- Current
- Browse
- Collections
-
For contributors
- For Authors
- Instructions to authors
- Article processing charge
- e-submission
- For Reviewers
- Instructions for reviewers
- How to become a reviewer
- Best reviewers
- For Readers
- Readership
- Subscription
- Permission guidelines
- About
- Editorial policy
Articles
- Page Path
- HOME > Diabetes Metab J > Volume 43(1); 2019 > Article
-
Original ArticleClinical Diabetes and Therapeutics Hospital-Based Korean Diabetes Prevention Study: A Prospective, Multi-Center, Randomized, Open-Label Controlled Study
-
Sang Youl Rhee, Suk Chon, Kyu Jeung Ahn, Jeong-Taek Woo
 , the Korean Diabetes Prevention Study Investigators
, the Korean Diabetes Prevention Study Investigators -
Diabetes & Metabolism Journal 2019;43(1):49-58.
DOI: https://doi.org/10.4093/dmj.2018.0033
Published online: November 2, 2018
Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, Seoul, Korea.
- Corresponding author: Jeong-Taek Woo. Department of Endocrinology and Metabolism, Kyung Hee University School of Medicine, 23 Kyungheedae-ro, Dongdaemun-gu, Seoul 02447, Korea. jtwoomd@khmc.or.kr
• Received: February 18, 2018 • Accepted: June 3, 2018
Copyright © 2018 Korean Diabetes Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- The prevalence of diabetes mellitus (DM) continues to increase, and the disease burden is the highest of any medical condition in Korea. However, large-scale clinical studies have not yet conducted to establish the basis for diabetes prevention in Korea.
-
Methods
- The hospital-based Korean Diabetes Prevention Study (H-KDPS) is a prospective, multi-center, randomized, open-label controlled study conducted at university hospitals for the purpose of gathering data to help in efforts to prevent type 2 DM. Ten university hospitals are participating, and 744 subjects will be recruited. The subjects are randomly assigned to the standard care group, lifestyle modification group, or metformin group, and their clinical course will be observed for 36 months.
-
Results
- All intervention methodologies were developed, validated, and approved by Korean Diabetes Association (KDA) multi-disciplinary team members. The standard control group will engage in individual education based on the current KDA guidelines, and the lifestyle modification group will participate in a professionally guided healthcare intervention aiming for ≥5% weight loss. The metformin group will begin dosing at 250 mg/day, increasing to a maximum of 1,000 mg/day. The primary endpoint of this study is the cumulative incidence of DM during the 3 years after randomization.
-
Conclusion
- The H-KDPS study is the first large-scale clinical study to establish evidence-based interventions for the prevention of type 2 DM in Koreans. The evidence gathered by this study will be useful for enhancing the health of Koreans and improving the stability of the Korean healthcare system (Trial registration: CRIS KCT0002260, NCT02981121).
- The prevalence of diabetes is rapidly increasing worldwide, and its prevalence in Korea has increased from 1.5% of adults aged 30 or older in 1971 to 11.1% of adults in 2013. It is estimated that it will continue to increase, reaching approximately 4.3 million by 2030 [12]. The rapid increase in diabetes patients has exacerbated the burden on global public health systems [34].
- Diabetes can be prevented through appropriate interventions. Clinical studies conducted in Finland, the United States, and China have shown that diabetes can be prevented through lifestyle interventions or medications [567891011]. Such interventions not only prevent diabetes but may also have significant preventive effects on complications closely related to diabetes [1213141516].
- In response to these findings, some countries have recently attempted to introduce diabetes prevention interventions into the public healthcare system. In countries with a high prevalence of diabetes, such as Korea, a systematic strategy for diabetes prevention is urgently needed. However, to prevent diabetes, we need to develop a differentiated strategy that reflects each country's unique social and cultural characteristics [1718].
- Until recently, no randomized controlled clinical studies aimed at establishing an objective basis for diabetes prevention had been conducted in Korea. However, the project group for the Korean Diabetes Prevention Study (KDPS) was recently organized by various researchers from the Korean Diabetes Association (KDA) to conduct multi-center clinical studies for the prevention of diabetes. The results of this clinical study are expected to contribute to the establishment of a unique diabetes prevention strategy that reflects the characteristics of Koreans. This article introduces the protocol for the hospital-based Korean Diabetes Prevention Study (H-KDPS), which is being implemented as part of this Korean diabetes prevention project.
INTRODUCTION
- Goals of the KDPS
- The goals of KDPS are as follows: (1) to develop a diabetes prevention program (lifestyle intervention and medication) that is suitable for Koreans at high risk for diabetes; (2) to conduct a prospective, randomized, controlled study and establish a cost-effective intervention program; and (3) to contribute to the national diabetes prevention program and promote public health by disseminating the results of the study.
