Increasing Age Associated with Higher Dipeptidyl Peptidase-4 Inhibition Rate Is a Predictive Factor for Efficacy of Dipeptidyl Peptidase-4 Inhibitors
Article information
Abstract
Background
It is not known which type 2 diabetes mellitus (T2DM) patients would most benefit from dipeptidyl peptidase-4 (DPP-4) inhibitor treatment. We aimed to investigate the predictors of response to DPP-4 inhibitors considering degree of DPP-4 inhibition.
Methods
This study is a post hoc analysis of a 24-week, randomized, double-blind, phase III trial that compared the efficacy and safety of a DPP-4 inhibitor (gemigliptin vs. sitagliptin) in patients with T2DM. Subjects were classified into tertiles of T1 <65.26%, T2=65.26%–76.35%, and T3 ≥76.35% by DPP-4 inhibition. We analyzed the change from baseline in glycosylated hemoglobin (HbA1c) according to DPP-4 inhibition with multiple linear regression adjusting for age, ethnicity, body mass index, baseline HbA1c, and DPP-4 activity at baseline.
Results
The mean age was greater in the high tertile group compared with the low tertile group (T1: 49.8±8.3 vs. T2: 53.1±10.5 vs. T3: 55.3±9.5, P<0.001) of DPP-4 inhibition. Although HbA1c at baseline was not different among tertiles of DPP-4 inhibition (P=0.398), HbA1c after 24-week treatment was lower in the higher tertile compares to the lower tertile (T1: 7.30%±0.88% vs. T2: 7.12%±0.78% vs. T3: 7.00%±0.78%, P=0.021). In multiple regression analysis, DPP-4 enzyme inhibition rate was not a significant determent for HbA1c reduction due to age. In subgroup analysis by tertile of DPP-4 inhibition, age was the only significant predictor and only in the highest tertile (R2=0.281, B=–0.014, P=0.024).
Conclusion
This study showed that HbA1c reduction by DPP-4 inhibitor was associated with increasing age, and this association was linked with higher DPP-4 inhibition.
Highlights
• The predictors of clinical response to DPP-4 inhibitors were age, ethnicity, and HbA1c at baseline.
• Older patients with DPP-4 inhibitor associated with higher degree of DPP-4 inhibition.
• The association between age and HbA1c reduction was dependent on higher degree of DPP-4 inhibition.
INTRODUCTION
Dipeptidyl peptidase-4 (DPP-4) inhibitors have been widely used as a therapeutic option for patient with type 2 diabetes mellitus (T2DM) [1]. They improve glycemic control in patients with T2DM by increasing circulating levels of incretins, endogenous gut-derived peptide hormones that enhance insulin secretion and suppress glucagon release in a dose-dependent manner [2]. Despite the widespread use of DPP-4 inhibitors; however, it has not yet been established which patients would benefit most from the treatment. Although previous studies have suggested predictors of better clinical response to DPP-4 inhibitors [3-6], the results were somewhat inconsistent.
It has been demonstrated that currently available DPP-4 inhibitors might differ in potency of DPP-4 inhibition [7]. For example, once-daily treatment with sitagliptin provided significantly greater DPP-4 inhibition than saxagliptin or vildagliptin administered once daily and was similar to that of vildagliptin administered twice daily [8]. Furthermore, a meta-analysis of alogliptin, saxagliptin, sitagliptin, and vildagliptin efficacy results demonstrated that weighted average inhibition of DPP-4, a summary of the time course of DPP-4 inhibition over the dosing interval, was a useful biomarker related to glycosylated hemoglobin (HbA1c) response after chronic therapy with DPP-4 inhibitor [9]. However, few studies have suggested the predictors of better clinical response to DPP-4 inhibitors based on degree of DPP-4 inhibition.
Gemigliptin (LC15-0444) is a potent, highly selective, and long acting DPP-4 inhibitor. Various studies have demonstrated the efficacy and safety of gemigliptin for treatment of T2DM [10,11]. In addition, gemigliptin and sitagliptin were more effective than glimepiride in reducing glycemic variability as measured by continuous glucose monitoring as an initial combination with metformin in patients with T2DM [12]. In this study, we aimed to investigate the predictors of better clinical response to DPP-4 inhibitors according to degree of DPP-4 inhibition.
