age <30 years) with T2DM for each year. In this study, patient with T2DM were defined as those with at least one claim with a principal or first additional diagnosis of diabetes, based on International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes for diabetes (E1000–E1090, type 1 diabetes mellitus [T1DM]; E1100–E1190, T2DM; E1200–E1290, malnutrition-related diabetes; E1300–E1390, diabetes associated with other diseases, other type of diabetes; and E1400–E1490, diabetes type unknown, unknown diabetes) and the concurrent prescription of anti-diabetic medications. We excluded patients with codes for T1DM (E1000–E1090) and those treated with only insulin for more than 1 year. Because it is a prevalence study, there are overlapping numbers between years. However, just because a patient received multiple claims in the same year does not count as a duplicate number. The data were extracted on August 30, 2018. For more detailed subanalyses, patients were stratified by sex and age group (0–4, 5–9, 10–14, 15–19, 20–24, and 25–29 years). Participants’ level of income was estimated using the health insurance premium and divided into five quintiles based on the National Health Insurance fee imposed on each household, categorized arbitrary as low group (quintile 1, <20% of insurance premium) or others (quintile 2–5, except for the low group).
Statistical analysis
All data were analyzed using the SAS version 9.3 software program (SAS Institute Inc., Cary, NC, USA). We conducted the analysis in accordance with sex and age groups (0–4, 5–9, 10–14, 15–19, 20–24, and 25–29 years). The prevalence rates of T2DM for each year from 2002 to 2016 were calculated by dividing the number of patients with T2DM by the total number of health insurance recipients. We used the Cochran-Armitage trend test to estimate the P value for the time trends. A P value of less than 0.05 was considered to be statistically significant.
RESULTS
Table 1 shows the prevalence of T2DM in 2002 and 2016, respectively. Between 2002 and 2016, the prevalence of T2DM in Korean children, adolescents, and young adults younger than 30 years of age was 2.27 and 10.08 per 10,000 people, respectively, exhibiting a significant increase by 4.43 times. The prevalence increased exponentially with age, being three-fold greater in the 15 to 19 years age group than in the 10 to 14 years age group in both 2002 and 2016.
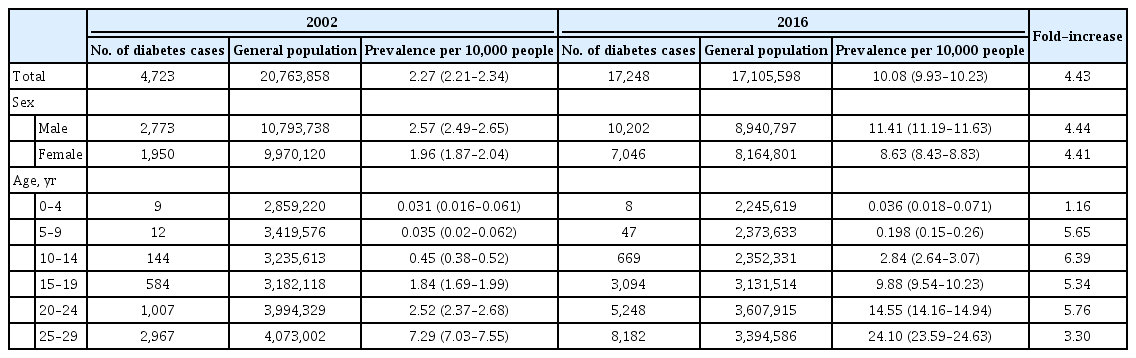
The prevalence of T2DM among Korean children, adolescents, and young adults younger than 30 years of age in 2002 and 2016
The prevalence rates of T2DM from 2002 to 2016 are presented in Table 2 and depicted graphically in Fig. 1. A gradual and constant increase in the prevalence of T2DM was observed, affecting both sexes similarly: in male and female individuals, respectively, the prevalence of T2DM steadily increased from 2.57 in 2002 to 11.41 in 2016 (P for trend <0.0001) and from 1.96 in 2002 to 8.63 in 2016 (P for trend <0.0001), respectively. Fig. 2 shows the prevalence of T2DM by sex and age group. In the age group of 0 to 4 years, no significant change was observed; however, the remaining four age groups experienced a remarkable increase the prevalence of T2DM from 2002 to 2016. Cases of T2DM displayed a female predominance from the ages of 5 to 14 years and a male predominance in those older than 20 years; male patients overtook female ones in terms of T2DM prevalence somewhere between 15 and 19 years of age.