- KDA project group for the KDPS
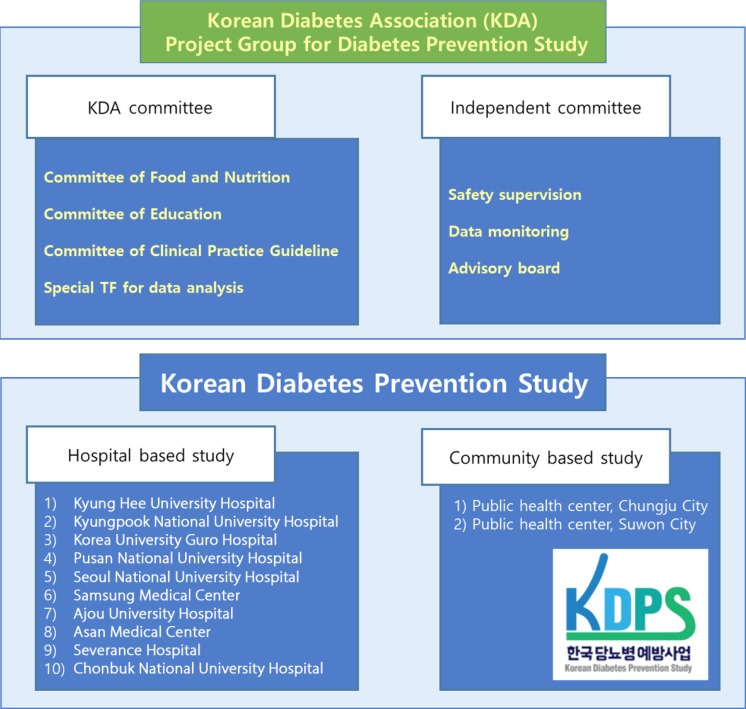
- The project group for the KDPS was organized in December 2015 to support and operate the KDPS (Fig. 1). In addition to the steering committee responsible for the actual research, KDA's multidisciplinary committee has supported the development of clinical research protocols. Committees independent of the steering committee were organized within the project team to perform data monitoring, subject safety management, and verification of results.
- H-KDPS study
- The KDPS is divided into two intervention studies: a hospital-based intervention study (H-KDPS) and a community web-based intervention study (community web-based Korean Diabetes Prevention Study [C-KDPS]). The H-KDPS is a hospital-based intervention study involving diabetes experts from 10 university hospitals. The study protocol of the H-KDPS was developed based on the recommendations of the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) statement on randomized trials [19]. The contents of this protocol have been reviewed and approved by the KDA board of directors. C-KDPS is a community-based intervention study, and related content has been published in another article [20].
- Study design
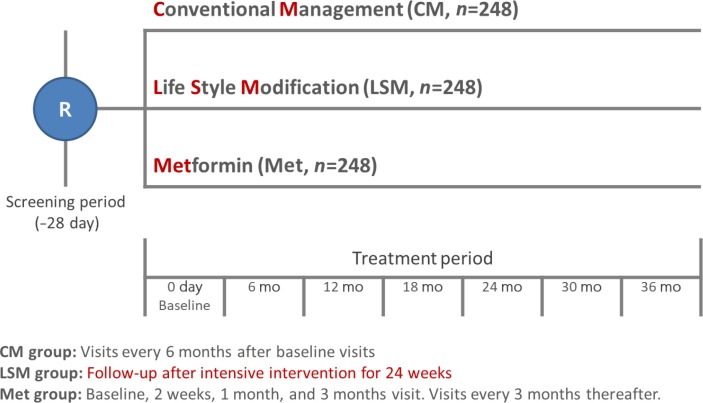
- The H-KDPS is a prospective, multi-center, randomized, open-label controlled study (Fig. 2). This clinical study was designed based on the following hypotheses: (1) a 5% weight loss through lifestyle interventions will prevent diabetes in participants at high risk for this condition; and (2) metformin will prevent diabetes in participants at high risk for this condition.
- Eligibility criteria
- The inclusion criteria of this study are as follows: (1) aged ≥30 and ≤70 years; (2) body mass index ≥23 kg/m2; and (3) at least one of the following conditions: a postprandial 2-hour glucose level of 140 to 199 mg/dL after a 75-g oral glucose tolerance test, a ≥8-hour fasting blood glucose level of 110 to 125 mg/dL, and a National Glycohemoglobin Standardization Program (NGSP) standardized glycosylated hemoglobin (HbA1c) level of 5.7% to 6.4%.
- The exclusion criteria of this study are as follows: (1) people with type 2 diabetes mellitus (DM); (2) people whose life expectancy is anticipated to shorten or whose risk of a shortened life span may increase due to the intervention; (3) people who are expected to influence the performance of clinical trials based on the researcher's evaluation; and (4) people taking medication or with a medical condition that may affect the diagnosis of diabetes.
- More detailed inclusion and exclusion criteria can be found at the clinical trial registration websites (https://clinicaltrials.gov/ct2/show/NCT02981121 and https://cris.nih.go.kr/cris/search/search_result_st01_en.jsp?seq=6414).
- Allocation and randomization
- An independent statistician prepared the randomization plan. The subjects will be divided into three groups and assigned 1:1:1 using stratified block randomization to reduce the predictability of assigned groups and to assign subjects according to the institution.
- Recruitment
- Subjects who satisfy the inclusion criteria were/will be recruited during health check-ups (national, workplace, or institutional health check-up program recipients) or during outpatient visits to the research institutions. We have been cooperating with various medical institutions and healthcare promotion centers for purposes of subject recruitment and will continue to do so (see Acknowledgments).
- Intervention
- The subjects will be randomly assigned to three treatment groups: conventional management (CM), lifestyle modification (LSM), and metformin (Met) treatment (Tables 1,2,3, Fig. 1). The total benefits received through participation in the research will be equal regardless of group assignment.
- The CM group will receive individual education based on the KDA clinical practice guidelines as the baseline for participation [21]. After that, subjects will attend follow-up visits every 6 months to monitor their clinical course.
