- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 44(1); 2020 > Article
-
Original ArticleDrug/Regimen An Electronic Health Record-Integrated Computerized Intravenous Insulin Infusion Protocol: Clinical Outcomes and
in Silico Adjustment -
Sung Woon Park1
 , Seunghyun Lee2
, Seunghyun Lee2 , Won Chul Cha3,4, Kyu Yeon Hur1, Jae Hyeon Kim1, Moon-Kyu Lee1, Sung-Min Park2
, Won Chul Cha3,4, Kyu Yeon Hur1, Jae Hyeon Kim1, Moon-Kyu Lee1, Sung-Min Park2 , Sang-Man Jin1,4
, Sang-Man Jin1,4
-
Diabetes & Metabolism Journal 2020;44(1):56-66.
DOI: https://doi.org/10.4093/dmj.2018.0227
Published online: October 21, 2019
1Division of Endocrinology and Metabolism, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
2Department of Creative IT Engineering, Pohang University of Science and Technology (POSTECH), Pohang, Korea.
3Department of Emergency Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
4Department of Digital Health, Samsung Advanced Institute for Health Sciences & Technology, Sungkyunkwan University, Seoul, Korea.
- Corresponding author: Sung-Min Park. Department of Creative IT Engineering, Pohang University of Science and Technology (POSTECH), 77 Cheongam-ro, Nam-gu, Pohang 37673, Korea. sungminpark@postech.ac.kr
- Corresponding author: Sang-Man Jin. Division of Endocrinology and Metabolism, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 06351, Korea. sangman.jin@samsung.com
- *Sung Woon Park and Seunghyun Lee contributed equally to this study as first authors.
Copyright © 2020 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- We aimed to describe the outcome of a computerized intravenous insulin infusion (CII) protocol integrated to the electronic health record (EHR) system and to improve the CII protocol in silico using the EHR-based predictors of the outcome.
-
Methods
- Clinical outcomes of the patients who underwent the CII protocol between July 2016 and February 2017 and their matched controls were evaluated. In the CII protocol group (n=91), multivariable binary logistic regression analysis models were used to determine the independent associates with a delayed response (taking ≥6.0 hours for entering a glucose range of 70 to 180 mg/dL). The CII protocol was adjusted in silico according to the EHR-based parameters obtained in the first 3 hours of CII.
-
Results
- Use of the CII protocol was associated with fewer subjects with hypoglycemia alert values (P=0.003), earlier (P=0.002), and more stable (P=0.017) achievement of a glucose range of 70 to 180 mg/dL. Initial glucose level (P=0.001), change in glucose during the first 2 hours (P=0.026), and change in insulin infusion rate during the first 3 hours (P=0.029) were independently associated with delayed responses. Increasing the insulin infusion rate temporarily according to these parameters in silico significantly reduced delayed responses (P<0.0001) without hypoglycemia, especially in refractory patients.
-
Conclusion
- Our CII protocol enabled faster and more stable glycemic control than conventional care with minimized risk of hypoglycemia. An EHR-based adjustment was simulated to reduce delayed responses without increased incidence of hypoglycemia.
- Hyperglycemia is associated with poor prognosis and increased mortality in critically ill patients [123] and is also a strong risk factor for surgical site infection [4]. In 2001, van den Berghe et al. [5] reported that tight glycemic control with intravenous insulin therapy in critically ill patients in a surgical intensive care unit (ICU) significantly lowered mortality. However, several multicenter randomized controlled studies failed to reproduce the decreased mortality with tight glycemic target (80 to 110 mg/dL) findings [678]. Consequentially, the American Association of Clinical Endocrinologists and the American Diabetes Association currently recommend a targeting range of 140 to 180 mg/dL in both critically and non-critically ill patients [9].
- At least in part, failure of intensive insulin therapy to reduce mortality in critically ill patients has been attributed to resultant increase in hypoglycemia [1011]. In a previous study [11], which evaluated 3,116 patients on IV insulin, almost one-third of the patients had suffered at least one episode of acute hypoglycemia (glucose level <60 mg/dL), and over 8% had severe acute hypoglycemia (glucose level <40 mg/dL). Although several studies of computerized intravenous insulin infusion protocols (CIIs) reported improved glycemic control and reduced hypoglycemia compared to paper-based protocols [1213141516], most of these studies either applied the CIIs only in the ICU rather than a general ward setting, or the software required frequent blood glucose measurement, sometimes requested in 30 minutes, making the protocol inappropriate in the general ward of most hospitals. More importantly, most of the target glucose range of these CII protocols were lower than the recommended glucose range of 140 to 180 mg/dL, failing to achieve near-elimination of hypoglycemia in most studies [1213141516].
- We developed a CII protocol fully integrated into the electronic health record (EHR) system for use in both the general ward and ICU, with a target glucose range of 140 to 180 mg/dL. This was fully integrated to the insulin prescription module in the EHR, eliminating the need for an additional record of glucose measurement and insulin dosing. The protocol was used in all hospital units, including general wards, ICUs, and emergency rooms. The purpose of this study was to describe the clinical outcome of our CII protocol and to adjust the CII protocol in silico using the factors independently associated with delayed target blood glucose achievement.
INTRODUCTION
- The protocol of this study was approved by the Institutional Review Board (IRB) of Samsung Medical Center (IRB file No. 2017-04-036-006). Informed consent was waived by the board.