The prevalence of T2DM among Korean children, adolescents, and young adults younger than 30 years of age from 2002 to 2016
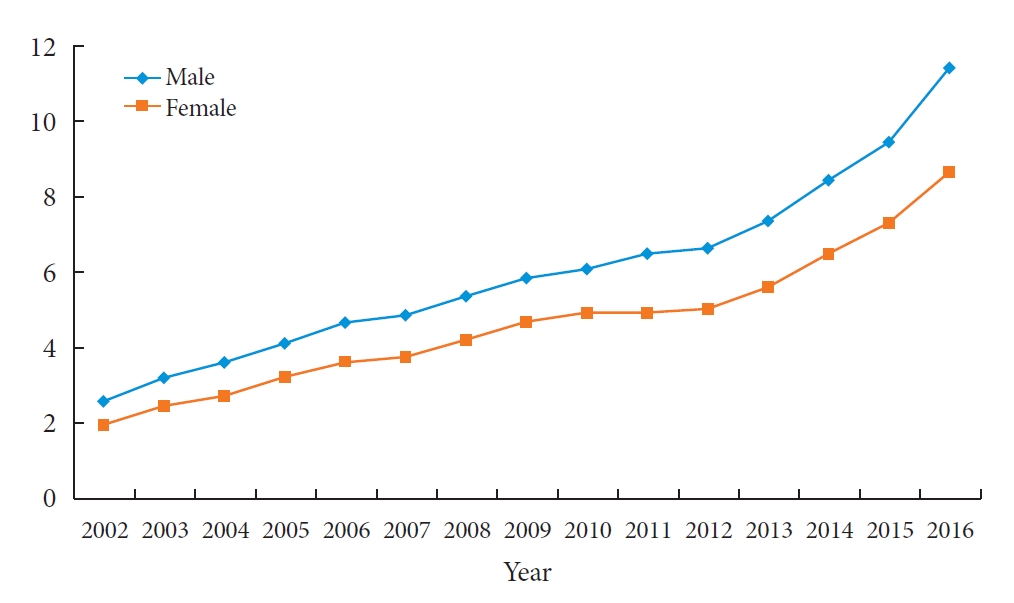
The prevalence of type 2 diabetes mellitus per 10,000 people among Korean children, adolescents, and young adults younger than 30 years of age from 2002 to 2016. A difference in the slope between male and female is confirmed with the test for trend, P (P=0.015).
