Lobeglitazone: A Novel Thiazolidinedione for the Management of Type 2 Diabetes Mellitus
Article information
Abstract
Type 2 diabetes mellitus (T2DM) is characterized by insulin resistance and β-cell dysfunction. Among available oral antidiabetic agents, only the thiazolidinediones (TZDs) primarily target insulin resistance. TZDs improve insulin sensitivity by activating peroxisome proliferator-activated receptor γ. Rosiglitazone and pioglitazone have been used widely for T2DM treatment due to their potent glycemic efficacy and low risk of hypoglycemia. However, their use has decreased because of side effects and safety issues, such as cardiovascular concerns and bladder cancer. Lobeglitazone (Chong Kun Dang Pharmaceutical Corporation), a novel TZD, was developed to meet the demands for an effective and safe TZD. Lobeglitazone shows similar glycemic efficacy to pioglitazone, with a lower effective dose, and favorable safety results. It also showed pleiotropic effects in preclinical and clinical studies. In this article, we summarize the pharmacologic, pharmacokinetic, and clinical characteristics of lobeglitazone.
INTRODUCTION
Type 2 diabetes mellitus (T2DM) is a chronic progressive metabolic disease characterized by insulin resistance and β-cell dysfunction [1]. Because the pathophysiology of T2DM is complex and multifactorial, a variety of oral antidiabetic agents (OADs) have been developed based on the underlying mechanisms associated with T2DM. Among the OADs developed so far, the thiazolidinedione (TZD) class is one of the classes of OADs which primarily targets insulin resistance [2].
TZDs improve insulin sensitivity by activating peroxisome proliferator-activated receptor γ (PPARγ) [3] and were introduced as an OAD class in the late 1990s. The first TZD approved for T2DM treatment was troglitazone, which was withdrawn from the market in 2000 because of hepatotoxicity [4]. Other TZDs, rosiglitazone and pioglitazone, were approved by the U.S. Food and Drug Administration in 1999 and have been used widely for T2DM treatment due to their potent glycemic efficacy and low risk of hypoglycemia. They have also shown excellent glycemic durability [5,6]. However, because of side effects and safety issues, such as cardiovascular concerns [7] and bladder cancer [8], the use of rosiglitazone and pioglitazone has been restricted or decreased significantly during the 2010s.