- The LSM group will receives an intervention based on the protocol developed by the Food and Nutrition Committee of the KDA. The LSM protocol consists of moderate exercise for more than 150 minutes per week and meal coaching and monitoring by healthcare professionals. The goal is loss of more than 5% of body weight within 6 months and subsequent maintenance of the weight loss. In the first 6 months of the study, an intensive intervention consisting of seven offline training sessions and eight telephone training sessions will be conducted. After that, subjects will attend follow-up visits every 3 months to monitor their clinical course.
- During the course of the observation period, the subjects will be encouraged to practice 10 basic living rules (“10 habits'”). The adherence of subjects is assessed using the session participation rate and the practice rate of “10 habits” using self-diary. And, even if subjects cannot achieve the weight loss target of 5% or more, we plan to observe the clinical course continuously without dropping. The details of the LSM protocol will be published separately. A digital activity tracker (ZIKTO Walk; ZIKTO Inc., Seoul, Korea) will be provided to all LSM subjects to evaluate the effects of the lifestyle intervention. However, the user will be able to arbitrarily select whether to use it continuously. We plan to assess whether voluntary use of an activity tracker has a significant effect on the desired endpoint.
- The Met group will take metformin according to the protocol approved by the KDA Clinical Practice Guidelines Committee. After randomization, subjects will take 250 mg/day metformin (Dalim Corporation, Seoul, Korea) for 2 weeks. If there are no side effects, the dose will be increased to 500 mg/day for an additional 2 weeks. The dose will be increased to up to 1,000 mg/day if there are no side effects. Dosage reduction will depend on the subject's symptoms and adherence. After that, subjects will attend follow-up visits every 3 months to monitor their clinical course.
- Ethics
- The H-KDPS protocol was approved by the Institutional Review Boards at Kyung Hee University Hospital (KHUH 2016-04-102-028), Kyungpook National University Hospital (KNUH 2016-07-002-015), Korea University Guro Hospital (KUGH16114-013), Pusan National University Hospital (D-1607-001-054), Samsung Medical Center (SMC 2016-06-006-014), Seoul National University Hospital (H-1606-110-771), Asan Medical Center (S2016-0978-0011), Severance Hospital (4-2016-0711), Ajou University Hospital (AJIRB-MED-CT4-16-202), and Chonbuk National University Hospital (CUH 2016-06-026-015). This trial has been registered with the Clinical Research Information Service (CRIS), Republic of Korea (KCT0002260), and ClinicalTrial.gov (NCT02981121). All subjects will provide written informed consent for participation.
METHODS
- Outcome measures
- The primary endpoint of this study is the cumulative incidence of diabetes for 3 years after randomization. The incidence of diabetes is defined as fasting glucose ≥126 mg/dL, postprandial 2-hour glucose ≥200 mg/dL after a 75-g oral glucose tolerance test, or HbA1c ≥6.5%. However, when the fasting glucose criterion is satisfied, the test will be repeated to confirm diabetes. After a subject is diagnosed with diabetes, standard therapy will be performed according to KDA clinical practice recommendations [21]. Diabetes treatment will be individualized according to patient characteristics. The clinical course of a subject with diabetes will be monitored over the course of the scheduled observation period.
- The secondary endpoints of the study are HbA1c at 1, 2, and 3 years after baseline; fasting blood glucose at 1, 2, and 3 years after baseline; insulin sensitivity index (homeostasis model assessment 2 [HOMA2] %S) at 1, 2, and 3 years after baseline; and insulin secretion index (HOMA2 %B) at 1, 2, and 3 years after baseline [22]. Additional analyses will be performed to determine differences in cardiovascular risk factors, diabetes risk factors, and adherence to the different interventions.
- After study completion, a further extension of the study will be conducted to identify the long-term outcomes of the subjects based on public healthcare data, such as insurance claims data, death certificates, and cancer registries, using the personal identifiers of the collected subjects.
- Timeline
- Recruitment of participants began in November 2016, and enrollment is expected to be completed by October 2018. The total observation period for each subject is planned to be 36 months.
- Sample size
- The number of enrolled subjects was calculated based on the only previous Asian randomized trial, the Indian Diabetes Prevention Program (IDPP) [23]. In this study, the cumulative incidence of diabetes at 3 years was 55.0% for control, 39.3% for LSM group, and 40.5% for Met group [23]. However, the prevalence of diabetes in IDPP is exaggerated in comparison to previous epidemiologic studies in Koreans [2425]. In order to overcome this problem, we performed conservative sample size estimation using Bonferroni correction considering multiple comparisons between the groups.
- A corrected significance level of 5% and a power of 80% yielded a total of 221 subjects per group. A total of two analyses, including one interim analysis, will be conducted for the major endpoints of this study. RB (2, 0.05, 0.2)=1.008 was applied to each group, and the dropout rate was assumed to be 10%. Therefore, 248 patients per group will be recruited for clinical research.
- Data collection and management
- Regular workshops for H-KDPS researchers and coordinators and twice-yearly additional training sessions will be conducted to ensure standardized interventions that are unaffected by participating organizations. Clinical information on the subjects will be collected and managed using the iCReaT system developed by the Korea National Institute of Health (http://icreat.nih.go.kr). The quality of the collected data will be confirmed through periodic audits.