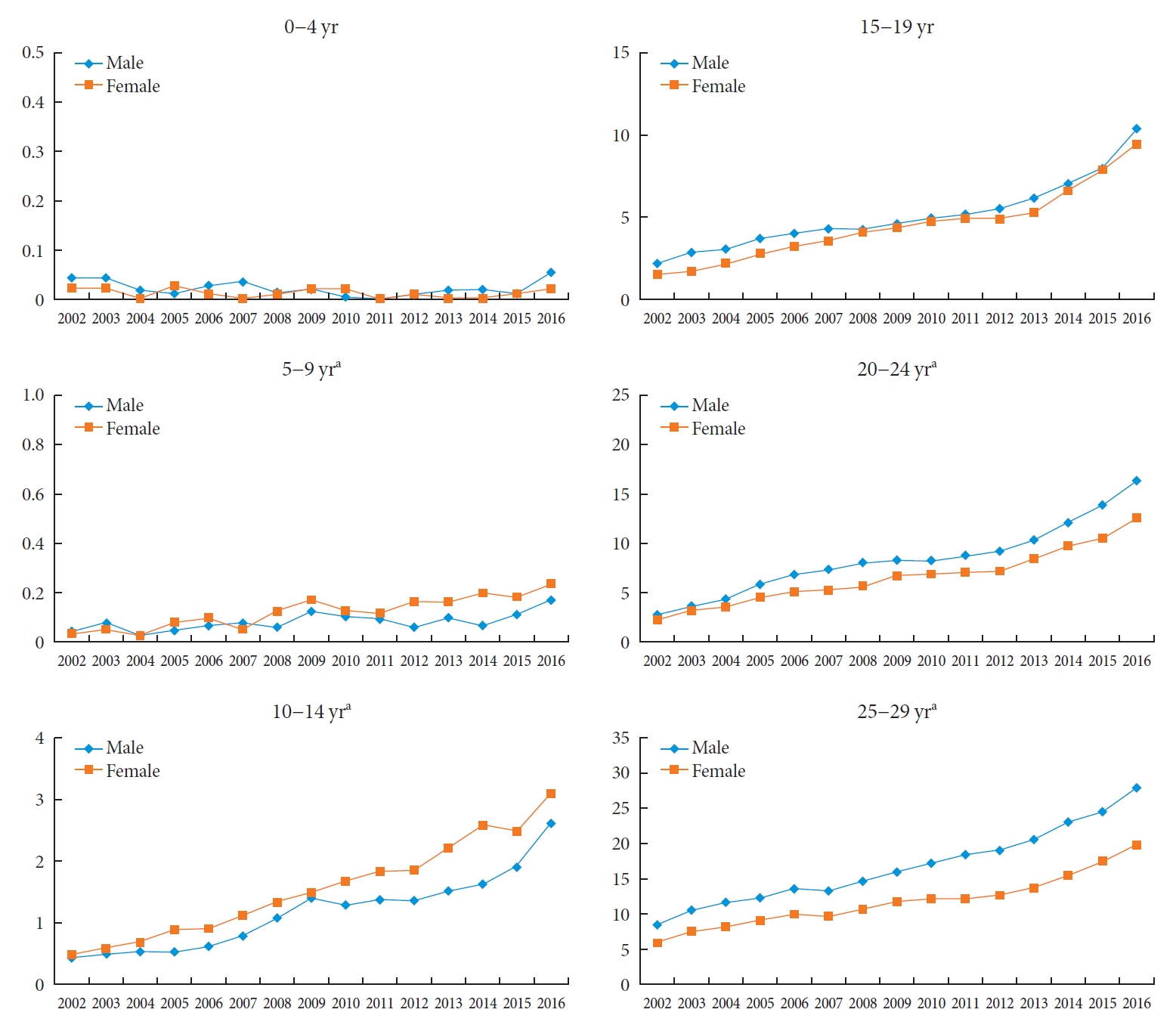
The prevalence of type 2 diabetes mellitus per 10,000 people stratified by sex and age among Korean children, adolescents, and young adults younger than 30 years of age from 2002 to 2016. Differences in the slope between groups are confirmed with the test for trend, P. aP for trend <0.05.
Also, the prevalence of T2DM increased rapidly among both boys and girls aged older than 10 years with low income (Table 3, Fig. 3, Supplementary Table 1). Notably, from 2006, the prevalence of T2DM has increased significantly in the male, low-income, and 10 to 14 years age group, while, in 2013, the increase was predominantly observed in the 20 years and older age group.

The prevalence of T2DM stratified by sex, age, and level of income among Korean children, adolescents, and young adults younger than 30 years of age from 2002 to 2016

The prevalence of type 2 diabetes mellitus per 10,000 people stratified by sex, age, and level of income among Korean children, adolescents, and young adults younger than 30 years of age from 2002 to 2016. Differences in the slope between groups are confirmed with the test for trend, P. Mo, male other (quintile 2–5); ML, male low income (quintile 1); Fo, female other (quintile 2–5); FL, female low income (quintile 1). aP for trend <0.05.