Lobeglitazone (Chong Kun Dang Pharmaceutical Corporation, Seoul, Korea) is a novel TZD, developed to meet the demands for an effective and safe TZD. Its development program was initiated in May 2000 by Chong Kun Dang Pharmaceutical Corporation and it was approved for the management of T2DM in Korea in July 2013.
Although there is still a need for further studies, lobeglitazone has shown good results in terms of efficacy and safety in studies performed to date. In this article, we will review the pharmacologic, pharmacokinetic, and clinical characteristics of lobeglitazone.
LOBEGLITAZONE CHARACTERISTICS
Chemistry
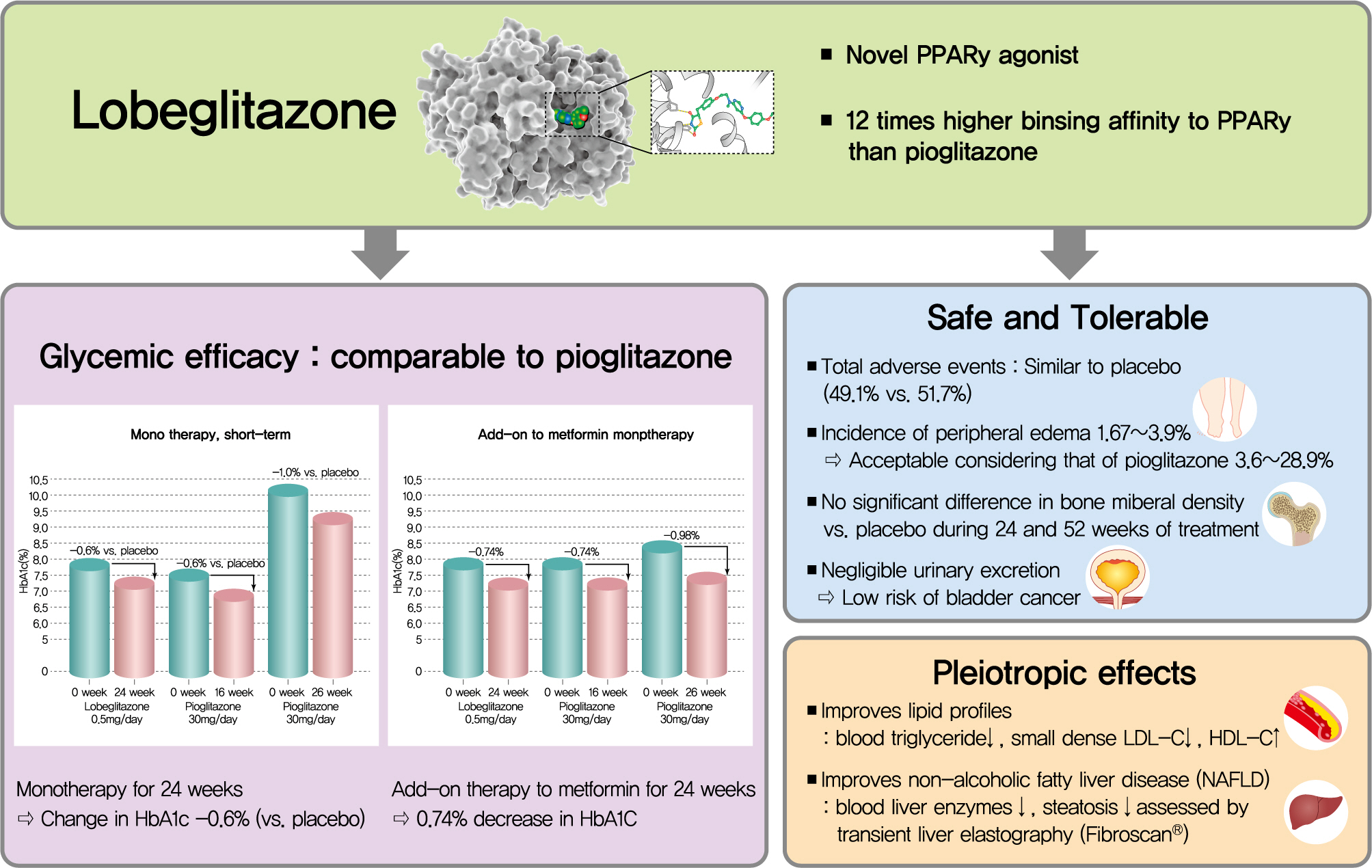
Lobeglitazone is a pharmacophore which has a 2,4-thiazolidinedione group with an ethoxy-benzyl N-methylamino group bound to this as a connecting link. Its structural formula is C24H24N4O5S, and the chemical name is 5-[4-(2-{[6-(4-Methoxy-phenoxy)-pyrimidin-4-yl]-methyl-amino}-ethoxy)- benzyl]-thiazolidine-2,4-dione hydrosulphuric acid. The cocrystal structure of lobeglitazone with PPARγ is shown in Supplementary Fig. 1.
Lobeglitazone was based on modification of the rosiglitazone structure to introduce a p-methoxyphenoxy group at the 4-position of the pyrimidine moiety [9]. This contributes to enhanced binding affinity of lobeglitazone for PPARγ, with docking analysis suggesting that the binding affinity of lobeglitazone is 12 times higher than that of rosiglitazone and pioglitazone [9]. The p-methoxyphenoxy group enables extended interaction with the hydrophobic pocket, and this could also affect the cyclin dependent kinase 5-mediated phosphorylation of PPARγ at Ser245, which changes the expression of genes such as adiponectin and adipsin that are associated with insulin sensitivity without general transcriptional activity of PPARγ [10].