- Study participants and setting
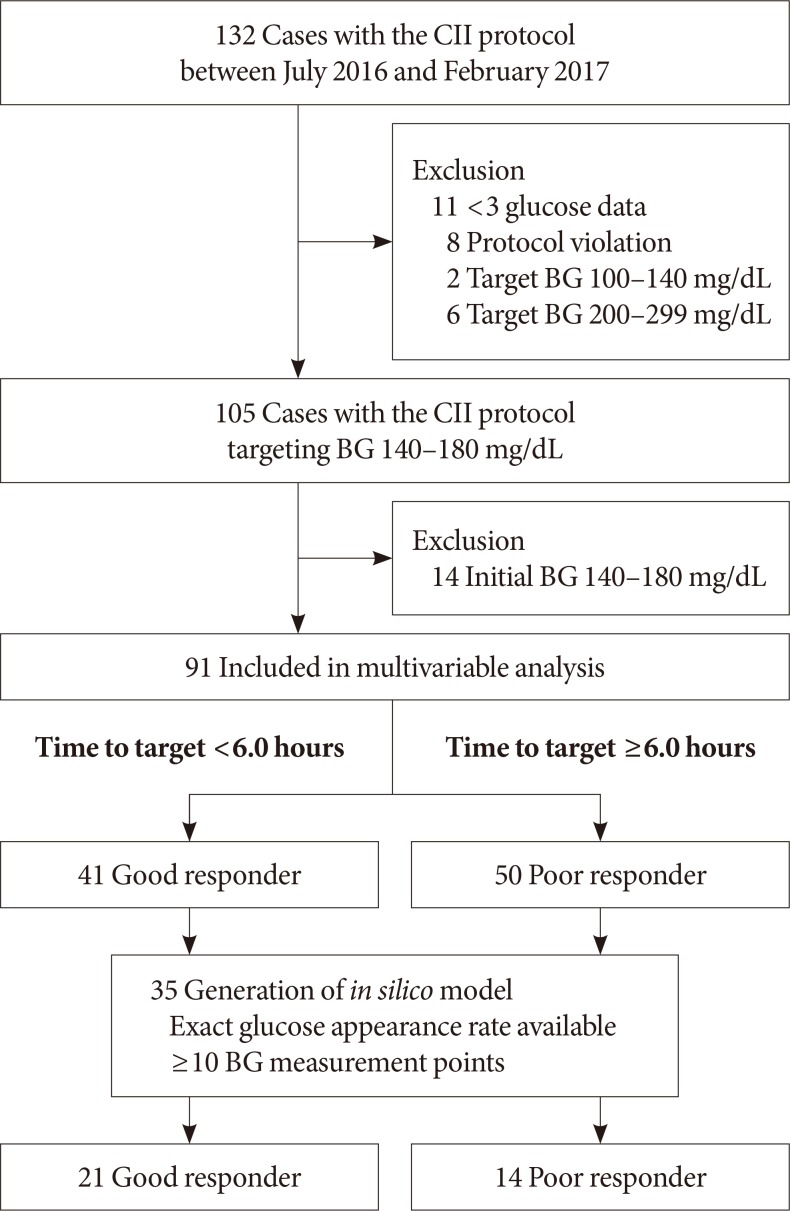
- The study was conducted in an academic tertiary care facility (Samsung Medical Center, Seoul, Korea) where a CII protocol was launched in June 2016. After launching the CII protocol, it had been advised to use in the patients with various potential indications for intravenous insulin therapy [17]. In perioperative cases, the CII was recommended when the patients underwent prolonged fasting for major surgery and had either glycosylated hemoglobin >8% or random glucose of >180 mg/dL at the day before operation. The CII protocol was applied at the initiation of fasting in perioperative period. We screened the 132 cases with the CII protocol between July 2016 and February 2017. Excluding the cases with less than three glucose measurements (n=11) and those with violation of the protocol (n=8), a total of 113 cases were included. Study participants in which the protocol targeting blood glucose was 100 to 140 mg/dL (n=2, patients who underwent cardiovascular surgery) and 200 to 299 (n=6, patients with hyperglycemic hyperosmolar state) were also excluded, leaving only those study participants who used the protocol targeting blood glucose range of 140 to 180 mg/dL (n=105) for inclusion in further analyses (Fig. 1).
- For each patient who used the protocol targeting blood glucose range of 140 to 180 mg/dL, a control patient was randomly selected by exact and propensity score matching. Controls were exactly matched for basal glucose level (mg/dL) category at the initiation of intravenous insulin infusion (<180, 180 to 249, ≥250 mg/dL), age group (10-year interval), reason for insulin infusion (perioperative, continuous enteral feeding, diabetic ketoacidosis [DKA], fasting for other reasons), and setting of admission (ICUs vs. general wards). Controls were also matched for the propensity score generated by other matching variables such as body mass index (<25 and ≥25 kg/m2), and estimated glomerular filtration rate (<60 and ≥60 mL/min/1.73 m2). Sixty controls were selected from the clinical database of 3,913 patients who underwent a conventional intravenous insulin infusion protocol, such as a modified version of Alberti's regimen, from January 2012 to June 2017 in Samsung Medical Center, Seoul, Korea.
- Among the 105 cases who used the CII protocol, 91 were studied in the multivariable analysis to identify the independent variables associated with delayed achievement of target blood glucose. The other 14 cases were excluded because of being within the target range from initiation of CII. The 91 cases were studied for glycemic outcomes including time required to achieve target blood glucose and percentage of time in target ranges. After determining the median time required to reach target blood glucose range using a Kaplan-Meier curve, these 91 cases were divided into good (less than median time) or poor (greater than or equal to median time) responder groups. The cases that failed to reach target range (n=7) were included in the poor responder group.
- CII protocol
- The CII protocol was fully integrated into an EHR system so that no additional manipulation other than insulin prescription was required. When nurses input a patient's current blood glucose level, the protocol automatically calculates insulin dose and time interval for next blood glucose check. The scheme for calculation of insulin dose adjustment was based on the Yale Insulin Infusion Protocol [18], but the target glucose range was adjusted to 140 to 180 mg/dL instead of 100 to 140 mg/dL (Supplementary Table 1). Blood glucose levels were checked with conventional fingerstick glucometers. The time interval for the next blood glucose check was set at 1 hour at onset of the CII protocol or when the previous blood glucose was not within the target range. If two consecutive blood glucoses are within the target range, the CII protocol instructs the user to increase the time interval for the next blood glucose check to 2 hours and then to 4 hours if an additional two blood glucoses are within the target range. If blood glucose is less than 70 mg/dL, the CII protocol orders immediate stoppage of insulin, initiation of 50% dextrose fluid infusion, and blood glucose recheck every 15 minutes until the blood glucose level recovers to 110 mg/dL. If the rechecked blood glucose rises above the target, the CII automatically calculates the reduced dosage of insulin to be infused.