DISCUSSION
This nationwide study using nationwide Korean claims data investigated trends in the prevalence of T2DM in individuals younger than 30 years of age. Despite concerns about increasing rates of obesity and T2DM, limited data exist regarding trends in the prevalence of T2DM in Koreans, including among children, adolescents, and young adults, especially stratified according to income level. To our knowledge, we present the first estimates of recent changes in T2DM prevalence rates among Koreans younger than 30 years of age using NHIS data according to age, sex, and level of income.
The growing worldwide prevalence of T2DM in young people [10] has emphasized the need for identification and treatment of this condition in children and adolescents. T2DM has accounted for a significant number of adolescents with diabetes recently. Available global data indicate considerable variations in incidence and prevalence, depending on ethnicity and geographical region. In the SEARCH for Diabetes in Youth study [11], the overall prevalence of T2DM among youth between 2001 and 2009 increased by 30.5%, rising with age. Relatively few population-based datasets are available concerning the prevalence of T2DM in Asia. In Japan, 80% of all new cases of diabetes in children and adolescents are diagnosed as T2DM [12]. Likewise, in Taiwan, 54.2% of new cases are diagnosed as T2DM, with an incidence of 6.5 per 100,000 people [12]. In China, in 2002 and 2012, the prevalence rates of diabetes were 0.24% and 0.52% among children and adolescents aged seven to 17 years, respectively [13]. Other Chinese data suggest the prevalence of T2DM among those younger than 18 years has increased from 4.1 in 1995 to 7.1 and 10.0 in 2010, respectively [14]. A gradual and constant increase in the prevalence rate of T2DM in Koreans younger than 30 years of age was observed from 2002 to 2016 in our study.
In this study, we found that T2DM occurs increasingly frequently as children progress through puberty. Puberty is associated with significant changes in physiology, including a transient reduction in insulin sensitivity [15]. As compensation, insulin secretion must increase reciprocally, which may lead to hyperglycemia in youth with limited β-cell capacity due to genetic, epigenetic, and/or lifestyle factors [5]. Thus, puberty creates a high-risk time for diabetes development in susceptible individuals.
Youth-onset T2DM has a sex ratio (male:female) that varies from 1:4 to 1:6 in native North Americans and is 1:1 in Asians and Libyan Arabs [16]. In the SEARCH study of American children and adolescents, the incidence of T2DM in girls was nearly twice that in boys [17]. In China, the prevalence of T2DM in boys is higher than in girls [14]. In this Korean study, the overall prevalence of T2DM showed a male predominance and the prevalence of diabetes in boys increased after the age of 15 years. It is presumed that this trend is probably due to the male predominance among children and adolescents in Korea with extreme obesity [18].
A substantial proportion of young people with T2DM live in poverty or socially disadvantaged households [19]. The SEARCH for Diabetes in Youth study reported a 44% prevalence rate of poverty [20], and while the Treatment Options for Type 2 Diabetes in Adolescents and Youth (TODAY) cohort demonstrated a 41% prevalence rate of poverty in adolescents and youth with T2DM [21]. According to the T1DM cohort of the Pediatric Diabetes Consortium [22], the family income and parental education were relatively high, but, in the T2DM cohort, 43% of participants lived in a household with an income of less than 25,000 United States Dollars per year [23]. Diabetes care providers need to be aware of these characteristics and the increased difficulties faced by youth with T2DM and should consider how this might affect patient management. A previous Korean nationwide study indicated that the incidence rates of T2DM and diabetes-related morbidity were higher among diabetic subjects who were over 30 years of age with a lower socioeconomic status [24]. In the United States and Europe, youth-onset T2DM has been predominantly found in populations characterized by a low socioeconomic and educational status, whereas, in emerging countries like China and India, more affluent children are more likely to develop T2DM [16]. In our study, there were varying prevalence rates of T2DM observed according to age, sex, and income level. Generally, the low-income group had a higher prevalence rate than did the high-income group, in correlation with findings from the United States and Europe. Boys in low-income families aged 10 to 19 years showed a remarkable increase in the prevalence of T2DM. Even though the prevalence was highest among those older than 20 years of age, the rise in prevalence rate had started already from 2006 in boys in low-income families aged 10 to 14 years, suggesting the trend initiated earlier than that seen in the age group of 20 to 29 years.