METHODS
Study population
This study is a post-hoc analysis of a multinational, randomized, active-controlled, parallel group, double-blind, phase 3 clinical trial in which the efficacy and safety of a DPP-4 inhibitor, gemigliptin versus sitagliptin added to metformin, were investigated in patients with T2DM (https://clinicaltrials.gov/ct2/show/NCT01602003). The detailed protocol of the study has been previously described [13]. Briefly, after screening (n=604), patients with T2DM (18 to 75 years of age) who were being treated with metformin monotherapy for at least 12 weeks with at least 4 weeks of 1,000 mg/day or higher dose of metformin before screening were randomized to one of the three treatment groups (sitagliptin 100 mg daily [n=142], gemigliptin 25 mg twice a day [n=141], or gemigliptin 50 mg daily [n=142]) at a 1:1:1 ratio. Each group was treated with the assigned treatment regimen for 24 weeks. We analyzed 323 patients who completed the treatment regimen and underwent measurement of DPP-4 activity at baseline and after 24 weeks. Approval was obtained from the Institutional Review Board of Kangbuk Samsung Hospital for the study protocol (KBSMC 2021-02-036). Informed consent was waived by the board.
Endpoint assessment
The primary endpoint of this post-hoc analysis was the change in HbA1c from baseline according to the degree of DPP-4 inhibition after treatment with DPP-4 inhibitors for 24 weeks.
Laboratory measurements
All blood samples for efficacy assessment were analyzed at the Seoul Clinical Laboratory, except for HbA1c and glucose collected during the first visit in Indian sites, which were analyzed at Super Religare Laboratories Ltd., Mumbai, India. Measurement techniques included the hexokinase method for glucose and enzymatic colorimetric assays (7600 Clinical Analyzer, Hitachi, Tokyo, Japan) for total cholesterol, low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and triglycerides. Immunoradiometric assays were used for insulin (INS-IRMA, BiosourceKit, Biosource, Nivelles, Belgium) and C-peptide (IRMA, Immunotech Kit, Immunotech, Prague, Czech Republic) measurements. HbA1c was measured using turbidimetric inhibition immunoassays (TINIA, A1C-2 [tina-quant hemoglobin A1c Gen2], Roche Diagnostics, Mannheim, Germany). Homeostasis model assessment of insulin resistance (HOMA-IR) and β-cell function (HOMA-β) at baseline were calculated as previously described [14]. Fasting plasma samples were stored at –80°C for measurement of DPP-4 activity using a continuous fluorometric assay with the substrate Gly-Pro-AMC (Bachem, Bubendorf, Switzerland) [15]. The degree of DPP-4 inhibition was calculated as 100×(1–W24/W0), where W0 was the enzyme activity measured before administration, and W24 was the activity measured after administration at week 24.
Statistical analysis
This post hoc analysis was based on the full analysis set population consisting of all patients at our institute who received the investigational drug at least once and showed signs of efficacy after randomization. Continuous variables were expressed as mean±standard deviation, whereas categorical variables were expressed as proportion (%). The tertile groups according to degree of DPP-4 inhibition were as follows: T1 <65.26%, T2=65.26%–76.35%, and T3 ≥76.35%. Demographic and biochemical characteristics of the study population according to degree of DPP-4 inhibition were compared using two-sample t-test or Wilcoxon’s rank sum test for continuous variables and the chi-square test for categorical variables. The difference in change of HbA1c from baseline to week 24 according to degree of DPP-4 inhibition was compared using Kruskal-Wallis test, while the change of HbA1c from baseline to week 24 within each tertile group by degree of DPP-4 inhibition was analyzed using Wilcoxon signed rank test. The association between degree of DPP-4 inhibition and HbA1c reduction was investigated by multiple linear regression, with HbA1c reduction as the dependent variable. This analysis was adjusted for age, ethnicity (Korean vs. Indian), body mass index, HbA1c at baseline, and DPP-4 activity at baseline. All statistical analyses were carried out with SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA).