- Identification of clinical and glycemic factors independently associated with time required to reach target blood glucose range
- We retrospectively reviewed clinical characteristics and glycemic outcomes of the cases with CII protocol and their controls. We extracted clinical information including age, sex, prior history of diabetes, and reason for instituting the insulin infusion protocol and selected laboratory results from the EHR system of Samsung Medical Center.
- Glycemic outcomes were compared between good and poor responder groups according to median time required to reach target blood glucose range among the 91 cases. We compared clinical characteristics and glycemic parameters between the good and poor responder groups and constructed a multivariable analysis model to identify the factors independently associated with delayed target achievement. Clinically significant hypoglycemia was defined as blood glucose less than 54 mg/dL, and the hypoglycemic alert value was defined as blood glucose less than 70 mg/dL. We considered a blood glucose level between 70 to 180 mg/dL, which has been used as a primary end-point for most clinical trials of artificial pancreas [19], as a clinically acceptable range of glucose levels. Calculated percentage of time in a glucose range of 70 to 180 mg/dL and time required to reach a glucose range of 70 to 180 mg/dL were analyzed.
- In silico evaluation
- To confirm whether the time to target blood glucose achievement can be reduced by temporarily increasing the insulin infusion rate, we developed an in silico patient group from the abstracted clinical CII data and simulated the outcome according to adjustment of insulin infusion rate in in silico patients. This adjustment was conducted in the presence of the factors found to be independently associated with delayed target achievement. Among the subjects included in the multivariable analysis, the in silico patients were developed from 35 cases. Excluded patients included those with an uncertain rate of glucose appearance such as patients in which continuous enteral feeding was applied or an evening meal was given right before initiation of the CII protocol so that uncertain glucose appearance through the gastrointestinal pathway might occur. Also excluded were patients in whom the number of blood glucose measurement points were less than 10, having non-positive degrees of freedom. The patients whose target blood glucose was not reached during the CII periods were also excluded.
- The virtual patients used Van Herpe's ICU minimal model (ICU-MM) [20] as a mathematical basis. For development, estimation was performed on nine glucose and insulin-related model parameters including basal glycemia and basal insulin. The ICU-MM was implemented in the Simbiology toolbox of MATLAB (R2018a; MathWorks, Natick, MA, USA) assuming the initial state to be basal. Initial parameter setting for optimization was set to the mean value of the ‘obese-low glucose tolerance patient group,’ as described in Bergman et al. [21]. Actual blood glucose values were used as initial search points of basal glycemic estimation.
- For simulation, the in silico patient model and CII protocols were implemented in Simulink (MATLAB R2018a). We categorized each factor found to be independently associated with delayed target achievement according to the cut-off values determined by Youden index in receiver operator characteristics (ROC) analysis. Additional insulin was infused when each parameter met the cut-off value. The insulin infusion rate was increased differentially according to the currently calculated insulin infusion rate (Supplementary Table 2).
- Statistical analysis
- Descriptive data are shown as mean±standard deviation or median with interquartile range. Student's t-test, Mann-Whitney test, chi-square test, or Fisher's exact test were used in univariable analysis as appropriate. Paired t-test, Wilcoxon signed rank test, or McNemar's test was used to compare the CII protocol cases and their matched controls as appropriate. Kaplan-Meier analysis was used to determine the median time required to reach the target blood glucose range. Multivariable binary logistic regression analysis (forward stepwise selection) was performed with the factors for which the P value was less than 0.2 in univariable analysis. Paired t-test was used to compare the time required to achieve a glucose range of 70 to 180 mg/dL before and after in silico adjustment of insulin infusion rate. Statistical analyses were performed with SPSS version 25 (IBM Co., Armonk, NY, USA) and R 3.4.3 (R Foundation for Statistical Computing, Vienna, Austria).
METHODS
- Clinical characteristics of and glycemic outcomes in each subject group
- The baseline characteristics of all the subjects who used the CII protocol targeting a blood glucose range of 140 to 180 mg/dL (n=105) are shown in Table 1. ICU patients accounted for 40.9% of cases with the CII protocol. The most common reason for use of the CII protocol was fasting in the perioperative period. In the 105 total patients, two cases with hypoglycemic alert value (glucose level <70 mg/dL) were reported. One case was due to sudden discontinuation of enteral feeding without adjustment of insulin infusion rate. The other case showed a blood glucose level of 69 mg/dL and successfully recovered to the target blood glucose range in 23 minutes with an automated algorithm for hypoglycemia in the CII protocol. No clinically significant hypoglycemia (glucose level <54 mg/dL) was reported in these patients. All of the 10 subjects who used the CII protocol for managing DKA were fully recovered from acidemia and met the criteria for transition to subcutaneous insulin therapy (glucose <200 mg/dL and two of the following; serum bicarbonate ≥15 mEq/L, venous pH >7.3, and anion gap ≤12 mEq/L) at the end of the CII protocol.