Metabolic effects: preclinical studies
The effect of lobeglitazone on PPARγ activity was assessed by time-resolved fluorescence resonance energy transfer (TRFRET) assay. The half maximal effective concentration (EC50) of lobeglitazone for PPARγ was 0.1374 μM, and it showed selectivity for PPARγ (PPARα/PPARγ EC50 ratio=3,976) [11]. The EC50 values of rosiglitazone and pioglitazone for PPARγ were 0.1076 and 0.5492 μM, respectively.
In in vivo studies, lobeglitazone showed a significant glucose-lowering effect in Zucker diabetic fatty (ZDF) rats, db/db and KK/Upi-Ay/J (KKAy) mice. Lobeglitazone acted in a dosedependent manner and was most effective at 0.3 mg/kg, at which dose it was equivalent to 30 mg/kg of pioglitazone [11]. Lobeglitazone increased glucose uptake in 3T3-L1 adipocytes and L6 muscle cells, with the effects greater than those observed with pioglitazone [11], and showed beneficial effects on pancreatic β-cell survival in db/db mice [12,13]. It was also found to inhibit protein tyrosine phosphatase 1B in vitro; this enzyme is involved in the regulation of insulin and leptin signaling and is implicated in the development of insulin resistance and T2DM [14].
Other metabolic effects of lobeglitazone have also been evaluated. The effect of lobeglitazone on lipid profiles was compared to that of pioglitazone in ZDF rats, with plasma triglyceride (TG) and free fatty acid (FFA) levels reduced similarly in both treatment groups (TG reduced by 77% with lobeglitazone 10 mg/kg and 75% with pioglitazone 10 or 30 mg/kg; FFA reduced by 98% with lobeglitazone 10 mg/kg and 97% with pioglitazone 10 or 30 mg/kg) [11]. Lobeglitazone enhanced lipid accumulation and, adipocyte differentiation through increasing the expression of genes regulated by PPARγ such as adipocyte proterin 2 (aP2), and CD36 to a greater extent than seen with other TZDs in vitro [11]. Long-term administration of lobeglitazone had a beneficial role on adipose tissue remodeling, by inducing differentiation of preadipocytes into insulinsensitive small adipocytes, and redistribution from visceral to subcutaneous adipose depots, and induced the differentiation of white adipocytes to beige adipocytes and was associated with the development of brown adipocytes [13]. Lobeglitazone also enhanced beige adipocyte formation and thermogenic gene expression in lean mice after cold exposure, to a greater extent than observed with pioglitazone or rosiglitazone [15].
Potential pleiotropic effects: preclinical studies
Similar to the therapeutic effect of pioglitazone on fatty liver, lobeglitazone also attenuated hepatic steatosis in obese mice by increasing insulin sensitivity and inhibiting hepatic lipogenesis [16,17].
A few studies have reported favorable effects of lobeglitazone on atherosclerosis in in vitro or in vivo models [18,19]. Lobeglitazone significantly reduced neointimal formation after balloon injury in rat carotid arteries and reduced the atheromatous burden in the aorta of ApoE−/− mice fed with a high-fat/high-cholesterol diet [18]. These results were in line with previous studies showing that PPARγ activation inhibits vascular smooth muscle cell proliferation and inflammatory pathways and, therefore, TZDs might be expected to protect or relieve the endothelium from atherosclerosis [20,21]. Lobeglitazone also inhibited the formation of lipid droplets in foam cells in an in vitro model [22].
Anti-inflammatory characteristics of lobeglitazone were evident in a murine model of asthma [23], and lobeglitazone improved renal fibrosis in mice by inhibiting transforming growth factor-β/Smad signaling [24].
These preclinical studies suggest that lobeglitazone might have beneficial effects on various organs; however, clinical studies are needed to evaluate the potential role of lobeglitazone in these settings.
Other studies evaluated potential safety concerns with TZDs, including bone health and cancer risk. Both adipocytes and osteoblasts differentiate from mesenchymal stem cells, a process that is in part regulated by PPARγ [25]. Consequently, there is a concern that long-term administration of TZDs could have adverse effects on bone, as was seen with rosiglitazone which reduced bone mineral density (BMD) [26,27]. Studies with lobeglitazone found that it did not inhibit osteoblast differentiation in vitro and had no adverse effect on BMD in mice [25].