RESULTS
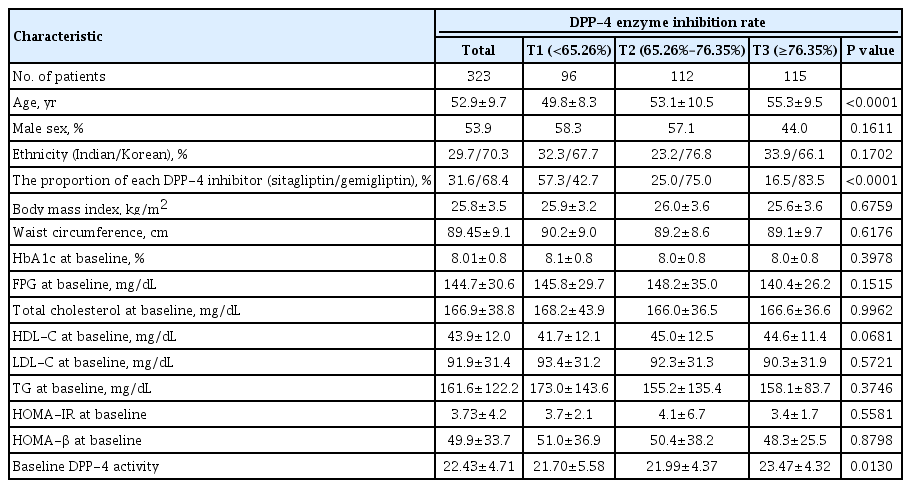
Demographics and baseline characteristics according to degree of DPP-4 inhibition
As previously described [13], 425 (296 from Korea and 129 from India) of 604 screened patients with T2DM were enrolled in the double-blind treatment period: 142 received sitagliptin 100 mg daily, 141 received gemigliptin 25 mg twice a day, and 142 received gemigliptin 50 mg daily. Among the 425 eligible patients, 323 had data for HbA1c level and DPP-4 activity at baseline and 24 weeks. Table 1 shows the baseline clinical and biochemical characteristics according to degree of DPP-4 inhibition. The average age of the study subjects was 52.9±9.7 years, and the average body mass index was 25.8±3.5 kg/m2. The mean HbA1c at baseline was 8.01%±0.8%, and mean fasting plasma glucose (FPG) value was 144.7±30.6 mg/dL. Sex (P=0.161), ethnicity (P=0.170), body mass index (P=0.676), waist circumference (P=0.618), HbA1c (P=0.398), FPG (P= 0.152), lipid profile (total cholesterol [P=0.996], HDL-C [P= 0.067], LDL-C [P=0.572], and triglyceride [P=0.375]), HOMA-IR (P=0.558), and HOMA-β (P=0.880) at baseline were similar across degrees of DPP-4 inhibition. However, the higher was the tertile by degree of DPP-4 inhibition, the higher was mean age (T1: 49.8±8.3 vs. T2: 53.1±10.5 vs. T3: 55.3± 9.5, P<0.001). And the higher tertile by degree of DPP-4 inhibition showed higher baseline DPP-4 activity (T1: 21.70±5.58 vs. T2: 21.99±4.37 vs. T3: 23.47±4.32, P=0.013)
HbA1c reduction according to degree of DPP-4 inhibition
Table 2 shows HbA1c at baseline and at week 24 after DPP-4 inhibitor treatment. The mean HbA1c at baseline was not different among the tertile groups by degree of DPP-4 inhibition (P=0.398) (Table 2). Overall, there was significant HbA1c reduction of –0.88%±0.76% after 24-week treatment (P<0.001) and in each tertile group by degree of DPP-4 inhibition (all P<0.001). The HbA1c at week 24 was significantly lower with increasing degree of DPP-4 inhibition (T1: 7.30%±0.88% vs. T2: 7.12%±0.78% vs. T3: 7.00%±0.78%, P=0.021). And the decrease of HbA1c from baseline to week 24 was higher with increasing degree of DPP-4 inhibition (T1: –9.60%±9.34% vs. T2: –10.29%±8.50% vs. T3: –12.12%±8.11%, P=0.062; P=0.037, between T1 and T3).
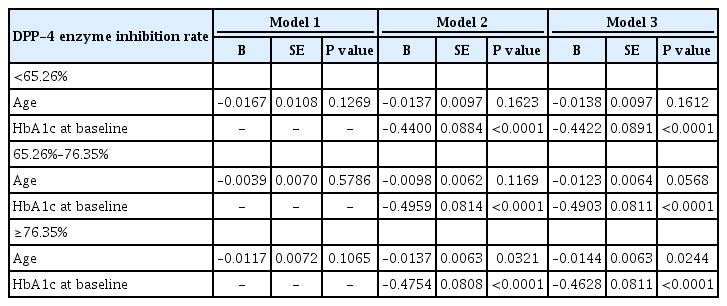
Independent association of degree of DPP-4 inhibition with HbA1c reduction
Table 3 shows the association of degree of DPP-4 inhibition with HbA1c reduction by multiple linear regression, with HbA1c reduction serving as the dependent variable.