- Among the 105 subjects who used the CII protocol, 60 patients whose matched controls were available were included in the case-control analysis (Supplementary Table 3). Use of the CII protocol was associated with fewer subjects with hypoglycemia alert values (1 case vs. 12 cases in cases and controls, P=0.003), earlier achievement of a glucose range of 70 to 180 mg/dL (6.0 hours vs. 10.0 hours, P=0.002), and greater percentage of time in a glucose range of 70 to 180 mg/dL in overall (62.72±27.19 vs. 52.57±25.67, P=0.017), and greater percentage of time in a glucose range of 70 to 180 mg/dL after entering a glucose range of 70 to 180 mg/dL (81.25±21.70 vs. 68.09±25.78, P=0.002) (Supplementary Table 4). No patients in the cases, but two patients in controls, had clinically significant hypoglycemia (glucose level <54 mg/dL).
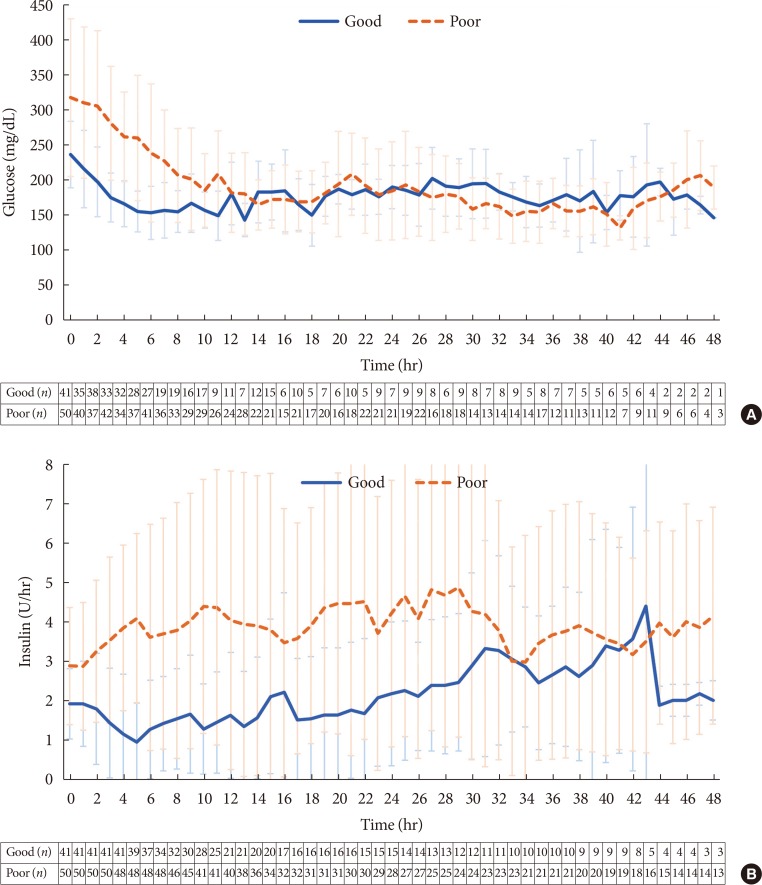
- In the 105 subjects who used the CII protocol, the median time required to achieve a glucose range of 70 to 180 mg/dL was 6.0 hours. The cases that took less than 6.0 hours were classified as a good responder group, and the cases that took 6.0 or more hours or those could not reach target range were classified as a poor responder group. The clinical characteristics of the two groups are compared in Table 2. The poor responders included more ICU patients (56.0% vs. 31.7%, P=0.02) and more patients with no prior treatment or newly diagnosed diabetes (46% vs. 19.5%, P=0.005). Although the good responders showed higher percentage of time in a glucose range of 70 to 180 mg/dL than poor responder group in overall (64.09±23.41 vs. 38.77±23.54, P=0.000), the % time in a glucose range of 70 to 180 mg/dL was not significantly different between groups after achieving a glucose range of 70 to 180 mg/dL (Supplementary Table 5). Although mean glucose levels were similar between groups after achieving a glucose range of 70 to 180 mg/dL, glucose levels of good responder group decreased rapidly in first few hours while those of poor responder group decreased slowly in first 2 hours with higher baseline glucose levels. The insulin requirement of poor responder group continuously increased during the first 5 hours, whereas the insulin requirement decreased in good responder group (Fig. 2).
- Factors independently associated with poor responder group
- To find factors independently associated with poor responder group, we constructed multivariable analysis models including clinical and glycemic parameters as covariates. In multivariable analysis model 1, the factors that could be determined at the initiation of the protocol were included. The covariates included the initial blood glucose level and other clinical factors such as age, sex, ICU or non-ICU patients, prior treatment for diabetes, and reason for insulin therapy. Among the covariates, initial blood glucose level and prior treatment for diabetes were selected by forward stepwise variable selection (Table 3). In multivariable model 2, we added the glycemic parameters that can be obtained within 3 hours after initiation of the CII protocol. The final model included initial blood glucose (odds ratio [OR], 1.027; 95% confidence interval [CI], 1.011 to 1.043), blood glucose change in first 2 hours (OR, 1.019; 95% CI, 1.002 to 1.036), and change in insulin infusion rate in first 3 hours (OR, 1.747; 95% CI, 1.058 to 2.885) (Table 3).
- In silico improvement of the CII protocol by automatic adjustments
- Based on the cut-off values determined by ROC analysis, additional insulin infusion was applied when any of the following conditions was met: (1) initial blood glucose was greater than or equal to 233 mg/dL; (2) decrease in blood glucose was less than or equal to 6 mg/dL at 2 hours; or (3) decrease in insulin infusion rate at 3 hours was less than or equal to 0.5 U/hr. Simulation was performed in the 35 in silico patients. Before additional insulin infusion was performed, 21 and 14 subjects were in the good and poor responder groups, respectively.