Finally, there is evidence that some PPAR agonists, such as pioglitazone, may increase the risk of bladder cancer [28,29]. Long-term studies in rats and mice suggested that lobeglitazone had a low carcinogenic potential and did not induce urothelial tumors [30,31].
Pharmacokinetics
Data on the pharmacokinetics of lobeglitazone have been obtained from healthy volunteers and patients with renal impairment.
Healthy adults
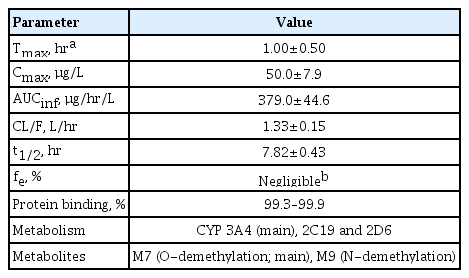
After single oral doses of lobeglitazone in healthy male subjects, peak plasma levels of lobeglitazone were reached at 1 to 3 hours and then declined monoexponentially with a half-life of 7.8 to 9.8 hours [32]. In males, peak plasma concentration (Cmax) and area under the plasma concentration–time curve from time 0 to infinity (AUCinf) increased in a dose-proportional manner across the dose range of 1 to 4 mg [32]. After multiple-dose administration (once-daily regimen) in healthy males, lobeglitazone reached steady-state concentrations by day 5, and no clinically significant drug accumulation was observed [32].
When a single doses of lobeglitazone was administered to healthy female subjects, systemic lobeglitazone exposure after 2 mg was similar to that seen in males, but after administration of 4 mg, exposure was 2.36-fold greater in females than in males [33]. However, dose adjustment based on sex alone is not necessary because the approved dose for clinical use is lower (0.5 mg/day). Pharmacokinetic parameters after a single dose of lobeglitazone 0.5 mg in males are summarized in Table 1.
Lobeglitazone clearance was mainly mediated by liver metabolism and the ratio of renal excretion was predicted to be <1.0% [32]. Based on an in vitro human liver microsomal study, the main cytochrome P450 (CYP) isoforms involved in the hepatic metabolism of lobeglitazone were cytochrome P450 3A4 (CYP3A4), 2C19, and 2D6 [11].
Food interaction
In a study that evaluated the effect of food on the pharmacokinetics of a single dose of lobeglitazone 1 mg, a slight increase in the AUC for lobeglitazone was observed in subjects who were receiving a high lipid diet compared with fasting subjects [11]. There were no significant between-group differences in the Cmax of lobeglitazone or its active metabolite M7, and the slight increase in bioavailability with a high lipid diet was not considered to be clinically significant.
In a study evaluating a fixed-dose combination tablet containing lobeglitazone 0.5 mg plus metformin extended-release 1,000 mg, lobeglitazone Cmax decreased by approximately 32% when administered after a high-fat meal compared with the fasting state; however, there was no significant difference between the fed and fasted states with respect to lobeglitazone AUC up to the last sampling time [34].
Patients with renal impairment
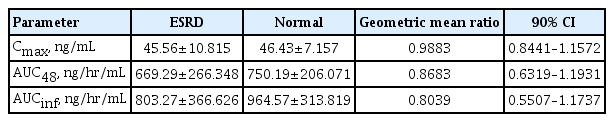
An open-label, parallel-group, single-dose, non-randomized study was conducted to evaluate the pharmacokinetic profile of lobeglitazone in patients with renal impairment [11]. The geometric means for Cmax and AUC in patients with end-stage renal disease (ESRD) were similar to those for subjects with normal renal function (Table 2). Cmax and AUCinf values for M7 (the active metabolite of lobeglitazone) were 1.22 and 1.31 times higher in the ESRD group compared with the control group. However, overall, the pharmacokinetic profiles did not differ significantly between the two groups (Table 2).