The relationship between degree of DPP-4 inhibition and degree of HbA1c reduction from baseline to week 24
In model 3 (R2=0.240) in which age, ethnicity, body mass index, HbA1c at baseline, DPP-4 activity at baseline, and degree of DPP-4 inhibition were adjusted, age (B=–0.013, P=0.001), ethnicity (Korean vs. Indian, B=0.169, P=0.047), and HbA1c at baseline (B=–0.470, P<0.001) were significant determinants for HbA1c reduction. In model 6 in which age was not adjusted, degree of DPP-4 inhibition (R2=0.240, B=–0.005, P<0.031) was a significant determinant for HbA1c reduction and ethnicity (Korean vs. Indian, B=0.213, P=0.013), as was HbA1c at baseline (B=–0.463, P<0.001).
Predictors of better clinical response to DPP-4 inhibitors by degree of DPP-4 inhibition
We conducted subgroup analysis by tertile group according to degree of DPP-4 inhibition (T1 <65.26%, T2=65.26%–76.35%, and T3 ≥76.35%), with HbA1c reduction serving as the dependent variable (Table 4). In subgroup analysis, baseline HbA1c was a significant predictor of better clinical response to DPP-4 inhibitors in all subgroups, and age was a significant predictor of better clinical response to DPP-4 inhibitors in the highest tertile (B=–0.014, P=0.024), marginal in the middle tertile (B=–0.012, P=0.057), and not significant in the lowest tertile (B=–0.014, P=0.161) (Table 4).
DISCUSSION
This present study investigates the predictors of better clinical response (HbA1c reduction) to DPP-4 inhibitors with consideration of pharmacokinetic response (degree of DPP-4 inhibition). The predictors of clinical response to DPP-4 inhibitors were age, ethnicity, and HbA1c at baseline in this study. In the consideration of pharmacokinetic response, the highest degree of DPP-4 inhibition was associated with higher age and with greater HbA1c reduction, but it was dependent on patient age.
Some studies have investigated the HbA1c response to DPP-4 inhibitors in patients with T2DM, but the results were heterogeneous. A meta-analysis by Kim et al. [16] reported that DPP-4 inhibitors exhibit better glucose-lowering efficacy in Asians than in other ethnic groups, and body mass index was significantly correlated with HbA1c reduction in response to DPP-4 inhibitors in patients with T2DM, especially in an Asian population. The Predicting Response to Incretin Based Agents in Type 2 Diabetes (PRIBA) study comprised mostly of Caucasians showed that markers of higher insulin resistance were associated with reduction in response to DPP-4 inhibitors [5]. This suggests that Asian populations have a higher risk of developing diabetes due to genetic defects affecting insulin secretory function and β-cell mass compared to the Caucasian population developing diabetes due to insulin resistance [17]. Like the above study, Korean of this study showed lower insulin resistance (HOMA-IR: 3.15±1.59 vs. 5.33±6.66, P<0.001) and lower β-cell function (HOMA-β: 42.54%±22.28% vs. 67.85%±45.15%, P<0.001) (Supplementary Table 1) comparing with Indian of this study. And ethnicity (Korean vs. Indian) was statistically significantly associated with HbA1c reduction but not degree of DPP-4 inhibition in our study. These showed that there was significantly different in HbA1c reduction in response to DPP-4 inhibitors between ethnicity, but this difference was not relying on the degree of DPP-4 inhibition. Body mass index was not statistically significantly associated with HbA1c reduction or degree of DPP-4 inhibition in our study despite the Asian population. In other meta-analysis with 98 randomized clinical trials (RCTs) with 100 arms, Esposito et al. [6] observed that baseline HbA1c level explained most of the variance in HbA1c reduction in response to DPP-4 inhibitors (34% of the variance between studies), but mean baseline age, duration of treatment, and previous diabetes drugs did not provide predictive power (less than 1%) to the DPP-4 inhibitor treatment response. However, in another meta-analysis with 63 RCTs, Monami et al. [3] reported the results of meta-analysis with 63 RCTs that DPP-4 inhibitors are more effective in older patients with mild to moderate fasting hyperglycemia, compatible with our study. Compared to previous study, the strengths of our study include inclusion of degree of DPP-4 inhibition in the analysis as a predictor of clinical response to DPP-4 inhibitors. Age is an independent predictor of clinical response to DPP-4 inhibitor based on degree of DPP-4 inhibition.