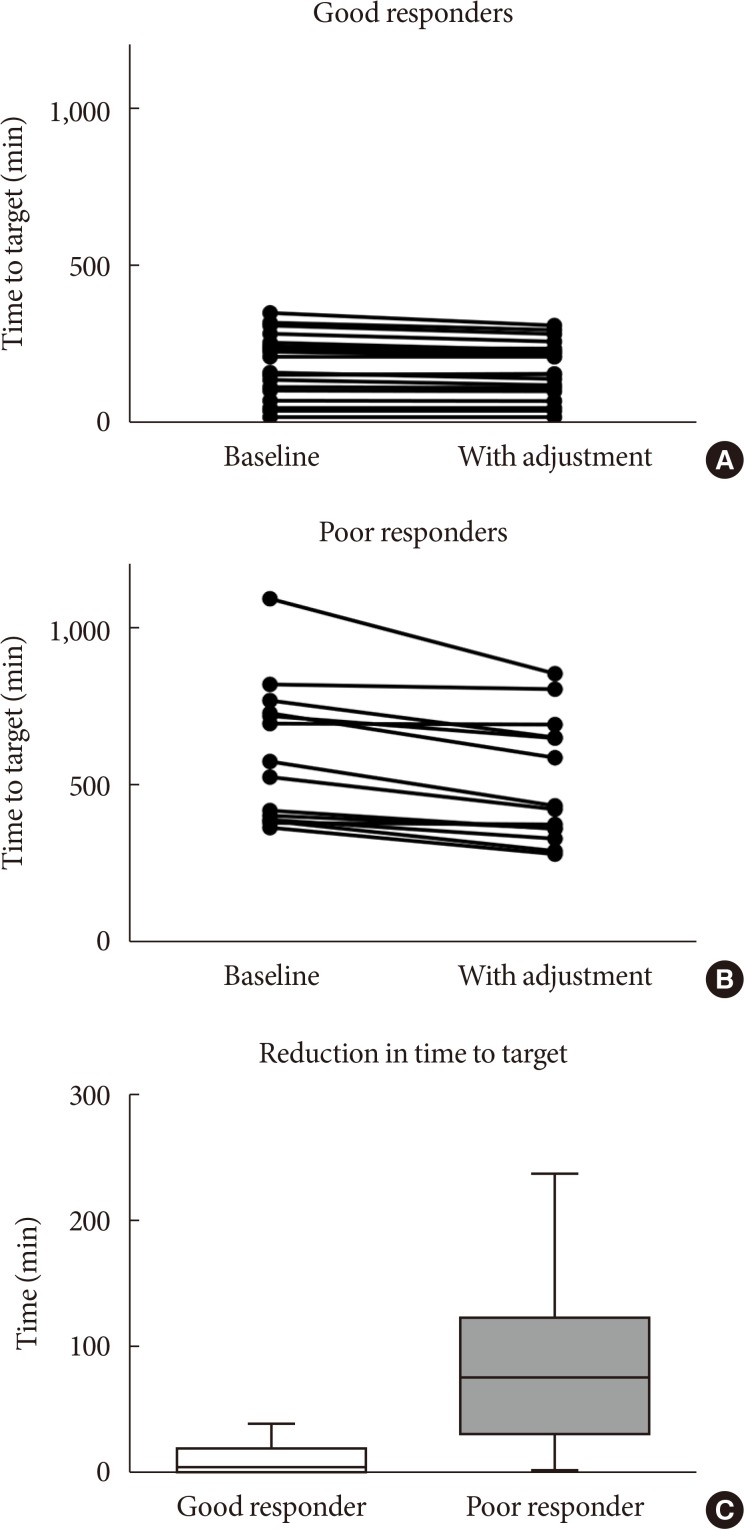
- Additional insulin infusion at the early glycemic predicting points resulted in a significant reduction in median time required to achieve a glucose range of 70 to 180 mg/dL (286 to 261 minutes) overall (P<0.0001 for paired t-test). The median time required to achieve a glucose range of 70 to 180 mg/dL was changed from 213 minutes (range, 22 to 352 minutes) to 213 minutes (range, 22 to 312 minutes) in the good responder group (P=0.0004 for paired t-test), and from 549.5 minutes (range, 365 to 1,089 minutes) to 428.5 minutes (range, 281 to 852 minutes) in the poor responder group (P=0.0003 for paired t-test), with a significantly greater reduction in time to achieve a glucose range of 70 to 180 mg/dL in poor responder group (P=0.0005). This resulted in reclassification of three subjects into the good responder group (Fig. 3). In this simulation, additional insulin infusion did not cause any hypoglycemic events (blood glucose <70 mg/dL).
RESULTS
- This study describes the clinical outcome of a fully EHR-integrated CII protocol that enabled faster and more stable glycemic control than conventional care with minimized risk of hypoglycemia. The convenience achieved by full integration to EHR allows application of this CII protocol in both critical and non-critical care settings. Furthermore, an automatic adjustment taking advantage of the fully EHR-integrated feature significantly improved the time required to achieve target glucose level in our in silico simulation without increased incidence of hypoglycemia.
- The low proportions of patients with hypoglycemia alert value (two of 105 patients) and absence of clinically significant hypoglycemia in our study population are distinguishing aspects of our CII protocol from previous CII protocol reports. Subsequent case-control analysis (n=60) also showed fewer proportion of the subjects with hypoglycemia alert values, and better quality of glycemic control in terms of earlier achievement of euglycemia and greater percent time in a glucose range of 70–180 mg/dL in the subjects who used the CII protocol. Several previous studies have demonstrated lower mean glucose level and fewer hypoglycemic patients managed by CII protocols than those of paper-based protocol controls [12141516182223]. Recently, Marvin et al. [24] compared the outcome of several CII protocols having different target blood glucose ranges of 100 to 139, 120 to 140, and 140 mg/dL as a fixed threshold. The proportion of protocol use period with hypoglycemia alert values (glucose <70 mg/dL) was 17.2%, 7.56%, and 5.8%, respectively. After their protocol was modified to include the addition of mid-protocol bolus insulin, the proportion was reduced to 0.095% [24]. However, the modified protocol required additional workload including hourly blood glucose checks and additional bolus doses of insulin, which are difficult to institute in settings outside of the ICU [24]. The CII protocol in our study adopted a higher target blood glucose range (140 to 180 mg/dL) than the previous studies, while allowing blood glucose check intervals no less than 1 hour and up to 2 to 4 hours after blood glucose stabilization. Therefore, caregiver workload was substantially reduced after entering euglycemia, and specialized equipment or continuous glucose monitoring was not required. Although 40.9% of our study population was in critical care setting, clinically significant hypoglycemia was absent even in ICU patients, in contrast to the findings of the previous studies conducted in ICU showing higher rate of hypoglycemia [12141516182223] but consistent with a recent preliminary report of a bedside computerized decision-support tool targeting the blood glucose range of 140 to 180 mg/dL in mixed ICU [25]. Although elimination of perioperative hypoglycemia has been also achieved with artificial pancreas systems even with lower blood glucose target [26272829], use of these systems in critical care settings with decreased blood perfusion would require specialized equipment and continuous peripheral blood sampling.