Although the overall prevalence of T2DM in American youth is low (0.046% in 2009 per the SEARCH for Diabetes in Youth study), it has increased by 30% from 2001 [11]. We can predict the same phenomenon is likely in the Korean youth. The prevalence of T2DM in Korean adults aged ≥30 years increased 0.2% to 0.5% annually, from 5.6% in 2006 to 8% in 2013 and 13.8% of Korean adults aged ≥30 years had diabetes in 2018 [2,25]. In this study, compared to 2002 and 2016, the fold-increase was 4.43 in Koreans younger than 30 years. Although the number of T2DM patients in Koreans younger than 30 years is smaller than adults aged ≥30 years, the trend seems to outweigh that of adults aged ≥30 years.
The prevalence of obesity among children and adolescents in Korea increased nine times in males and four times in females from 1979 to 2002 [26,27]. Also after the 2000’s, the prevalence of obesity increased from 8.7% in 2007 to 15.0% in 2017 in the National School Health Examination in Korean children aged 6 to 18 years [28]. The prevalence of adult obesity increased across all age groups, especially in the 20s, 30s [29]. From 2009 to 2018, the prevalence of obesity in males aged 20 to 29 years was 18.5% in 2009 to 27.5%. In females aged 20 to 29 years, the prevalence of obesity increased from 8.6% in 2009 to 15.4% in 2018 [29]. This increase in obesity may have affected to the rapid increase in the prevalence of T2DM in Koreans younger than 30 years.
In future research, increasing the number of patients is important, but it is not a good indicator for the early detection of the changes in disease patterns. Even results from a small study population should be paid attention to if the increase in prevalence or incidence of a disease is large. Furthermore, risk-based screening for T2DM and/or prediabetes should be considered in youth at the onset of puberty or ≥10 years of age in overweight and obese youth [15,30,31].
This study has some limitations, including the fact that we could not assess trends before 2002. Our observation period was relatively short and further surveillance will produce better estimates of changes. Another limitation was there were not the adjustment of the individual obesity. Finally, since the NHIS data is billing data, diagnosis information could only be confirmed by disease code. In order to reduce errors, only cases which the diagnosis and drug prescription were simultaneously counted as the patient group. Nevertheless, the strengths of this study were that it included a large national dataset and the fact that it is the first study to date to example the trend in the prevalence of T2DM in children, adolescents, and young adults in Korea.
In conclusion, it should be noted that the prevalence of T2DM in low-income children and adolescents has increased significantly. Given the growing and high prevalence rate of T2DM in Korean younger than 30 years of age, obese children and adolescents at risk of diabetes should educate themselves on the condition and actively undergo screening tests. Research on T2DM in children and adolescents is scarce and conditions for testing are insufficient. It is important to monitor T2DM in children, including via continuous trend analysis. Future studies and collaborations with social support systems to prevent T2DM at an early age group should be performed.
Supplementary Materials
Supplementary materials related to this article can be found online at https://doi.org/10.4093/dmj.2021.0038.
The prevalence of T2DM stratified by sex, age, and level of income among Korean children, adolescents, and young adults younger than 30 years of age from 2002 to 2016
Notes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conception or design: Y.H.H., S.C.
Acquisition, analysis, or interpretation of data: Y.H.H., I.H.C., K.H., S.C.
Drafting the work or revising: Y.H.H., S.C.
Final approval of the manuscript: Y.H.H., I.H.C., K.H., S.C.
FUNDING
This work was partially supported by the Korean society for the study of obesity, which had not role in study design, data collection, data analysis, data interpretation, or writing of the report. This work was supported by the Soonchunhyang University Research Fund.
Acknowledgements
We thank the Korean National Health Insurance Corporation and all the participants of the study.