Age is one of the most important risk factors for T2DM, and there is evidence of differences in pathophysiology of T2DM in older compared with younger patients. It has been hypothesized that impaired insulin secretion, rather than insulin resistance, is responsible for most diabetes in elderly populations compared with younger populations. It has been repeatedly reported that the capacity of pancreatic β-cells to provide adequate insulin for metabolic demand is decreased with increasing age [18-21]. This age-related loss of β-cell secretory capability has been attributed to several factors, including attenuation of the enteroinsular axis, incretin potentiate insulin secretion. Faerch et al. [22] have suggested that a reduction in GLP-1 response to oral glucose could predispose one to T2DM. Other longitudinal study showed that a low tertile of GLP-1 response to the oral glucose tolerance test was associated with a steeper increase in fasting glucose level than higher tertiles during 7 years of follow-up [23]. A recent longitudinal study showed that fasting GIP and GLP-1 and glucose-stimulated GLP-1 decreased significantly over a mean period of 5.9 years and suggested that reduction in incretin hormone responses with aging may predispose one to the glucose intolerance and T2DM [24]. A major mechanism of action of DPP-4 inhibitors is potentiation of β-cell insulin secretion and inhibition of glucagon secretion by elevating endogenous GLP-1 in a glucosedependent mode of action [25]. In our study, we divided study population into younger (<53 years old) and older (≥53 years old) group with median age (Supplementary Table 2), there were significant different in HOMA-IR between younger group (4.328±15.354) and older group (3.281± 15.354, P<0.001) but HOMA-β (%) at baseline were not different between younger group (53.13±42.47) and older group (46.94±23.15, P=0.10). Although there was significant difference in HbA1c difference from baseline between younger and older groups (–0.76±0.79 vs. –1.00±0.72, P<0.001), there was no proof that the insulin secretory function was more improved by DPP-4 inhibitors therapy in the older group compared to younger group. HOMAβ (%) change from baseline to week 24 were not different between younger and older groups (18.06±53.48 vs. 30.53± 123.35, P=0.25). And these were also similar in highest tertile of DPP-4 inhibition rate group (21.28±40.02 vs. 41.87± 123.35, P=0.45) (Supplementary Table 3). Therefore, there was possibility for other mechanism for HbA1c reduction by DPP-4 inhibitors in older patients with high DPP-4 inhibition rate, further study needed.
There are some limitations to this study. First, due to the nature of post hoc analysis, the sample size could be inadequate to find other less significant predictors of clinical response to DPP-4 inhibitors. Second, the relatively short-term follow-up of the study (24 weeks) could hinder evaluation of predictors of clinical response to DPP-4 inhibitors. However, previous randomized studies showed great HbA1c reduction within the first 8 to 12 weeks of such treatment [26,27]. Third, we described as the elder patients were more beneficial in the treatment with DPP-4 inhibitors. However, the average age of T3 was 55.3 years old and not very advanced age and we did not suggest a cut off of any age which was more beneficial in the treatment with DPP-4 inhibitors. Therefore ‘elder’ patients should not restrict to ‘elderly’ patients.
In conclusion, our study showed that age, ethnicity, and HbA1c at baseline were predictors of clinical response to DPP4 inhibitors. In elder but not younger patients with diabetes, DPP-4 enzyme inhibition rate was associated with HbA1c reduction. Therefore, a DPP-4 inhibitor might be adequate treatment in elder patient with diabetes.
Supplementary Materials
Supplementary materials related to this article can be found online at https://doi.org/10.4093/dmj.2020.0253
Baseline clinical and biochemical characteristics according to ethnicity
Baseline clinical and biochemical characteristics according to age group
Change of HOMA-β from baseline to week 24 according to degree of DPP-4 inhibition and age groups
Notes
CONFLICTS OF INTEREST
Sangmo Hong, Chang Hee Jung, and Cheol-Young Park had no potential conflict of interest relevant to this article. Song Han was hired by LG Chem Ltd., Seoul.
AUTHOR CONTRIBUTIONS
Conception or design: S.H., C.H.L., C.Y.P.
Acquisition, analysis, or interpretation of data: S.H, C.H.L., S.H., C.Y.P.
Drafting the work or revising: S.H., C.H.L.
Final approval of the manuscript: S.H., C.Y.P.
FUNDING
None
Acknowledgements
The authors thank the staff who conducted the original study, the patients for their participation and the industry sponsor (Life Sciences, LG Chem Ltd., Seoul, Korea) for coordination and funding of the original study.