- In this study, the median time required to enter a blood glucose range of 70 to 180 mg/dL was 6 hours. Because entering euglycemia in 6 hours might secure 12 to 18 hours duration of euglycemia before operation if the patients were admitted the day before the operation, we grouped the cases with the CII protocol into good and poor responders according to whether the time required to enter a blood glucose range of 70 to 180 mg/dL was less than 6 hours. We also found that the most distinct differences of changes in glucose levels and insulin requirement over time between good and poor responders were obtained during the first 2 to 3 hours, but it became stable after the glucose levels entered the range of 70 to 180 mg/dL except in only a few patients with exceptionally long duration of using the CII protocol. Therefore, we focused on reducing the time required for achievement of a glucose range of 70 to 180 mg/dL through automatic adjustment based on the information gathered in the first 2 to 3 hours of CII. To improve our CII protocol, we started with identifying several clinical and glycemic factors related to delayed achievement of target glucose range through multivariable analyses. Among the covariates available at initiation of the CII, ‘no prior treatment for diabetes’ was an independent risk factor for the poor response. This is in line with the previous reports that inpatient hyperglycemia without prior diagnosis of diabetes, commonly from the stress and highly catabolic status, is associated with the highest risk for perioperative complications [30]. When we constructed Model 2 in this study by adding the covariates from glycemic factors during the first 3 hours of CII to the clinical covariates, all of the chosen covariates were glycemic factors rather than the clinical factors.
- Indeed, in our in silico simulation, we found that temporarily increasing insulin infusion rate based on the glycemic factors found in Model 2 shortened the time required to achieve a glucose range of 70 to 180 mg/dL. More prominent reduction in the time to achievement of a glucose range of 70 to 180 mg/dL was observed in the poor responder group. Importantly, such an improved outcome was achieved without causing hypoglycemia in the CII simulation. These results indicate that delayed achievement of target blood glucose in this CII protocol, presumably due to the relatively high target blood glucose range, could be individually prevented. In clinical settings where a CII protocol is available and adjustable, prevention could be achieved by applying a simple calculation based on the variables available in the first 3 hours of the CII. Alternatively, when CII protocols are not available, it could be practically suggested that increasing insulin infusion rate when the patients have a high glucose level (greater than or equal to 233 mg/dL) along with slow progress in lowering glucose levels and insulin requirement could be an option for rapid glucose stabilization.
- We recognize limitations in this study. This was a single center study with a limited number of cases because the CII protocol was recently launched; a limited number of patients had sufficient numbers of blood glucose measurements required for generation of an in silico model. Further study with a large number of qualified in silico patients and an actual clinical trial will enable development of a more optimized CII protocol for generalized use.
- In conclusion, our EHR-integrated CII protocol successfully maintained target glucose level with minimized risk of hypoglycemia. We identified parameters available during the first 3 hours of CII that were independently associated with delayed target achievement and used these parameters in this CII to shorten the time required for target blood glucose achievement in our in silico patients.
DISCUSSION
-
Acknowledgements
- We thank the Department of Bio Statistics, Samsung Medical Center for assistance with statistical analysis including exact and propensity score matching. This research was supported by the MSIT (Ministry of Science and ICT), Korea, under the ICT Consilience Creative program (IITP-2019-2011-1-00783) supervised by the IITP (Institute for Information & communications Technology Planning & Evaluation). This research was also supported by the Technology Innovation Program (or Industrial Strategic Technology Development Program (20001841; Development of System for Intelligent ContextAware Wearable Service based on Machine Learning) funded by the Ministry of Trade, Industry & Energy (MOTIE, Sejong, Korea).
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS:
Conception or design: S.M.P., S.M.J.
Acquisition, analysis, or interpretation of data: S.W.P., S.L., S.M.P., S.M.J.
Critical review of the manuscript: W.C.C., K.Y.H., J.H.K., M.K.L., S.M.P., S.M.J.
Drafting the work or revising: S.W.P., S.L., S.M.P., S.M.J.
Final approval of the manuscript: S.W.P., S.L., W.C.C., K.Y.H., J.H.K., M.K.L., S.M.P., S.M.J.