- Biospecimens
- Biospecimens will be collected from subjects according to the standard operating procedures of the Korean National Health and Nutrition Examination and processed and stored in a central laboratory (Seegene Medical Foundation, Seoul, Korea) [26]. The baseline laboratory parameters are HbA1c, fasting plasma glucose, 75-g oral glucose tolerance test (0, 30, 60, 90, and 120 minutes glucose; 0 and 30 minutes insulin and C-peptide), and blood chemistry, including blood urea nitrogen, creatinine, aspartate aminotransferase, alanine aminotransferase, γ-glutamyl transferase, total cholesterol, triglycerides, high density lipoprotein cholesterol, and low density lipoprotein cholesterol. Additional biospecimens for genomics and novel biomarker-related studies will be collected and stored.
- Statistical analysis
- The results of this study will be validated by independent committee members of KDA. Evaluations of the validity and safety of the intervention will be based on the full data set. Interim analyses will be performed to compare the baseline characteristics of and treatment effects in the three groups with regard to primary endpoints and to consider early termination of the study.
- For primary endpoint evaluation, we will present the cumulative incidence of diabetes for 3 years after randomization. Differences among the groups will be presented as Kaplan-Meier curves. The descriptive statistics for changes in the main clinical variables at 1, 2, and 3 years after baseline will be presented and compared among the three groups using analysis of variance (ANOVA) or the Kruskal-Wallis test, depending on the distribution of the data. Additionally, paired t-tests or Wilcoxon's signed rank tests will be used for within-group comparisons. Categorical data will be tested using the chi-square test or Fisher's exact test. Risk factors related to cardiovascular disease and diabetes will be analyzed using the Cox proportional hazards model. Univariate analysis and multivariate analysis using logistic regression will be performed for each DM risk factor.
- The incidence of all adverse events that occur during the clinical trial, of adverse events that result in dropout, and of significant adverse events will be presented. The chi-square test or Fisher's exact test will be used to compare the three groups.
RESULTS
- The personal and social burdens of diabetes and its complications are increasing, with adverse effects on the stability of the public health system. According to the 2014 Korean national death statistics, diabetes was the sixth most common cause of death [27]. Additionally, diabetes is a closely related to cancer, cardiovascular, and cerebrovascular diseases, which are the top three major causes of death in Koreans. Recently, a study using health insurance data determined that diabetes was associated with the highest disease burden in Korea [28].
- The increase in diabetes patients is closely related to the increase in those at high risk for diabetes (i.e., prediabetic subjects). Non-diabetic hyperglycemia that does not satisfy the diagnostic criteria for DM is generally known as pre-diabetes mellitus (preDM) [2930]. The preDM stage is a period characterized by the dynamic pathophysiological changes associated with increased insulin resistance and reduced insulin secretion, although the clinical symptoms are not significant [3132]. It is generally known that 5% to 10% of preDM subjects develop DM every year [3033]. The prevalence of preDM in Korea is estimated at approximately 25% of adults over 30 years of age [2].
- At the preDM stage, aggressive intervention can prevent diabetes and improve the long-term prognosis. Several key clinical studies on diabetes prevention have been published. Although there were differences in the characteristics of the subjects and the methods of intervention in each study, they confirmed that diabetic incidence was significantly reduced through LSM or medication [567891011]. Notably, the intervention effect continued even after the end of the study, which had a significant effect on the long-term outcomes of subjects [1213141516]. The diabetes prevention program is economical and is considered to improve the stability of the national healthcare system [3435].
- Based on these findings, some countries have implemented diabetes prevention programs as major public health policies [3637]. In recent years, the incidence of diabetes in the United States has decreased, suggesting that the spread of diabetes prevention programs may have partially contributed to this outcome [38]. In the United States, an increasing number of diabetes prevention programs are being implemented, reflecting the benefits of such programs [37]. In some countries, such as the United Kingdom, the use of metformin has been reflected in insurance benefits to prevent diabetes [39].
- For successful diabetes prevention, an individualized intervention strategy should be developed that reflects each country's unique social and cultural characteristics; then, a well-designed clinical trial should be conducted to establish solid evidence. However, a reliable clinical trial for diabetes prevention in Korea has not yet been conducted. For this reason, a KDA project group for studying diabetes prevention has been organized and is conducting the H-KDPS and C-KDPS studies. The H-KDPS, in particular, is expected to produce representative evidence in Korea due to the participation of many KDA experts in all areas of study design, protocol development, research execution, data monitoring, data analysis, and consulting.
- The H-KDPS study has some important differences from previously conducted diabetes prevention trials. First, it is designed to enable long-term outcome observation by linking the clinical information of the subjects with public data (e.g., health insurance claims, cancer registries, and death statistics) even after the intervention period, which is scheduled for 36 months. It is expected that the long-term clinical outcomes of the subjects will be ascertained in greater detail than in previous studies owing to the advantage of the National Health Insurance system in Korea.
- Second, the study was designed to enable various biomarker studies in addition to the primary endpoints as biospecimens will be collected via the quality management system of the Korea National Health and Nutrition Survey. In the future, these biospecimens are likely to be used as valuable resources for understanding the pathophysiology of preDM and early-stage diabetes in Korea. Third, the study was designed to provide digital activity trackers for the lifestyle intervention group to investigate the clinical utility of digital healthcare devices for diabetes prevention. This will help establish objective evidence regarding new technologies that will be widely used in chronic disease interventions in the future.