Drug interactions
In studies evaluating potential drug–drug interactions in healthy male volunteers, no significant changes were observed in the pharmacokinetics of either drug when lobeglitazone was coadministered with other antidiabetic agents including metformin, glimepiride, dapagliflozin, and sitagliptin [11,35-37]. In general, the 90% confidence intervals (CIs) for the geometric mean ratios (combination vs. single drug) for Cmax and AUC were within the conventional equivalence range of 80% to 120%. Exceptions were a slightly increased steady-state Cmax for sitagliptin (ratio, 1.1694; 90% CI, 1.0740 to 1.27320) and a slightly decreased Cmax for glimepiride (ratio, 0.910547; 90% CI, 0.78246 to 1.05960), when each was coadministered with lobeglitazone; however, neither of these changes were considered to be clinically significant [11,37].
There were also no significant changes in the pharmacokinetics of either drug when lobeglitazone was coadministered with amlodipine or warfarin [38,39]. However, coadministration with ketoconazole, a strong CYP3A4 inhibitor, increased the level of exposure to lobeglitazone by about 33% (geometric mean ratio for AUCinf, 1.33; 90% CI, 1.23 to 1.44) [40].
LOBEGLITAZONE CLINICAL PROFILE
Glycemic control in type 2 diabetes mellitus
Monotherapy
The short-term glycemic efficacy of lobeglitazone was initially evaluated in a randomized, double-blind, placebo-controlled, phase II study. Patients with T2DM (n=241) received lobeglitazone 0.5 mg (n=55), 1 mg (n=54), or 2 mg (n=50), or placebo (n=55) for 8 weeks [11]. Changes in fasting plasma glucose levels after 8 weeks of treatment (primary endpoint) were –20.65 mg/dL (0.5 mg), –23.38 mg/dL (1 mg), –33.69 mg/dL (2 mg), and –1.8 mg/dL (placebo). The frequency of edema increased with the higher doses of lobeglitazone, and therefore the minimum effective dose of 0.5 mg once daily was selected as the standard dose for further evaluation.
Subsequently, a phase III study compared the glycemic efficacy of lobeglitazone 0.5 mg (n=115) with placebo (n=58) over 24 weeks [41]. Mean changes in glycosylated hemoglobin (HbA1c) from baseline to week 24 (primary endpoint) were –0.44% in the lobeglitazone group versus 0.16% in the placebo group (mean difference, –0.6%; P<0.0001). In a 28-week openlabel extension study, patients who completed the 24-week study either continued on lobeglitazone (n=65) or switched from placebo to lobeglitazone (n=29) [42]. In those patients who received lobeglitazone for 52 weeks, the decrease in HbA1c seen at week 24 was maintained during the extension period; the mean change in HbA1c from baseline to week 52 was –0.50%. In patients who switched from placebo to lobeglitazone at week 25, HbA1c improved after administration of lobeglitazone and the mean change in HbA1c from baseline to week 52 was –0.52%, similar to that seen in the group who remained on lobeglitazone for the entire 52 weeks.
Combination therapy
The antihyperglycemic effect of lobeglitazone in combination with metformin was evaluated in a 24-week, randomized, double-blind, non-inferiority trial [43]. Patients with T2DM whose blood glucose levels were inadequately controlled (HbA1c 7% to 10%) despite a stable dose of metformin (≥1,000 mg/day) were randomized to receive lobeglitazone 0.5 mg daily (n=128) or pioglitazone 15 mg daily (n=125) as addon therapy to metformin. After 24 weeks of lobeglitazone addon therapy, the mean change in HbA1c (primary endpoint) was −0.74%, which was almost the same as the change seen in the pioglitazone group (mean difference, 0.01%; 95% CI, –0.16% to 0.18%), indicating that lobeglitazone was not inferior to pioglitazone as add-on therapy to metformin with respect to the glucose-lowering effect.
In another randomized controlled study, 24 weeks of treatment with lobeglitazone (n=121) as an add-on to metformin was non-inferior to sitagliptin add-on therapy (n=126) with respect to glycemic efficacy (mean change in HbA1c –0.79% vs. –0.86%; difference, 0.08%; 95% CI, –0.14% to 0.30%) [44].