NOTES
SUPPLEMENTARY MATERIALS
Supplementary Table 2
Supplementary Table 3
Supplementary Table 4
Supplementary Table 5
- 1. Falciglia M, Freyberg RW, Almenoff PL, D'Alessio DA, Render ML. Hyperglycemia-related mortality in critically ill patients varies with admission diagnosis. Crit Care Med 2009;37:3001-3009. ArticlePubMedPMC
- 2. Norhammar AM, Ryden L, Malmberg K. Admission plasma glucose. Independent risk factor for long-term prognosis after myocardial infarction even in nondiabetic patients. Diabetes Care 1999;22:1827-1831. ArticlePubMedPDF
- 3. Yendamuri S, Fulda GJ, Tinkoff GH. Admission hyperglycemia as a prognostic indicator in trauma. J Trauma 2003;55:33-38. ArticlePubMed
- 4. Golden SH, Peart-Vigilance C, Kao WH, Brancati FL. Perioperative glycemic control and the risk of infectious complications in a cohort of adults with diabetes. Diabetes Care 1999;22:1408-1414. ArticlePubMedPDF
- 5. van den Berghe G, Wouters P, Weekers F, Verwaest C, Bruyninckx F, Schetz M, Vlasselaers D, Ferdinande P, Lauwers P, Bouillon R. Intensive insulin therapy in critically ill patients. N Engl J Med 2001;345:1359-1367. ArticlePubMed
- 6. Brunkhorst FM, Engel C, Bloos F, Meier-Hellmann A, Ragaller M, Weiler N, Moerer O, Gruendling M, Oppert M, Grond S, Olthoff D, Jaschinski U, John S, Rossaint R, Welte T, Schaefer M, Kern P, Kuhnt E, Kiehntopf M, Hartog C, Natanson C, Loeffler M, Reinhart K. German Competence Network Sepsis (SepNet). Intensive insulin therapy and pentastarch resuscitation in severe sepsis. N Engl J Med 2008;358:125-139. ArticlePubMed
- 7. NICE-SUGAR Study Investigators. Finfer S, Chittock DR, Su SY, Blair D, Foster D, Dhingra V, Bellomo R, Cook D, Dodek P, Henderson WR, Hebert PC, Heritier S, Heyland DK, McArthur C, McDonald E, Mitchell I, Myburgh JA, Norton R, Potter J, Robinson BG, Ronco JJ. Intensive versus conventional glucose control in critically ill patients. N Engl J Med 2009;360:1283-1297. ArticlePubMed
- 8. Preiser JC, Devos P, Ruiz-Santana S, Melot C, Annane D, Groeneveld J, Iapichino G, Leverve X, Nitenberg G, Singer P, Wernerman J, Joannidis M, Stecher A, Chiolero R. A prospective randomised multi-centre controlled trial on tight glucose control by intensive insulin therapy in adult intensive care units: the Glucontrol study. Intensive Care Med 2009;35:1738-1748. ArticlePubMedPDF
- 9. Moghissi ES, Korytkowski MT, DiNardo M, Einhorn D, Hellman R, Hirsch IB, Inzucchi SE, Ismail-Beigi F, Kirkman MS, Umpierrez GE. American Association of Clinical Endocrinologists. American Diabetes Association. American Association of Clinical Endocrinologists and American Diabetes Association consensus statement on inpatient glycemic control. Endocr Pract 2009;15:353-369. ArticlePubMedPDF
- 10. Wilson M, Weinreb J, Hoo GW. Intensive insulin therapy in critical care: a review of 12 protocols. Diabetes Care 2007;30:1005-1011. PubMed
- 11. Zhang Y. Predicting occurrences of acute hypoglycemia during insulin therapy in the intensive care unit. Conf Proc IEEE Eng Med Biol Soc 2008;2008:3297-3300. Article
- 12. Boord JB, Sharifi M, Greevy RA, Griffin MR, Lee VK, Webb TA, May ME, Waitman LR, May AK, Miller RA. Computer-based insulin infusion protocol improves glycemia control over manual protocol. J Am Med Inform Assoc 2007;14:278-287. ArticlePubMedPMC
- 13. Davidson PC, Steed RD, Bode BW. Glucommander: a computer-directed intravenous insulin system shown to be safe, simple, and effective in 120,618 h of operation. Diabetes Care 2005;28:2418-2423. PubMed
- 14. Dortch MJ, Mowery NT, Ozdas A, Dossett L, Cao H, Collier B, Holder G, Miller RA, May AK. A computerized insulin infusion titration protocol improves glucose control with less hypoglycemia compared to a manual titration protocol in a trauma intensive care unit. JPEN J Parenter Enteral Nutr 2008;32:18-27. ArticlePubMedPDF
- 15. Newton CA, Smiley D, Bode BW, Kitabchi AE, Davidson PC, Jacobs S, Steed RD, Stentz F, Peng L, Mulligan P, Freire AX, Temponi A, Umpierrez GE. A comparison study of continuous insulin infusion protocols in the medical intensive care unit: computer-guided vs. standard column-based algorithms. J Hosp Med 2010;5:432-437. ArticlePubMedPDF
- 16. Saur NM, Kongable GL, Holewinski S, O'Brien K, Nasraway SA Jr. Software-guided insulin dosing: tight glycemic control and decreased glycemic derangements in critically ill patients. Mayo Clin Proc 2013;88:920-929. ArticlePubMed
- 17. Kelly JL. Continuous insulin infusion: when, where, and how? Diabetes Spectr 2014;27:218-223. ArticlePubMedPMCPDF
- 18. Marvin MR, Inzucchi SE, Besterman BJ. Computerization of the Yale insulin infusion protocol and potential insights into causes of hypoglycemia with intravenous insulin. Diabetes Technol Ther 2013;15:246-252. ArticlePubMedPMC
- 19. Maahs DM, Buckingham BA, Castle JR, Cinar A, Damiano ER, Dassau E, DeVries JH, Doyle FJ 3rd, Griffen SC, Haidar A, Heinemann L, Hovorka R, Jones TW, Kollman C, Kovatchev B, Levy BL, Nimri R, O'Neal DN, Philip M, Renard E, Russell SJ, Weinzimer SA, Zisser H, Lum JW. Outcome measures for artificial pancreas clinical trials: a consensus report. Diabetes Care 2016;39:1175-1179. ArticlePubMedPMCPDF
- 20. Van Herpe T, Pluymers B, Espinoza M, Van den Berghe G, De Moor B. A minimal model for glycemia control in critically ill patients. Conf Proc IEEE Eng Med Biol Soc 2006;1:5432-5435.