- The results of the KDPS study will be used to establish the Korea Diabetes Prevention Program (KDPP), which is a unique diabetes prevention program in Korea. KDPP, which reflects the unique characteristics of Koreans and the Korean medical system, will contribute to the effective implementation of diabetes prevention programs in diverse environments, such as communities, public health centers, and medical clinics. Successful KDPP operations will be used as important evidence supporting the nationalization of diabetes prevention interventions in Korea. To achieve this goal, the KDA project group for DPS will continue its efforts.
DISCUSSION
-
Acknowledgements
- The following are the major public institutions that are helping to conduct the H-KDPS: Coordinating Center for National Healthcare Clinical Research, National Evidence-based Healthcare Collaborating Agency (NECA); Korea Centers for Disease Control & Prevention; Korea Association of Health Promotion; Dongdaemun-gu Public Health Center; and Songpa-gu Public Health Center.
- This research was supported by a grant from the Korean Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HC15C3364).
- The full list of all participating university hospitals and contributors is as follows: Korean Diabetes Prevention Study.
- Moon-Kyu Lee (Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul), Kyong Soo Park (Department of Internal Medicine, Seoul National University College of Medicine, Seoul), Bong Soo Cha (Department of Internal Medicine, Yonsei University College of Medicine, Seoul), Sei-Hyun Baik (Division of Endocrinology and Metabolism, Department of Internal Medicine, Korea University Medical Center, Korea University College of Medicine, Seoul), Joong Yeol Park (Department of Internal Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul), Kwan Woo Lee (Department of Endocrinology and Metabolism, Ajou University School of Medicine, Suwon), In-Kyu Lee (Division of Endocrinology and Metabolism, Department of Internal Medicine, Kyungpook National University School of Medicine, Daegu), Tae Sun Park (Department of Internal Medicine, Chonbuk National University Hospital, Chonbuk National University Medical School, Jeonju), In Joo Kim (Department of Nuclear Medicine and Biomedical Research Institute, Pusan National University Hospital, Pusan National University School of Medicine, Busan), Jung Min Lee (Department of Nutrition Care, Severance Hospital, Yonsei University College of Medicine, Seoul), Jung Hwa Lee (Nutrition Team, Kyung Hee University Medical Center, Kyung Hee University School of Medicine, Seoul), Jeong Hyun Lim (Department of Food Service and Nutrition Care, Seoul National University Hospital, Seoul National University College of Medicine, Seoul), Jae Won Cho (Department of Dietetics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul), Meera Kweon (Department of Food Service and Nutrition Care, Seoul National University Hospital, Seoul National University College of Medicine, Seoul), Ji-Hyun Lee (Food Service and Clinical Nutrition Team, Ajou University Hospital, Ajou University School of Medicine, Suwon), Young Jung Cho (Department of Internal Medicine, National Medical Center, Seoul), Jee-Hyun Lee (Department of Social Work, Gangnam Severance Hospital, Yonsei University College of Medicine, Seoul), Sung-Chul Lim (Department of Social Work, Kyung Hee University Medical Center, Kyung Hee University School of Medicine, Seoul), Jin Sun Choi (Department of Dietetic, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul), Jeongim Hong (Department of Nutrition, Yeouido St. Mary's Hospital, College of Medicine, The Catholic University of Korea, Seoul), Hyun Jung Lim (Department of Medical Nutrition, Kyung Hee University Graduate School of East-West Medical Science, Seoul), Dong-Hyeok Cho (Division of Endocrinology and Metabolism, Department of Internal Medicine, Chonnam National University Hospital, Chonnam National University Medical School, Gwangju), Sung Wan Chun (Department of Internal Medicine, Soonchunhyang University Cheonan Hospital, Soonchunhyang University College of Medicine, Cheonan), So Hun Kim (Department of Internal Medicine, Inha University School of Medicine, Incheon), Seo Young Sohn (Division of Endocrinology, Department of Medicine, Myongji Hospital, Goyang), Hyeok Lee (Nutrition Team, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul), You Cheol Hwang (Department of Endocrinology and Metabolism, Kyung Hee University Hospital at Gangdong, Kyung Hee University School of Medicine, Seoul), Justin Y. Jeon (Department of Family Medicine, Yonsei University College of Medicine, Seoul), Mi Kyoung Lee (Department of Sport Industry Studies, Exercise Medicine Center for Diabetes and Cancer Patients, Yonsei University, Seoul), Eunjeong Kang (Department of Endocrinology and Metabolism, Kyung Hee University Hospital, Kyung Hee University School of Medicine, Seoul), and Jihye Choi (Department of Endocrinology and Metabolism, Kyung Hee University Hospital, Kyung Hee University School of Medicine, Seoul).