Apart from metformin, no prospective randomized studies comparing the glycemic efficacy of dual-combination therapy containing lobeglitazone versus other OADs have been published to date. However, a retrospective study assessed the glycemic efficacy of lobeglitazone as monotherapy and in various forms of combination therapy [45]. The study, conducted in a routine clinical practice setting, analyzed 423 patients who took lobeglitazone for more than 180 consecutive days (average age 62.7 years, average duration of T2DM 8.5 years, baseline HbA1c 7.5%). After a median of 350 days of lobeglitazone treatment, the mean change in HbA1c was –0.6% overall; for lobeglitazone monotherapy it was –0.34%, for combination therapy with metformin it was –0.52%, in combination with dipeptidyl peptidase-4 (DPP-4) inhibitors –0.63%, and in combination with a sulfonylurea –0.33%. Regarding the triple therapy, the mean change in HbA1c was –0.84% for patients taking lobeglitazone, metformin, and DPP-4 inhibitor, –0.88% for those taking lobeglitazone, DPP-4 inhibitor and sulfonylurea, and –0.33% for those taking lobeglitazone, metformin, and sulfonylurea. These results suggested that when lobeglitazone is treated in combination with DPP-4 inhibitor, it would improve blood glucose levels more potently than other lobeglitazone-combining regimens.
Recently, a prospective observational study specifically evaluated the effect of initial triple combination therapy including lobeglitazone for drug-naïve patients with T2DM with an HbA1c level from 9.0% to 12.0% [46]. This study enrolled patients consecutively and used matching based on age and body mass index to select a comparator group who were treated with conventional stepwise dual therapy. After 12 months, HbA1c had decreased by a mean of 4.05% among recipients of initial triple therapy comprising metformin 1,000 mg/day plus sitagliptin 100 mg/day plus lobeglitazone 0.5 mg/day compared with 3.28% among recipients of uptitrated treatment with metformin ≥1,000 mg/day plus glimepiride ≥2 mg/day (P<0.05). Although this study focused on the effectiveness of the initial triple therapy, it also provided evidence for the efficacy and safety of lobeglitazone as a combination therapy for T2DM.
Indirect comparison with pioglitazone
No randomized studies have directly compared lobeglitazone with pioglitazone as monotherapy. An indirect comparison of the glycemic efficacy of lobeglitazone with pioglitazone based on relevant studies is summarized in Table 3, Fig. 1 [41-43,47-51]. Among patients who had a similar level of glycemic control at baseline, lobeglitazone appeared to show similar glucose-lowering efficacy to pioglitazone. Although the results of some studies seemed to suggest that pioglitazone might have a more potent effect, it should be noted that those studies recruited patients with higher baseline HbA1c.
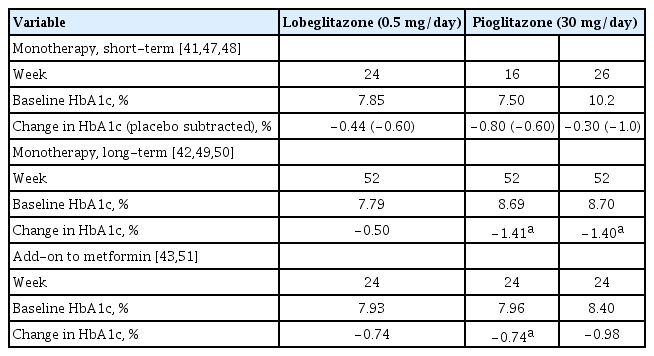
Glycemic efficacy of lobeglitazone in patients with type 2 diabetes mellitus: indirect comparison with pioglitazone based on prospective randomized controlled studies

Indirect comparison of glycemic efficacy between lobeglitazone and pioglitazone in patients with type 2 diabetes mellitus based on previous prospective randomized, controlled studies. (A) Monotherapy, short-term, (B) monotherapy, longterm, and (C) add-on to metformin monotherapy. HbA1c, glycosylated hemoglobin.
