Predicting Mortality of Critically Ill Patients by Blood Glucose Levels
Article information
Abstract
Background
The aim of this study is to observe the outcome of critically ill patients in relation to blood glucose level at admission and to determine the optimal range of blood glucose at admission predicting lower hospital mortality among critically ill patients.
Methods
We conducted a retrospective cohort study of a total 1,224 subjects (males, 798; females, 426) admitted to intensive care unit (ICU) from 1 January 2009 to 31 December 2010. Blood glucose levels at admission were categorized into four groups (group 1, <100 mg/dL; group 2, 100 to 199 mg/dL; group 3, 200 to 299 mg/dL; and group 4, ≥300 mg/dL).
Results
Among 1,224 patients, 319 patients were already known diabetics, and 296 patients died in ICU. Five hundred fifty-seven subjects received insulin therapy, and 118 received oral hypoglycemic agents. The overall mortality rate was 24.2% (296 patients). The causes of death and mortality rates of diabetic patients were not different from nondiabetic subjects. The mortality curve showed J shape, and there were significant differences in mortality between the groups of blood glucose levels at admission. Group 2 had the lowest mortality rate (P<0.05).
Conclusion
These results suggest that serum glucose levels upon admission into ICU is associated with clinical outcomes in ICU patients. Blood glucose level between 100 and 199 mg/dL at the time of ICU admission could predict lower hospital mortality among critically ill patients.
INTRODUCTION
Hyperglycemia is common in critically ill patients during hospital admission, even in those who have not been diagnosed with diabetes mellitus [1,2]. The prevalence of hyperglycemia among hospitalized patients is estimated to be around 40% [3]. The release of stress hormones (e.g., epinephrine and cortisol), leads to increase in adipose tissue lipolysis and skeletal muscle proteolysis. Hepatic glycogenolysis mediated by catecholamine, as well as the direct sympathetic stimulation of glycogen breakdown, lead to hyperglycemia. The use of exogenous glucocorticoids and catecholamines, in addition to the release of proinflammatory cytokines, such as tumor necrosis factor, interleukin (IL)-1, and IL-6 in cases of sepsis or surgical trauma, also contribute to hyperglycemia in critically ill patients [4]. The use of intravenous dextrose commonly used in parenteral nutrition and solutions of intravenous medication could also contribute to hyperglycemia [5]. Hyperglycemia is linked to poor immune response, inflammatory changes, vascular response, thrombosis, increased cardiovascular events, brain cell response, delayed healing, and other problems [6]. Therefore, poorly controlled hyperglycemia is associated with increased morbidity and mortality in critically ill patients [7]. There has been some clinical trials of glucose management in critically ill patients. These trials examining the effects of tight glucose control have revealed conflicting results [8-16]. Moreover, systematic reviews and meta-analyses have derived different conclusions [17,18].
In this study, we aimed to observe the outcome of critically ill patients in relation to blood glucose levels upon admission to intensive care unit (ICU) and to identify the optimal range of blood glucose upon ICU admission that predicts lower hospital mortality among critically ill patients.
METHODS
Subjects
We obtained data on 1,247 patients who were admitted to ICU of Yeungnam University Medical Center from January 1 2009 to December 31 2010. 23 subjects who spent less than 24 hours in ICU were excluded from this study. The total subjects were composed of 798 males and 426 females.
Study design
We performed a retrospective cohort study involving both medical and surgical patients admitted to the ICU. Our ICU is a closed unit that does not service cardiologic and neurosurgical patients. Admission blood glucose level was defined as the first nonfasting serum blood glucose level measured at the time of the admission to ICU. All subjects were assigned to one of the four groups according to admission blood glucose levels based on previously studied clinical trials on admission blood glucose (group 1 [n=110], below 100 mg/dL; group 2 [n=799], above 100 mg/dL and below 200 mg/dL; group 3 [n=246], above 200 mg/dL and below 300 mg/dL; and group 4 [n=69], above 300 mg/dL) [19,20].
Primary outcome was defined as death from any cause. The causes of death were divided into four categories: multiple organ failure with proven septic focus, multiple organ failure without detectable septic focus, severe brain damage, and acute cardiovascular collapse. The mortality was analyzed according to presence of diabetes and the use of intravenous insulin to control blood glucose.
The protocol was approved by the Institutional Review Board of Yeungnam University Medical Center. Informed consent was exempted by the board due to the retrospective nature of the study.