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
NOTES
- 1. Kim DJ. The epidemiology of diabetes in Korea. Diabetes Metab J 2011;35:303-308. ArticlePubMedPMC
- 2. Noh J. The diabetes epidemic in Korea. Endocrinol Metab (Seoul) 2016;31:349-353. ArticlePubMedPMCPDF
- 3. Bommer C, Heesemann E, Sagalova V, Manne-Goehler J, Atun R, Barnighausen T, Vollmer S. The global economic burden of diabetes in adults aged 20-79 years: a cost-of-illness study. Lancet Diabetes Endocrinol 2017;5:423-430. ArticlePubMed
- 4. Lee KW. Costs of diabetes mellitus in Korea. Diabetes Metab J 2011;35:567-570. ArticlePubMedPMC
- 5. Pan XR, Li GW, Hu YH, Wang JX, Yang WY, An ZX, Hu ZX, Lin J, Xiao JZ, Cao HB, Liu PA, Jiang XG, Jiang YY, Wang JP, Zheng H, Zhang H, Bennett PH, Howard BV. Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study. Diabetes Care 1997;20:537-544. ArticlePubMedPDF
- 6. Tuomilehto J, Lindstrom J, Eriksson JG, Valle TT, Hamalainen H, Ilanne-Parikka P, Keinanen-Kiukaanniemi S, Laakso M, Louheranta A, Rastas M, Salminen V, Uusitupa M. Finnish Diabetes Prevention Study Group. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med 2001;344:1343-1350. ArticlePubMed
- 7. Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, Nathan DM. Diabetes Prevention Program Research Group. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med 2002;346:393-403. ArticlePubMedPMC
- 8. Chiasson JL, Josse RG, Gomis R, Hanefeld M, Karasik A, Laakso M. STOP-NIDDM Trail Research Group. Acarbose for prevention of type 2 diabetes mellitus: the STOP-NIDDM randomised trial. Lancet 2002;359:2072-2077. ArticlePubMed
- 9. DREAM (Diabetes REduction Assessment with ramipril and rosiglitazone Medication) Trial Investigators. Gerstein HC, Yusuf S, Bosch J, Pogue J, Sheridan P, Dinccag N, Hanefeld M, Hoogwerf B, Laakso M, Mohan V, Shaw J, Zinman B, Holman RR. Effect of rosiglitazone on the frequency of diabetes in patients with impaired glucose tolerance or impaired fasting glucose: a randomised controlled trial. Lancet 2006;368:1096-1105. ArticlePubMed
- 10. Kawamori R, Tajima N, Iwamoto Y, Kashiwagi A, Shimamoto K, Kaku K. Voglibose Ph-3 Study Group. Voglibose for prevention of type 2 diabetes mellitus: a randomised, double-blind trial in Japanese individuals with impaired glucose tolerance. Lancet 2009;373:1607-1614. ArticlePubMed
- 11. DeFronzo RA, Tripathy D, Schwenke DC, Banerji M, Bray GA, Buchanan TA, Clement SC, Henry RR, Hodis HN, Kitabchi AE, Mack WJ, Mudaliar S, Ratner RE, Williams K, Stentz FB, Musi N, Reaven PD. ACT NOW Study. Pioglitazone for diabetes prevention in impaired glucose tolerance. N Engl J Med 2011;364:1104-1115. ArticlePubMed
- 12. Li G, Zhang P, Wang J, Gregg EW, Yang W, Gong Q, Li H, Li H, Jiang Y, An Y, Shuai Y, Zhang B, Zhang J, Thompson TJ, Gerzoff RB, Roglic G, Hu Y, Bennett PH. The long-term effect of lifestyle interventions to prevent diabetes in the China Da Qing Diabetes Prevention Study: a 20-year follow-up study. Lancet 2008;371:1783-1789. ArticlePubMed
- 13. Gong Q, Gregg EW, Wang J, An Y, Zhang P, Yang W, Li H, Li H, Jiang Y, Shuai Y, Zhang B, Zhang J, Gerzoff RB, Roglic G, Hu Y, Li G, Bennett PH. Long-term effects of a randomised trial of a 6-year lifestyle intervention in impaired glucose tolerance on diabetes-related microvascular complications: the China Da Qing Diabetes Prevention Outcome Study. Diabetologia 2011;54:300-307. ArticlePubMedPDF
- 14. Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the Diabetes Prevention Program Outcomes Study. Lancet Diabetes Endocrinol 2015;3:866-875. ArticlePubMedPMC
- 15. Diabetes Prevention Program Research Group. Knowler WC, Fowler SE, Hamman RF, Christophi CA, Hoffman HJ, Brenneman AT, Brown-Friday JO, Goldberg R, Venditti E, Nathan DM. 10-Year follow-up of diabetes incidence and weight loss in the Diabetes Prevention Program Outcomes Study. Lancet 2009;374:1677-1686. ArticlePubMedPMC
- 16. Li G, Zhang P, Wang J, An Y, Gong Q, Gregg EW, Yang W, Zhang B, Shuai Y, Hong J, Engelgau MM, Li H, Roglic G, Hu Y, Bennett PH. Cardiovascular mortality, all-cause mortality, and diabetes incidence after lifestyle intervention for people with impaired glucose tolerance in the Da Qing Diabetes Prevention Study: a 23-year follow-up study. Lancet Diabetes Endocrinol 2014;2:474-480. ArticlePubMed
- 17. Baker MK, Simpson K, Lloyd B, Bauman AE, Singh MA. Behavioral strategies in diabetes prevention programs: a systematic review of randomized controlled trials. Diabetes Res Clin Pract 2011;91:1-12. ArticlePubMed
- 18. Ackermann RT, Marrero DG. Adapting the Diabetes Prevention Program lifestyle intervention for delivery in the community: the YMCA model. Diabetes Educ 74;33:6974-75. 77-78. ArticlePDF
- 19. Chan AW, Tetzlaff JM, Gotzsche PC, Altman DG, Mann H, Berlin JA, Dickersin K, Hrobjartsson A, Schulz KF, Parulekar WR, Krleza-Jeric K, Laupacis A, Moher D. SPIRIT 2013 explanation and elaboration: guidance for protocols of clinical trials. BMJ 2013;346:e7586. ArticlePubMedPMC
- 20. Cha SA, Lim SY, Kim KR, Lee EY, Kang B, Choi YH, Yoon KH, Ahn YB, Lee JH, Ko SH. Community-based randomized controlled trial of diabetes prevention study for high-risk individuals of type 2 diabetes: lifestyle intervention using web-based system. BMC Public Health 2017;17:387ArticlePubMedPMCPDF
- 21. Korean Diabetes Association. 2015 Treatment Guideline for Diabetes updated 2016 Oct 12. Available from: http://www.diabetes.or.kr/pro/publish/guide.php?code=guide&number=638&mode=view.