Potential pleiotropic effects
Lipid profiles
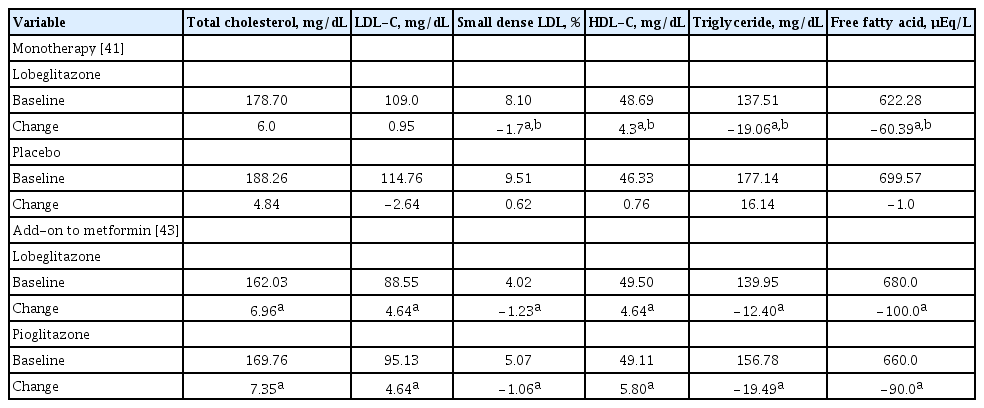
Treatment with lobeglitazone for 24 weeks improved blood levels of TG, high-density lipoprotein cholesterol, and small dense low-density lipoprotein cholesterol (small dense LDLC) [41]. Improvements in TG and small dense LDL-C remained statistically significant during a 28 week-extension period [42]. Similar effects on lipid profiles were seen in the study of lobeglitazone as add-on therapy to metformin [43]. In that study, there were no significant differences in lipid parameters between lobeglitazone and pioglitazone. The effect of lobeglitazone on lipid profiless is summarized in Table 4.
Non-alcoholic fatty liver disease
TZDs are one of the few oral agents recommended as a treatment for non-alcoholic fatty liver disease (NAFLD) [52]. The effects of lobeglitazone on NAFLD were investigated in a single-arm study using transient liver elastography (Fibroscan; Echosens, Paris, France), as well as glycemic, lipid, and liver enzyme profiles [53]. Fifty T2DM patients with NAFLD, defined as controlled attenuation parameter (CAP) ≥250 dB/m, received lobeglitazone 0.5 mg once daily. After 24 weeks, mean CAP significantly decreased (from 313.4±30.9 to 297.8±39.1 dB/m, P=0.016). Liver enzymes including aspartate transaminase, alanine transaminase, and gamma glutamyl transferase also decreased. When patients were grouped as lobeglitazone responders and non-responders in terms of CAP changes, mean changes in HbA1c levels did not differ significantly between the groups, indicating that the therapeutic effect in NAFLD by lobeglitazone was independent of its glycemic efficacy.
Safety and tolerability
Lobeglitazone was generally well tolerated as monotherapy and in combination with metformin (with or without sitagliptin) [41-43,46]. The proportions of adverse events were similar between the lobeglitazone and placebo groups in the placebo-controlled monotherapy trial [41,42] and the lobeglitazone and pioglitazone groups (Table 5) [43] as well as between the lobeglitazone and sitagliptin groups [44] in the active-controlled metformin combination therapy trials.
Peripheral edema, one of the most frequent side effects associated with TZDs, occurred in 3.6% of patients treated with lobeglitazone monotherapy for 24 weeks [41], and 3.1% of patients by the end of the monotherapy extension study (total 52 weeks) [42]. No patients had severe edema requiring treatment discontinuation. When lobeglitazone was administered in combination with metformin, 3.9% of patients experienced edema within 24 weeks, with no cases of severe edema [43]. In another study, peripheral edema and facial edema occurred in 1.67% and 3.33% of patients treated with lobeglitazone as addon therapy to metformin [44]. The incidence of peripheral edema seems to be acceptable, considering that the frequency of peripheral edema in clinical studies using pioglitazone monotherapy ranged from 3.6% to 28.9% [47-49,54-56].