Statistical analysis
The baseline characteristics of subjects were collected, including demographic, clinical characteristics, past medical history, and medication history. Mean±standard deviation was calculated for continuous variables. Comparisons of continuous variables between groups were carried out using Student t-test and one-way analysis of variance when appropriate. A two-level logistic regression analysis was used to determine the relationship between admission blood glucose level and mortality. The mortality rates between the groups were compared with Kaplan-Meier curves. All statistical analyses were performed using SPSS version 20.0 for Windows (IBM Co., Armonk, NY, USA) with 5% significance level (P<0.05).
RESULTS
Study population
The baseline characteristics of the subjects are shown in Table 1. The mean age of subjects was 63.48±13.68 years, and 65.2% of subjects were male. Of 1,224 total subjects, 319 were already known diabetics, and 905 were not. Five hundred and fifty-seven patients received insulin therapy, and 118 had received oral hypoglycemic agents (OHAs) during ICU stay. The history of hypertension, frequency of insulin or OHA using and mean serum glucose, blood urea nitrogen, creatinine were significantly higher among diabetic patients (P<0.05). The diagnosis at admission was divided into nine categories: respiratory, gastrointestinal or liver, hemato-oncologic, other sepsis, cardiovascular, neurologic, renal, metabolic diseases, and others. The distribution of diagnosis at admission was not different between diabetics and nondiabetics. The mean length of stay in ICU was 25.44 days, which was not different between the two groups.
Mortality
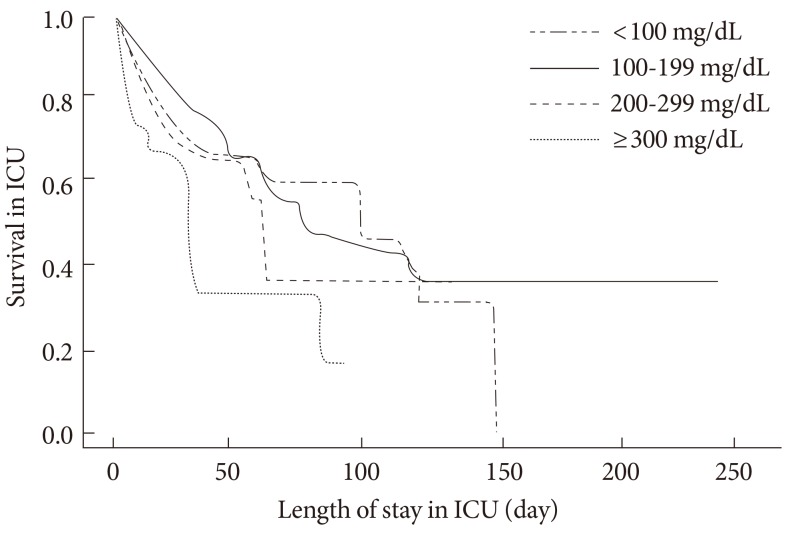
The total mortality rate was 24.2% (296 subjects), and it was not different between diabetic and nondiabetic subjects (Table 1). The mortality rate was not significantly different between diabetics and nondiabetic subjects (26.0% vs. 23.4%; P=0.40), and the causes of death were also not different. Mortality rates according to admission blood glucose levels are shown in Fig. 1. The mortality rate was the lowest in the group 2, and mortality curve showed J shape (group 1, 31%; group 2, 20%; group 3, 24%; group 4, 52%; P<0.05 for trend). These tendencies were maintained when the subjects were divided into the diabetic (group 1, 25%; group 2, 21%; group 3, 25%; group 4, 68%; P<0.05 for trend) and nondiabetic patients (group 1, 34%; group 2, 20%; group 3, 25%; group 4, 45%; P<0.05 for trend). In a two-level logistic regression analysis, the odds ratio of mortality for group 1, group 3, and group 4 were 1.41 (95% confidence interval [CI], 1.36 to 1.49), 1.33 (95% CI, 1.26 to 1.39), and 1.97 (95% CI, 1.83 to 2.12) compared to group 2 (P<0.05). Group 2 had the highest survival rate in Kaplan-Meier curves (Fig. 2). The mortality rates were not different statistically between insulin group (28.7%, n=160) and noninsulin group (20.3%, n=136).
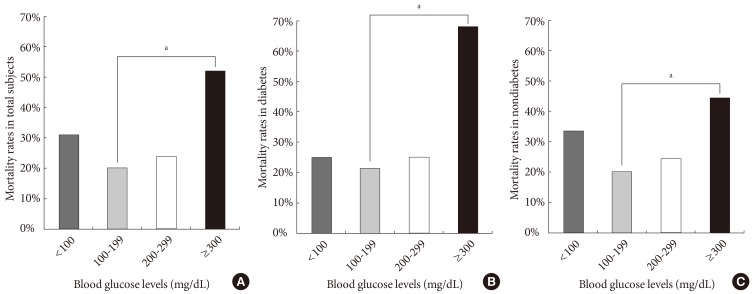
(A-C) Relationship between blood glucose levels and mortality rate in each subjects group (P<0.05 for trend). aP<0.05.