- 22. Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care 2004;27:1487-1495. ArticlePubMedPDF
- 23. Ramachandran A, Snehalatha C, Mary S, Mukesh B, Bhaskar AD, Vijay V. Indian Diabetes Prevention Programme (IDPP). The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1). Diabetologia 2006;49:289-297. ArticlePubMedPDF
- 24. Um HD, Lee DC, Lee SY, Kim YS. A prospective cohort study of exercise and the incidence of type 2 diabetes in impaired fasting glucose group. J Prev Med Public Health 2008;41:45-50. ArticlePubMed
- 25. Han SJ, Kim HJ, Kim DJ, Lee KW, Cho NH. Incidence and predictors of type 2 diabetes among Koreans: a 12-year follow up of the Korean Genome and Epidemiology Study. Diabetes Res Clin Pract 2017;123:173-180. ArticlePubMed
- 26. Kweon S, Kim Y, Jang MJ, Kim Y, Kim K, Choi S, Chun C, Khang YH, Oh K. Data resource profile: the Korea National Health and Nutrition Examination Survey (KNHANES). Int J Epidemiol 2014;43:69-77. ArticlePubMedPMC
- 27. Shin HY, Lee JY, Song J, Lee S, Lee J, Lim B, Kim H, Huh S. Cause-of-death statistics in the Republic of Korea, 2014. J Korean Med Assoc 2016;59:221-232.ArticlePDF
- 28. Yoon J, Oh IH, Seo H, Kim EJ, Gong YH, Ock M, Lim D, Lee WK, Lee YR, Kim D, Jo MW, Park H, Yoon SJ. Disability-adjusted life years for 313 diseases and injuries: the 2012 Korean Burden of Disease Study. J Korean Med Sci 2016;31(Suppl 2):S146-S157. ArticlePubMedPMCPDF
- 29. Rhee SY, Woo JT. The prediabetic period: review of clinical aspects. Diabetes Metab J 2011;35:107-116. ArticlePubMedPMC
- 30. Tabak AG, Herder C, Rathmann W, Brunner EJ, Kivimaki M. Prediabetes: a high-risk state for diabetes development. Lancet 2012;379:2279-2290. ArticlePubMedPMC
- 31. Rhee SY, Kim JY, Chon S, Hwang YC, Jeong IK, Oh S, Ahn KJ, Chung HY, Woo JT, Kim SW, Kim JW, Kim YS. The changes in early phase insulin secretion in newly diagnosed, drug naive korean prediabetes subjects. Korean Diabetes J 2010;34:157-165. ArticlePubMedPMC
- 32. Rhee SY, Woo JT, Chon S, Hwang YC, Oh S, Ahn KJ, Chung HY, Kim SW, Kim JW, Kim YS. Characteristics of insulin resistance and insulin secretory capacity in Korean subjects with IFG and IGT. Diabetes Res Clin Pract 2010;89:250-255. ArticlePubMed
- 33. Forouhi NG, Luan J, Hennings S, Wareham NJ. Incidence of type 2 diabetes in England and its association with baseline impaired fasting glucose: the Ely study 1990–2000. Diabet Med 2007;24:200-207. ArticlePubMed
- 34. Icks A, Rathmann W, Haastert B, Gandjour A, Holle R, John J, Giani G. KORA Study Group. Clinical and cost-effectiveness of primary prevention of type 2 diabetes in a ‘real world’ routine healthcare setting: model based on the KORA Survey 2000. Diabet Med 2007;24:473-480. ArticlePubMed
- 35. Diabetes Prevention Program Research Group. The 10-year cost-effectiveness of lifestyle intervention or metformin for diabetes prevention: an intent-to-treat analysis of the DPP/DPPOS. Diabetes Care 2012;35:723-730. PubMedPMC
- 36. Ackermann RT, Liss DT, Finch EA, Schmidt KK, Hays LM, Marrero DG, Saha C. A randomized comparative effectiveness trial for preventing type 2 diabetes. Am J Public Health 2015;105:2328-2334. ArticlePubMedPMC
- 37. Mensa-Wilmot Y, Bowen SA, Rutledge S, Morgan JM, Bonner T, Farris K, Blacher R, Rutledge G. Early results of states' efforts to support, scale, and sustain the National Diabetes Prevention Program. Prev Chronic Dis 2017;14:E130ArticlePubMedPMC
- 38. Selvin E, Ali MK. Declines in the incidence of diabetes in the U.S.: real progress or artifact? Diabetes Care 2017;40:1139-1143. ArticlePubMedPMCPDF
- 39. Halliday M. First drug approved for diabetes prevention updated 2017 May 22. Available from: https://www.mims.co.uk/first-drug-approved-diabetes-prevention/diabetes/article/1434274.