Weight gain is expected in patients treated with TZDs [2,57]. In the monotherapy trial, the mean bodyweight gain in the lobeglitazone group after 24 weeks was 0.89 kg, whereas in the placebo group bodyweight changed by –0.63 kg (mean difference, 1.52 kg; P<0.0001) [41]. When used as add-on therapy to metformin, mean weight gain was 0.92 kg in the lobeglitazone group, which did not differ significantly from that of the pioglitazone group (0.76 kg, P=0.569) [43]. Weight gain was greater with lobeglitazone than with sitagliptin when used as add-on therapy to metformin (mean difference, 1.34 kg; P<0.0001) [44].
Hypoglycemia was not reported in the monotherapy trial [41,42], while one subject (0.8%) in the lobeglitazone group versus three subjects (2.4%) in the pioglitazone group reported hypoglycemia when they were used as add-on therapy to metformin [43].
A potential concern with TZDs is an increased risk of bone fractures associated with reduced BMD [2]. The monotherapy trial [41,42] incorporated an evaluation of femur neck and total hip BMD using dual-energy X-ray absorptiometry at week 24 and the week 52, and found no statistically significant difference in change in BMD between lobeglitazone and placebo [58]. Longer-term and mechanistic studies are needed to confirm this finding.
Other potential concerns with some TZDs include heart failure and bladder cancer [2]. Neither of these were observed in either the lobeglitazone monotherapy or combination therapy trials. Due to the small sample sizes and short study durations, these studies cannot definitively confirm the long-term clinical benefits and risks, and further research is needed to assess cardiovascular outcomes and the risk of bladder cancer with lobeglitazone. However, the risk of bladder cancer with lobeglitazone is considered low based on preclinical studies [30,31] and its negligible urinary excretion [9,32].
Ongoing studies
Several clinical studies instigated by CKD Pharmaceutical Corp are currently ongoing, and the results are not yet available. These include efficacy and safety trials of lobeglitazone as add-on combination therapy with DPP-4 inhibitors (ClinicalTrial.gov registration number: NCT03641352), sodium glucose cotransporter-2 (SGLT-2) inhibitors (ClinicalTrial.gov registration number: NCT03627182) and insulin (cris.nih. go.kr registration number: KCT0002852), a comparison of changes in HbA1c between lobeglitazone 0.25 and 0.5 mg in patients with T2DM (ClinicalTrial.gov registration number: NCT03770052), and additional pharmacokinetic and drug– drug interaction studies with DPP-4 inhibitors (ClinicalTrial. gov registration number: NCT04431687) and SGLT-2 inhibitors (ClinicalTrial.gov registration number: NCT04334213).
CONCLUSIONS
Based on the studies summarized in this review, lobeglitazone is a novel TZD with potent efficacy and a favorable safety profile. Lobeglitazone shows similar glycemic efficacy to pioglitazone, with a lower effective dose, owing to its higher affinity to PPARγ. Because lobeglitazone is mainly metabolized by the liver, with negligible renal excretion, it is expected that it can be used in patients with renal insufficiency without dose reduction, and it may have a lower risk of bladder cancer than other TZDs. Clinical trials conducted to date also have shown favorable results on safety of lobeglitazone. Additional clinical and preclinical studies are expected to provide further evidence of the beneficial effects of lobeglitazone and its mechanism of action.
Supplementary Materials
Supplementary materials related to this article can be found online at https://doi.org/10.4093/dmj.2020.0272
X-ray crystallographic structure of lobeglitazone (green) bound to the active site of peroxisome proliferator-activated receptor γ.
Notes
CONFLICTS OF INTEREST
The authors report no potential conflict of interest relevant to this article. Laboratory data analysis and calibration support was provided by Chong Kun Dang Pharmaceutical Corporation, Seoul, Korea.
FUNDING
None
Acknowledgements
Under the direction of the authors, medical writing assistance was provided by Kathy Croom and David P. Figgitt PhD, ISMPP CMPPTM, Content Ed Net.