DISCUSSION
Hyperglycemia is associated with increased mortality and poor outcome in hospitalized patients, even those who have not previously had diabetes. Traditionally, acute hyperglycemia has been defined as random glucose concentration greater than 200 mg/dL [21], but in 2010, the American Diabetes Association proposed a threshold of 140 mg/dL [22].
Stress hyperglycemia occurs in acute injury and illness due to several mechanisms, and it alters the immune system, mediators of inflammation, vascular response, and brain-cell response [3]. Hyperglycemia itself causes immunosupression leading to infection, and it also increases blood pressure and natriuretic peptide levels. Hyperglycemia also results in numerous adverse effects on the cardiovascular system, including impaired protective mechanism against cardiac insult. Acute hyperglycemia induces platelet hyperactivity causing thrombotic events in hospitalized patients and is associated with enhanced neuronal damage inducing brain ischemia [23]. Therefore, the recently published guidelines have recommended a blood glucose target between 140 and 180 mg/dL for critically ill patients [24,25].
In one study of 6,891 community acquired pneumonia patients, those with mild hyperglycemia upon admission (serum glucose levels, 108 to 198 mg/dL) had a significantly increased risk of death at 90 days (hazard ratio, 1.56), which increased to 2.37 when admission blood glucose levels were ≥252 mg/dL [26]. In another study of 1,550 critically ill children, patients with high serum glucose level in the first day of pediatric ICU admission (>200 mg/dL) had longer mechanical ventilation days, longer pediatric ICU length of stay, and lower percentage of survival than those with normal serum glucose (≤200 mg/dL) [27]. Furthermore, patients with admission hyperglycemia (>135 mg/dL) had higher hospital mortality than those with normoglycemia (<100 mg/dL) in polytraumatised patients [19]. Although this study did not include cardiologic and neurosurgical patients, admission blood glucose was also associated with in-hospital mortality in acute ST-elevation myocardial infarction [28] and with severity, mortality, and poor functional recovery in patients with intracerebral hemorrhage [20,29].
Our study showed that blood glucose levels upon admission influences mortality. The patients who had admission blood glucose ranging between 100 and 199 mg/dL had the lowest mortality rate, which is similar to previous studies. The mortality demonstrated a J shaped curve. A recent large randomized NICE-SUGAR study including 6,104 patients in ICU reported that intensive glucose control group (target blood glucose range, 81 to 108 mg/dL) had higher risk of severe hypoglycemia and increased mortality than conventional control group (target of 180 mg/dL or less) among patients in ICU [16]. Additionally, in a study of 66,684 critically ill patients, early hypoglycemia (within 24 hours of admission, blood glucose <81 mg/dL) was associated with increased risk of hospital death (odds ratio, 1.36) [30]. It is possible that the risk of hypoglycemia is higher in group 1, which may have contributed to increased mortality of this group.
The admission blood glucose range with the lowest mortality rate was not significantly different between diabetes and nondiabetes nor was the mortality rate of subjects with diabetes different from those who without diabetes. Furthermore, the mortality rate was not influenced by insulin therapy. The lack of differences in mortality rate between diabetics and nondiabetics or insulin therapy and oral pharmacologic therapy may be due to stress hyperglycemia per se more influencing mortality more powerfully than the pathophysiology underlying the hyperglycemia.
Our study has several limitations. We didn't divide subjects into surgical or medical patients. The assessment of degrees of disease severity, such as Acute Physiology and Chronic Health Evaluation score, which was missed in our analysis could affect mortality. Another limitation is the lack of duration of diabetes and hemoglobin A1c on ICU admission, which may be important confounders. In addition, other confounders, such as the use of glucocorticoids, exogenous catecholamine, and parenteral nutrition, were not evaluated in this study, which could also influence hyperglycemia and mortality. However, since this study evaluated the association between mortality and blood glucose levels upon ICU admission, rather than those during ICU stay, we could exclude the effects of glucose-lowering interventions on mortality. This study was an observational study which identified the optimal range of admission serum glucose concentrations predicting lower in-hospital mortality.
In conclusion, the blood glucose concentrations upon ICU admission was associated with clinical outcomes in ICU patients, and admission blood glucose ranges of 100 to 199 mg/dL predicted lower hospital mortality among critically ill patients.
Notes
No potential conflict of interest relevant to this article was reported.