REFERENCES
Table 1
![dmj-43-49-i001.jpg]()
Intervention schedule at a glance: conventional management (CM) group
| Visit date | Screening | Baseline | Intervention | |||||
|---|---|---|---|---|---|---|---|---|
| −28 day | 0 day | 6 mo | 12 mo | 18 mo | 24 mo | 30 mo | 36 mo | |
| Visit window | ±7 day | |||||||
| Education and evaluation | ||||||||
| Conventional educationa | √ | |||||||
Table 2
![dmj-43-49-i002.jpg]()
Intervention schedule at a glance: lifestyle modification (LSM) group
Table 3
![dmj-43-49-i003.jpg]()
Intervention schedule at a glance: metformin (Met) group
Figure & Data
References
Citations
Citations to this article as recorded by 

- Estimating insulin sensitivity and β-cell function from the oral glucose tolerance test: validation of a new insulin sensitivity and secretion (ISS) model
Joon Ha, Stephanie T. Chung, Max Springer, Joon Young Kim, Phil Chen, Aaryan Chhabra, Melanie G. Cree, Cecilia Diniz Behn, Anne E. Sumner, Silva A. Arslanian, Arthur S. Sherman
American Journal of Physiology-Endocrinology and Metabolism.2024; 326(4): E454. CrossRef - Development and Adaptability of Smartphone-based Dietary Coaching Program for Patients Undergoing Diabetes and Prediabetes with Continuous Glucose Monitoring Device
Myoung Soo Kim, Jung Mi Ryu, Minkyeong Kang, Jiwon Park, Yeh Chan Ahn, Yang Seok Kim
Journal of Health Informatics and Statistics.2023; 48(1): 36. CrossRef - Improving Machine Learning Diabetes Prediction Models for the Utmost Clinical Effectiveness
Juyoung Shin, Joonyub Lee, Taehoon Ko, Kanghyuck Lee, Yera Choi, Hun-Sung Kim
Journal of Personalized Medicine.2022; 12(11): 1899. CrossRef - Impaired fasting glucose levels in overweight or obese subjects for screening of type 2 diabetes in Korea
Jin-Hee Lee, Suk Chon, Seon-Ah Cha, Sun-Young Lim, Kook-Rye Kim, Jae-Seung Yun, Sang Youl Rhee, Kun-Ho Yoon, Yu-Bae Ahn, Jeong-Taek Woo, Seung-Hyun Ko
The Korean Journal of Internal Medicine.2021; 36(2): 382. CrossRef - Development and Validation of a Deep Learning Based Diabetes Prediction System Using a Nationwide Population-Based Cohort
Sang Youl Rhee, Ji Min Sung, Sunhee Kim, In-Jeong Cho, Sang-Eun Lee, Hyuk-Jae Chang
Diabetes & Metabolism Journal.2021; 45(4): 515. CrossRef - 2021 Clinical Practice Guidelines for Diabetes Mellitus of the Korean Diabetes Association
Kyu Yeon Hur, Min Kyong Moon, Jong Suk Park, Soo-Kyung Kim, Seung-Hwan Lee, Jae-Seung Yun, Jong Ha Baek, Junghyun Noh, Byung-Wan Lee, Tae Jung Oh, Suk Chon, Ye Seul Yang, Jang Won Son, Jong Han Choi, Kee Ho Song, Nam Hoon Kim, Sang Yong Kim, Jin Wha Kim,
Diabetes & Metabolism Journal.2021; 45(4): 461. CrossRef - Short-Term Effects of the Internet-Based Korea Diabetes Prevention Study: 6-Month Results of a Community-Based Randomized Controlled Trial
Jin-Hee Lee, Sun-Young Lim, Seon-Ah Cha, Chan-Jung Han, Ah Reum Jung, Kook-Rye Kim, Kun-Ho Yoon, Seung-Hyun Ko
Diabetes & Metabolism Journal.2021; 45(6): 960. CrossRef - 2021 Clinical Practice Guidelines for Diabetes Mellitus in Korea
Seung-Hyun Ko
The Journal of Korean Diabetes.2021; 22(4): 244. CrossRef - Optimal fasting plasma glucose and haemoglobin A1c levels for screening of prediabetes and diabetes according to 2‐hour plasma glucose in a high‐risk population: The Korean Diabetes Prevention Study
Seon‐Ah Cha, Suk Chon, Jae‐Seung Yun, Sang Youl Rhee, Sun‐Young Lim, Kun‐Ho Yoon, Yu‐Bae Ahn, Seung‐Hyun Ko, Jeong‐Taek Woo, Jin‐Hee Lee
Diabetes/Metabolism Research and Reviews.2020;[Epub] CrossRef - How was the Diabetes Metabolism Journal added to MEDLINE?
Hye Jin Yoo
Science Editing.2020; 7(2): 201. CrossRef - Commercial Postural Devices: A Review
Nicole Kah Mun Yoong, Jordan Perring, Ralph Jasper Mobbs
Sensors.2019; 19(23): 5128. CrossRef - Changes in Metabolic Profile Over Time: Impact on the Risk of Diabetes
Yunjung Cho, Seung-Hwan Lee
Diabetes & Metabolism Journal.2019; 43(4): 407. CrossRef - Metformin for prevention or delay of type 2 diabetes mellitus and its associated complications in persons at increased risk for the development of type 2 diabetes mellitus
Kasper S Madsen, Yuan Chi, Maria-Inti Metzendorf, Bernd Richter, Bianca Hemmingsen
Cochrane Database of Systematic Reviews.2019;[Epub] CrossRef

 KDA
KDA

 PubReader
PubReader Cite
Cite