- 21. Bergman RN, Phillips LS, Cobelli C. Physiologic evaluation of factors controlling glucose tolerance in man: measurement of insulin sensitivity and beta-cell glucose sensitivity from the response to intravenous glucose. J Clin Invest 1981;68:1456-1467. ArticlePubMedPMC
- 22. Juneja R, Roudebush CP, Nasraway SA, Golas AA, Jacobi J, Carroll J, Nelson D, Abad VJ, Flanders SJ. Computerized intensive insulin dosing can mitigate hypoglycemia and achieve tight glycemic control when glucose measurement is performed frequently and on time. Crit Care 2009;13:R163. ArticlePubMedPMC
- 23. Dumont C, Bourguignon C. Effect of a computerized insulin dose calculator on the process of glycemic control. Am J Crit Care 2012;21:106-115. ArticlePubMedPDF
- 24. Marvin MR, Inzucchi SE, Besterman BJ. Minimization of hypoglycemia as an adverse event during insulin infusion: further refinement of the Yale protocol. Diabetes Technol Ther 2016;18:480-486. ArticlePubMedPMC
- 25. Telford ED, Franck AJ, Hendrickson AL, Dietrich NM. A bedside computerized decision-support tool for intravenous insulin infusion management in critically ill patients. Jt Comm J Qual Patient Saf 2018;44:299-303. ArticlePubMed
- 26. Hanazaki K, Kitagawa H, Yatabe T, Munekage M, Dabanaka K, Takezaki Y, Tsukamoto Y, Asano T, Kinoshita Y, Namikawa T. Perioperative intensive insulin therapy using an artificial endocrine pancreas with closed-loop glycemic control system: the effects of no hypoglycemia. Am J Surg 2014;207:935-941. ArticlePubMed
- 27. Ichimori S, Nishida K, Shimoda S, Sekigami T, Matsuo Y, Ichinose K, Shichiri M, Sakakida M, Araki E. Development of a highly responsive needle-type glucose sensor using polyimide for a wearable artificial endocrine pancreas. J Artif Organs 2006;9:105-113. ArticlePubMedPDF
- 28. Matsuo Y, Shimoda S, Sakakida M, Nishida K, Sekigami T, Ichimori S, Ichinose K, Shichiri M, Araki E. Strict glycemic control in diabetic dogs with closed-loop intraperitoneal insulin infusion algorithm designed for an artificial endocrine pancreas. J Artif Organs 2003;6:55-63. ArticlePubMedPDF
- 29. Namikawa T, Munekage M, Kitagawa H, Yatabe T, Maeda H, Tsukamoto Y, Hirano K, Asano T, Kinoshita Y, Hanazaki K. Comparison between a novel and conventional artificial pancreas for perioperative glycemic control using a closed-loop system. J Artif Organs 2017;20:84-90. ArticlePubMedPDF
- 30. Kwon S, Thompson R, Dellinger P, Yanez D, Farrohki E, Flum D. Importance of perioperative glycemic control in general surgery: a report from the Surgical Care and Outcomes Assessment Program. Ann Surg 2013;257:8-14. ArticlePubMed
REFERENCES
The mean glucose levels and insulin doses in good and poor responder groups of the cases using the computerized intravenous insulin infusion protocol during the 48 hours of intravenous insulin infusion. (A) Mean glucose level versus time mapped hourly for the initial 48 hours. (B) Insulin doses versus time mapped hourly for the initial 48 hours. Error bars indicate interquartile range. Numbers below each panel indicate the number of subjects for each time point. In panel B, the insulin infusion rates of good responders include data of a case in which insulin infusion at a rate of 8 to 10 U/hr was abruptly stopped at 44th hour.
Distribution of the time required to reach the target ranges with the computerized intravenous insulin infusion (CII) protocol and additional insulin infusion at early glycemic predicting points. (A) Good responder group. (B) Poor responder group. (C) Reduction in time required to reach a glucose range of 70 to 180 mg/dL. Box and whisker plots indicate median, interquartile range, and range.

Baseline characteristics of all the cases of the computerized intravenous insulin infusion protocol targeting blood glucose 140 to 180 mg/dL (n=105)
Values are presented as mean±standard deviation, number (%), or median (interquartile range).
BMI, body mass index; ICU, intensive care unit; eGFR, estimated glomerular filtration rate; IV, intravenous; NPO, nil per os; HbA1c, glycosylated hemoglobin.
aGastrointestinal bleeding (n=7), uncontrolled hyperglycemia (n=7), acute pancreatitis (n=2), intubation state (n=2), aspiration pneumonia (n=2), severe sepsis or septic shock (n=2), altered mentality (n=3), acute cholecystitis (n=1), diabetes gastropathy (n=1), peritonsilar abscess (n=1), for esophagogastroduodenoscopy (n=1), after total pancreatectomy (n=1).
Clinical characteristics of the good and poor responders in cases using the computerized insulin infusion protocol according to time required to achieve a glucose range of 70 to 180 mg/dL
Factors independently associated with delayed achievement of a glucose range of 70 to 180 mg/dL
Figure & Data
References
Citations

- Response: An Electronic Health Record-Integrated Computerized Intravenous Insulin Infusion Protocol: Clinical Outcomes and in Silico Adjustment (Diabetes Metab J 2020;44:56–66)
Sung Woon Park, Seunghyun Lee, Won Chul Cha, Kyu Yeon Hur, Jae Hyeon Kim, Moon-Kyu Lee, Sung-Min Park, Sang-Man Jin
Diabetes & Metabolism Journal.2020; 44(2): 358. CrossRef - Letter: An Electronic Health Record-Integrated Computerized Intravenous Insulin Infusion Protocol: Clinical Outcomes and in Silico Adjustment (Diabetes Metab J 2020;44:56–66)
Dongwon Yi
Diabetes & Metabolism Journal.2020; 44(2): 354. CrossRef

 KDA
KDA
 PubReader
PubReader Cite
Cite